��Ŀ����
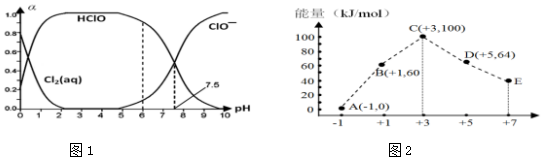
5�� ij������Ʒ�к��������Ȼ������ʣ�������ͼ��ʾװ�����ⶨ������Ʒ��̼���Ƶ���������������̨�����е���ͼ�о�����ȥ����ʵ�鲽�����£�
ij������Ʒ�к��������Ȼ������ʣ�������ͼ��ʾװ�����ⶨ������Ʒ��̼���Ƶ���������������̨�����е���ͼ�о�����ȥ����ʵ�鲽�����£��ٰ�ͼ����װ�ã�����������ԣ�
��ȷ�Ƶ�ʢ�м�ʯ�ң������������ƺ���ʯ�ҵĻ����ĸ����D������Ϊ85.4g��
��ȷ�Ƶ�6g������Ʒ��������b�У�
�ܴ�Һ©��a����������������ϡ���ᣬ�����ٲ�������Ϊֹ��
�ݴ��ɼУ����Թ�A�л���������������ӣ�Ȼ��Ƶø����D��������Ϊ87.6g���Իش�
��1�����ܢ�������ʵ�����̫�죬��ᵼ�²ⶨ���ƫС���ƫ��ƫС������
��2�����������Ŀ���ǽ�װ���ڲ����Ķ�����̼ȫ���ų����ﵽ��ȷ������Ŀ�ģ�װ��A���Լ�XӦѡ������������Һ��
��3����û��Cװ�ã���ᵼ�²ⶨ���ƫ���ƫ��ƫС������
��4��Eװ�õ������Ƿ�ֹ������CO2��ˮ��������D�ĸ�����У�
��5��װ��B�з�����Ӧ�����ӷ���ʽCO32-+2H+�TH2O+CO2����
��6������ʵ���в�õ��й����ݣ������������ƷNa2CO3����������Ϊ88.3%������������һλС������
���� ��ʵ����ͨ��̼���ƺ����ᷴӦ����������̼���ü�ʯ�������ն�����̼�����ݲ���������̼������������̼���Ƶ���������������������������ʵ�������Ӧ��ֹ������ˮ�Ͷ�����̼����ʵ�飬�Ѳ����Ķ�����̼ȫ������ʯ�����գ�
��1��������Ӧ�dz�������̼����Բⶨ�����Ӱ�죻
��2������װ�õ��ص㼰ʵ��Ŀ�ģ��������������һ������Ŀ�ĺ�װ��A�м�Һ�����ã�
��3����װ��C�����ÿ�֪�Բⶨ�����Ӱ�죻
��4�����Ը��ݿ����еijɷ�������װ��E������װ�����������ã�
��5���������εķ�Ӧ���ɣ���Ӧ��������д����Ӧ���ӷ���ʽ��
��6������ʵ��ǰ��װ��D�����仯�����ɴ���������������Ʒ��̼���Ƶ������������Ʒ��̼���Ƶ�����������
��� �⣺��1����Ӧ�����ʹ���������������̼û����ȫ��Dװ���м�ʯ�����գ����ٹ��������Ҳ��ʹװ���ڲ���������̼���ܱ�Dװ�ü�ʯ����ȫ���գ�������̼ƫС���ⶨ���ƫС���ʴ�Ϊ��ƫС��
��2���������֪������ͨ���ⶨ������̼���������ⶨ̼���Ƶ����������ģ�����Ҫ�ų������еĶ�����̼����ʵ����������A��װ�˼�����Һ�����տ����еĶ�����̼����װ��A���Լ���ѡ��NaOH���ʴ�Ϊ����װ���ڲ����Ķ�����̼ȫ���ų����ﵽ��ȷ������Ŀ�ģ�����������Һ��
��3��Ũ���������ˮ�ԣ���Cװ����������Bװ���ų������л��е�ˮ�����������˴�װ�����ʹ�����е�ˮ������Dװ���м�ʯ�����գ���ʹ�ⶨ����ƫ�ʴ�Ϊ��ƫ��
��4�����Dװ��ֱ��������������ͨ��������е�ˮ�����Ͷ�����̼�����Dװ�ö��Բⶨ�������Ӱ�죬����װ��E���������Ƿ�ֹ������ˮ�����Ͷ�����̼����װ��D�У��ʴ�Ϊ����ֹ������CO2��ˮ��������D�ĸ�����У�
��5���������εķ�Ӧ���ɣ���Ӧ��������д����Ӧ����ʽ��Na2CO3+H2SO4�TNa2SO4+H2O+CO2�������ӷ���ʽΪ��CO32-+2H+�TH2O+CO2����
�ʴ�Ϊ��CO32-+2H+�TH2O+CO2����
��6����Ӧ�зų�������̼������Ϊ��87.6g-85.4g=2.2g��
Na2CO3+H2SO4�TNa2SO4+H2O+CO2����
106g 44g
m 2.2g
m=$\frac{106g��2.2g}{44g}$=5.3g��������Ʒ��̼���Ƶ���������Ϊ��$\frac{5.3g}{6g}$��100%=88.3%���ʴ�Ϊ��88.3%��
���� ������Ҫ����̼���ƴ��ȵIJⶨ��ͨ����������Ҫ֪�������ʵ��ʱҪ�����ܵ��ų����ܶ�ʵ��������Ӱ������أ����籾���п����ж�����̼��ʵ������Ӱ�죬ˮ������Ӱ��ȣ��ѶȲ���

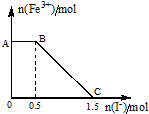
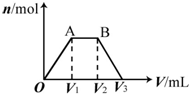 ��KOH��Ca��OH��2�Ļ����Һ��pH=13��ȡ����Һ500mL��������ͨ��CO2��ͨ��CO2�������V�������ɳ��������ʵ�����n���Ĺ�ϵ��ͼ��ʾ��ͼ�У�V1��Ϊ112mL����״��������ͼ��V2��V3���������λΪmL���ֱ��ǣ�������
��KOH��Ca��OH��2�Ļ����Һ��pH=13��ȡ����Һ500mL��������ͨ��CO2��ͨ��CO2�������V�������ɳ��������ʵ�����n���Ĺ�ϵ��ͼ��ʾ��ͼ�У�V1��Ϊ112mL����״��������ͼ��V2��V3���������λΪmL���ֱ��ǣ�������| A�� | 560��672 | B�� | 1008��1120 | C�� | 2240��2352 | D�� | 392��504 |
| A�� | ������Һɱ����Ӿ���е����� | |
| B�� | ��CaCl2�ڻ�·��Ļ�ѩ | |
| C�� | ��ʯ�����������ʴ������ | |
| D�� | �ô���������β���е�CO��NOת��Ϊ������ |
| ѡ�� | ʵ��Ŀ�� | ���� |
| A | ��ȥ���������е����� | ��NaOH��Һϴ�ӣ���Һ��Ȼ����� |
| B | ��ȥ��������Һ�е��Ҵ� | ��KMnO4������������ |
| C | ����ʳ�����Ƿ�����KIO3 | ʳ���ܽ⣬Ȼ����������Һ |
| D | �ⶨ������ҺŨ�� | ��̪��ָʾ�����ñ�NaOH��Һ�ζ� |
| A�� | A | B�� | B | C�� | C | D�� | D |

| A�� | ��a�Թ����ȼ���Ũ���ᣬȻ���ҡ���Թܱ����������Ҵ����ټӱ����� | |
| B�� | �Թ�b�е������¶˹ܿڲ��ܽ���Һ���ԭ���Ƿ�ֹʵ������з����������� | |
| C�� | ʵ��ʱ�����Թ�a��Ŀ���Ǽ�ʱ�����������������ӿ췴Ӧ���� | |
| D�� | �Թ�b��Na2CO3�������dz�ȥ��������������������������Ҵ�������������������Һ�е��ܽ�� |
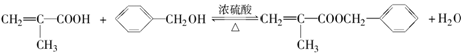 ��
��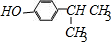 ��
�� ��1�����������嵥��Am��Bn����֪7.2g Am��6.3g Bn������ԭ�Ӹ�����ͬ�����Ӹ���֮��ȴΪ2��3����֪Aԭ����L���Ӳ㺬��������K���Ӳ��3����ͨ������ش�д��B��Ԫ�����Ƶ�
��1�����������嵥��Am��Bn����֪7.2g Am��6.3g Bn������ԭ�Ӹ�����ͬ�����Ӹ���֮��ȴΪ2��3����֪Aԭ����L���Ӳ㺬��������K���Ӳ��3����ͨ������ش�д��B��Ԫ�����Ƶ�