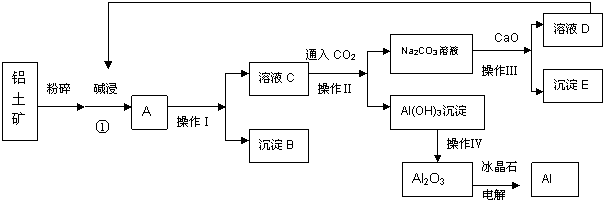
��Ŀ����
12����ҵ���������β���к��е�������NOx��NO��NO2�Ļ������費��N2O4��������̬���������ཡ�������ϴ����в����1����ҵ�Ͽ��ð������շ�����NOx����Ӧԭ�����£�4xNH3+6NOx$\frac{\underline{\;����\;}}{\;}$��2x+3��N2+6xH2O��ij��ѧ��ȤС��ģ��ô������̵�ʵ��װ�����£�
��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ2NH4Cl+Ca��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$CaCl2+2NH3��+2H2O��
��װ��D�м�ʯ�ҵ������dz�ȥ�����к��е�ˮ������
��2����ҵ��Ҳ����Na2CO3��Һ���շ�����NOx��
��֪��NO������Na2CO3��Һ��Ӧ��
NO+NO2+Na2CO3�T2NaNO2+CO2����
2NO2+Na2CO3�TNaNO2+NaNO3+CO2����
�ٵ�NOx��Na2CO3��Һ��ȫ����ʱ��x��ֵ��������C������ĸ����
A��1.9 B��1.7 C�� 1.2
�ڽ�1mol NOxͨ��Na2CO3��Һ�У�����ȫ����ʱ����Һ�����ɵ�NO3-��NO2-�������ӵ����ʵ�����x�仯��ϵ��ͼ2��ʾ��
ͼ���߶�a��ʾNO2-�����ʵ�����xֵ�仯�Ĺ�ϵ������������������Ϊ42.4%�� Na2CO3��Һ���գ�����ҪNa2CO3��Һ����125g��
����������Na2CO3��Һ��ȫ����NOx��ÿ����22.4L����״����CO2��ȫ���ݳ���ʱ������Һ����������44g����NOx�е�xֵΪ1.875��
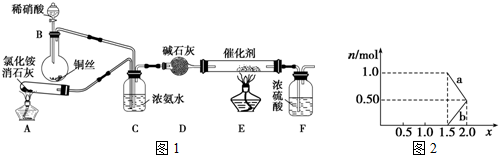
���� ��1��A����ʯ�����Ȼ���ڼ��������·�Ӧ���ɰ�����B��ϡ������ͭ��Ӧ����NO��������NO��C���ӡ�D�������E�д������·�Ӧ���ɵ�����FΪ����װ�ã�
���ڼ��������£��Ȼ�狀��������Ʒ�Ӧ�����Ȼ��ơ�������ˮ��
�ڰ������ڼ������壬���ü������ʸ��
��2���ٵ�NOx��Na2CO3��Һ��ȫ����ʱ����n��NO2����n��NO����
�����ü������غ㷨���������
�����ò���������NO��NO2�����ʵ������ٸ���ƽ��Ħ������������xֵ��
��� �⣺��1��A����ʯ�����Ȼ���ڼ��������·�Ӧ���ɰ�����B��ϡ������ͭ��Ӧ����NO��������NO��C���ӡ�D�������E�д������·�Ӧ���ɵ�����FΪ����װ�ã�
���ڼ��������£��Ȼ�狀��������Ʒ�Ӧ�����Ȼ��ơ�������ˮ��2NH4Cl+Ca��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$CaCl2+2NH3��+2H2O��
�ʴ�Ϊ��2NH4Cl+Ca��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$CaCl2+2NH3��+2H2O��
�ڼ�ʯ��������ˮ�ֶ�����������������ڼ������壬�������ü�ʯ�Ҹ��
�ʴ�Ϊ����ȥ�����к��е�ˮ������
��2���ٵ�NOx��Na2CO3��Һ��ȫ����ʱ����n��NO2����n��NO������n��NO2����n��NO��=1ʱxֵ��С��x��СֵΪ$\frac{2+1}{2}$=1.5����Ϊ����NO������x���ֵ��2����x��ȡֵ��ΧΪ1.5��x��2������x��ֵ��������1.2��
�ʴ�Ϊ��C��
���ü�������x=1.5����ӦΪNO��NO2�������ʵ�����Ϊ1��1������ʽ��Ӧ��û��NO3-����aӦ�ñ�ʾNO2-��
���غ㷨����Ӧ���ɵ�NaNO3��NaNO2�е�Ԫ������Ԫ��֮��Ϊ1��1������1mol NOx����ȫ����������̼����0.5mol������Ϊ53g�������̼������Һ������Ϊ$\frac{53g}{42.4%}$=125g��
�ʴ�Ϊ��NO2-��125��
������NO2�ʹ��Ӧ����CO2Ϊamol��
��NO��NO2�봿�Ӧ������CO2Ϊbmol��
2NO2+Na2CO3=NaNO2+NaNO3+CO2 ��������
1mol��m=48g
amol 48ag
NO+NO2+Na2CO3=2NaNO2+CO2 ��������
1mol��m=32g
bmol 32bg
$\left\{\begin{array}{l}{a+b=1}\\{48a+32b=44}\end{array}\right.$���$\left\{\begin{array}{l}{a=0.75mol}\\{b=0.25mol}\end{array}\right.$
n��NO2��=0.75mol��2+0.25mol=1.75mol
n��NO��=0.25mol
x=$\frac{0.25mol��1+1.75mol��2}{0.25mol+1.75mol}=\frac{15}{8}$=1.875��
�ʴ�Ϊ��1.875��
���� �����Ե�������Ϊ���忼�������ʼ�ķ�Ӧ��Ϊ�߿��������ͣ�������ѧ���ķ�����ʵ��ͼ��������Ŀ��飬��ȷ���ʵ������ǽⱾ��ؼ����ѵ��ǣ�2����ļ��㣬Ҫ��Ϸ���ʽ�и���������֮��Ĺ�ϵʽ��𣬻���������Ϣ����xֵ���Ѷ��еȣ�

��1����֪̼��������Ӧ�ڲ�ͬ�¶���ƽ�ⳣ���Ķ���ֵ��lgK�����±���
| ������Ӧʽ | 1gK | ||
| 700K | 900K | 1200K | |
| C��s��+H2O��g��=CO��g��+H2��g�� | -2.64 | -0.39 | 1.58 |
| C��s��+2H2O��g��=CO2��g��+2H2��g�� | -1.67 | -0.03 | 1.44 |
��2��ҵ�Ϻϳɼ״��ķ�ӦΪ��CO+2H2?CH3OH����֪��H2��g����CO��g����CH3OH��l����ȼ���ȡ�H�ֱ�Ϊ-285.8kJ/mol��-283.0kJ/mol��-726.5kJ/mol����CH3OH��l������ȫȼ������CO��Һ̬H2O���Ȼ�ѧ��Ӧ����ʽΪCH3OH��l��+O2��g��=CO��g��+2H2O��l������H=-443.5kJ/mol��
��3����һ���¶ȡ�ѹǿ�ʹ������£���ҵ����H2CO��CO�ϳ�CH3OCH3��
3H2��g��+3CO��g��?CH3OCH3��g��+CO2��g����H=-246.4KJ•mol-1
����һ�������µ��ܱ������У��÷�Ӧ�ﵽƽ�⣬ֻ�ı�һ��������ͬʱ��߷�Ӧ���ʺ�CO��ת���ʵ���cd������ĸ��ţ���
a�������¶� b��������� c����С������� d������H2��Ũ�� e������CO��Ũ��
����һ����ɱ���ܱ������г���3molH2��3molCO��1molCH3OCH3��1molCO2����һ���¶Ⱥ�ѹǿ�·���������Ӧ����һ��ʱ��ﵽƽ�⣬���ƽ��ʱ���������ܶ���ͬ��ͬѹ����ʼ��1.6������Ӧ��ʼʱ�����淴Ӧ���ʵĴ�С��v����v�棨�������������=������ƽ��ʱCO�����ʵ�������Ϊ15%��
��4��һ�������¿��ü״���CO��Ӧ���ɴ�������CO��Ⱦ�������£���amol•L-1�Ĵ�����bmol•L-1��Ba��OH��2��Һ�������ϣ���ַ�Ӧ����Һ�д���2c��Ba2+��=c��CH3COO-������û����Һ�д���ĵ��볣��Ka=$\frac{2b}{a-2b}$��10-7 mol•L-1���ú�a��b�Ĵ���ʽ��ʾ������仯���Բ��ƣ���
| A�� | �۵㣺Na��MgO��SiO2 | B�� | ˮ���ԣ�SO2��H2S��HCl | ||
| C�� | ���ȶ��ԣ�HF��H2O��NH3 | D�� | �е㣺HF��HCl��HBr |
��2���ڸ�����һ����̼�ɽ���������ԭΪ������
��C��s��+O2��g��=CO2��g����H1=-393.5kJ•mol
��CO2��g��+C��s��=2CO��g����H2=+172.5kJ•mol
��S��s��+O2��g��=SO2��g����H3=-296.0kJ•mol
��CO��SO2��Ӧ���Ȼ�ѧ����ʽ��2CO��g��+SO2��g��=CO2��g��+S��s����H=-270kJ/mol��
��3�������������Һ��ϡ���ᷴӦ�Ļ�ѧ����ʽΪNa2S2O3+H2SO4�TNa2SO4+SO2��+S��+H2O�����и���ʵ�������ȳ��ֻ��ǵ���D������ĸ���ţ���
| ʵ�� | ��Ӧ�¶�/�� | Na2S2O3��Һ | ϡH2SO4 | H2O | ||
| V/mL | c/��mol•L-1�� | V/mL | c/��mol•L-1�� | V/mL | ||
| A | 25 | 5 | 0.1 | 10 | 0.1 | 5 |
| B | 25 | 5 | 0.2 | 5 | 0.2 | 10 |
| C | 35 | 5 | 0.1 | 10 | 0.1 | 5 |
| D | 35 | 5 | 0.2 | 5 | 0.2 | 10 |
| A�� | ���۵��ɵ͵�������I2��CO2��Hg��KCl��SiO2 | |
| B�� | ��ʯӢ�����У�ÿ����ԭ�Ӻ����ڵ�������ԭ���Թ��ۼ���� | |
| C�� | �л��������̼ԭ�ӳɼ���ʽ�����з�ʽ�ж����Լ�ͬ���칹����Ĵ��ڶ����л���������ԭ�� | |
| D�� | ��Ϊʯī�ǽ��ʯ��ͬ�������壬���Զ��߿ռ�ṹ���ƣ���ѧ�������� |
 ��ʾ�ķ���ʽΪC6H14��������2-�����飮
��ʾ�ķ���ʽΪC6H14��������2-�����飮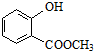 �к��еĹ����ŵ�����Ϊ�ǻ���������
�к��еĹ����ŵ�����Ϊ�ǻ���������