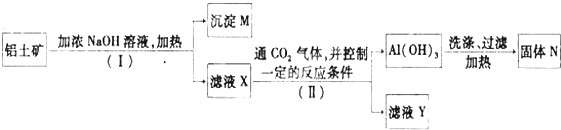
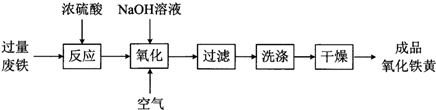
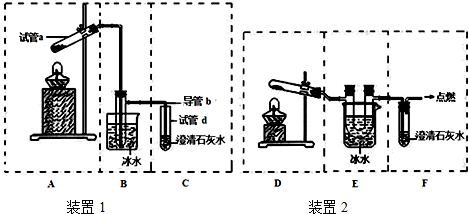
��Ŀ����
17����NH4��2Fe��SO4��2•6H2O��M=392g•mol-1���ֳ�Ī���Σ����FAS����������Դ�ڵ¹���ѧ��Ī����Karl Friedrich Mohr��������dz����ɫ�ᾧ���ĩ���Թ����У��ڿ����л��绯��������������ˮ�������������Ҵ���ijʵ��С�����ù�ҵ����м��ȡĪ���Σ����ⶨ�䴿�ȣ���Ī���ε���ȡ
�ش��������⣺
��1������ڱ�����ʣ��������мʱ���й��ˣ���ԭ���Ƿ�ֹFe2+��������
��2����ȡ��Ī�������Ҫ��bϴ�ӣ�����ĸ��ţ���
a������ˮ b���Ҵ� c����Һ
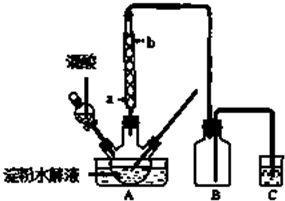
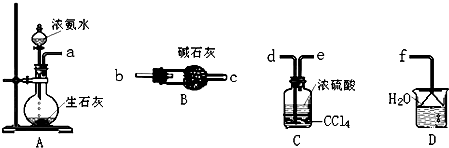
��3������ͼ��ѡȡ�����õ��������ӳ�ʵ������ȡ��NH4��2SO4��Һ��װ�ã����ӵ�˳���ýӿ���ĸ��ʾ����a��d��e��f��a��d��e��f��a��d��e��f��
��FAS���Ȳⶨ
Ϊ�ⶨFAS���ȣ�ȡm g��Ʒ���Ƴ�500mL��Һ������������ɣ��ס�����λͬѧ�������������ʵ�鷽����
��1��������ȡ20.00mLFAS��Һ����0.1000mol•L-1������K2Cr2O7��Һ���еζ���
i��д���˷�Ӧ�����ӷ���ʽ��6Fe2++Cr2O72-+14H+=6Fe3++2Cr3++7H2O��
ii�� �ζ�ʱ����ѡ�õ������Ǣ٢ܢݢޣ�

��2���ҷ�����ȡ20.00mLFAS��Һ��������ʵ�飮
����Һ$\stackrel{������BaCl_{2}��Һ}{��}$$\stackrel{����}{��}$$\stackrel{ϴ��}{��}$$\stackrel{����}{��}$$\stackrel{����}{��}$wg����
�г�����FAS���崿�ȵĴ���ʽ$\frac{392��25W}{2��233m}$��100%�����ü�����������
��3����֪ʵ���������ȷ��ȴ���ּ����IJⶨ�������С���ҷ���������ܵ�ԭ����Fe2+������������������ƼĻ�ѧʵ����֤�����Ʋ�ȡ����FAS��Һ����������KSCN��Һ������Һ��Ϊ��ɫ��˵��Fe2+�ѱ���������������
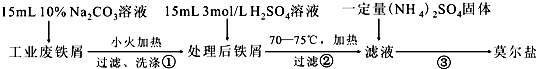
���� ����м��ϡ�����ܽ⣬�õ�����������ҺA��������茶��干ͬ�ܽ⣬��������Ũ������ȴ���塢���ˡ�ϴ�ӡ�����õ�Ī���Σ��Դ˷������
��1�����������ױ���������������ԭ�������ɵ�Fe3+��
��2���Ҵ��ӷ��������ƾ���ϴȥ����������ʣ�
��3����ȡ�İ��������ᷴӦ��������泥�ʣ��İ�����ˮ���գ�
��NH4��2Fe��SO4��2•6H2O��M=392g•mol-1���ֳ�Ī���Σ����FAS��
��1��i����ȡ20.00mLFAS��Һ����0.1000mol•L-1������K2Cr2O7��Һ���еζ���������������ԭ��Ӧ���������ӱ��ظ��������Ϊ�����ӣ����ɸ����ӣ�
ii����ע���ǵζ�ʱѡ�õ�����������������Һ��Ҫ����������Ӧ��ѡ����ʽ�ζ��ܡ���ƿ������̨�����У�
��2�����ݶ�����ϵ���㣬��NH4��2Fe��SO4��2•6H2O��2SO42-��2BaSO4��
��3��ʵ���������ȷ��ȴ���ּ����IJⶨ�������С���ҷ����������������ӱ�������������������������������KSCN��Һ���ɫ���ʵ������Ƿ�������
��� �⣺��1�����������ױ���������������ԭ�������ɵ�Fe3+����Ӧ�����ӷ���ʽΪFe+2Fe3+=3Fe2+���ʴ�Ϊ��Fe+2Fe3+=3Fe2+��
��2���Ҵ��ӷ����������ƾ���ϴȥ����������ʣ���ˮ��ʹĦ�����ܽ���ʧ������Һ��������ʣ����Ҵ����ȥ�������ˮ�֣����پ���ĸ����ֹ���������ʣ�
�ʴ�Ϊ��b��
��3����ȡ�İ��������ᷴӦ��������泥�ʣ��İ�����ˮ���գ�������˳��Ϊa��d��e��f��
�ʴ�Ϊ��a��d��e��f��a��d��e��f��a��d��e��f��
��NH4��2Fe��SO4��2•6H2O��M=392g•mol-1���ֳ�Ī���Σ����FAS��
��1��i����ȡ20.00mLFAS��Һ����0.1000mol•L-1������K2Cr2O7��Һ���еζ���������������ԭ��Ӧ���������ӱ��ظ��������Ϊ�����ӣ����ɸ����ӣ���Ӧ�����ӷ���ʽΪ��6Fe2++Cr2O72-+14H+=6Fe3++2Cr3++7H2O��
�ʴ�Ϊ��6Fe2++Cr2O72-+14H+=6Fe3++2Cr3++7H2O��
ii������K2Cr2O7��Һ����ǿ�����ԣ�Ӧ��ʹ�â���ʽ�ζ��ܣ�ʢװ����Һ��Ҫʹ�â���ƿ���ζ�����Ҫ�̶����õ�������̨��������
�ʴ�Ϊ���٢ܢݢޣ�
��2��ȡm g��Ʒ���Ƴ�500mL��Һ��ȡ20.00mLFAS��Һ����ʵ�飬�������ᱵ����Ϊwg�����ݶ�����ϵ���㣬
��NH4��2Fe��SO4��2•6H2O��2SO42-��2BaSO4 ��
1 2
n $\frac{w}{233}$mol
n=$\frac{w}{2��233}$mol��
��500mL��Һ��Ϊ$\frac{w}{2��233}$mol��$\frac{500}{20}$��
FAS���崿�ȵĴ���ʽ��$\frac{392��25W}{2��233m}$��100%��
�ʴ�Ϊ��$\frac{392��25W}{2��233m}$��100%��
��3����֪ʵ���������ȷ��ȴ���ּ����IJⶨ�������С���ҷ���������ܵ�ԭ����Fe2+������������������ƼĻ�ѧʵ����֤�����Ʋ��ʵ�����Ϊ��ȡ����FAS��Һ����������KSCN��Һ������Һ��Ϊ��ɫ��˵��Fe2+�ѱ���������������
�ʴ�Ϊ��Fe2+����������������ȡ����FAS��Һ����������KSCN��Һ������Һ��Ϊ��ɫ��˵��Fe2+�ѱ���������������
���� ���⿼���Ʊ�ʵ�鷽������ơ��ζ�ʵ����̷���Ӧ�ã��ۺ��Խ�ǿ��ע���������Ϣ��װ���ص㼰���ʵ����ʷ�������Ŀ�Ѷ��еȣ�

 �ݾ�ѵ������ϵ�д�
�ݾ�ѵ������ϵ�д� С����ȫ�ܼ��ϵ�д�
С����ȫ�ܼ��ϵ�д�| A�� | ʵ��ʱ��������У�Ӧ������ˮ��ϴ��������գ�ۣ��������ִ����۾� | |
| B�� | ���������������Һ��Fe2+�ķ����ǣ��ȵμ����ư�ˮ��μ�KSCN��Һ | |
| C�� | ֤��������������ʴ�ķ����ǣ��ڶ�п��Ƥ�ϵ�1��3�κ���̪�ı���ʳ��ˮ������1��2min���۲����� | |
| D�� | ��Ϊ��������һ�ּ�����������Գ�������ɫ�����Ϳ�� |
| A�� | -OH�� ����ʾ�ǻ� ����ʾ�ǻ� | B�� | ��ͼ��ʳ�ξ���ľ��� | ||
| C�� | CH4���ӵ����ģ�ͣ� | D�� | ��������ӵĵ���ʽ�� |
| A�� | 3.36L | B�� | 1.12L | C�� | 2.24L | D�� | 4.48L |
| A�� |  ��ʾij���ȷ�Ӧ�ֱ����С�������������·�Ӧ�����е������仯 | |
| B�� |  ��ʾ��Mg2+��Al3+��NH4+������Һ�еμ�NaOH��Һʱ�����������ʵ�����NaOH��Һ����Ĺ�ϵ�����������ӵ����ʵ���֮�ȱ�Ϊ��Mg2+������Al3+����n��NH4+��=2��3��2 | |
| C�� | 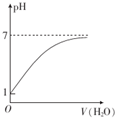 ��ʾ������0.10 mol•L-1��CH3COOH��Һ��ˮϡ��ʱ��Һ��pH�仯 | |
| D�� |  ��ʾ��10.00 mL 0.050 mol•L-1��������μ���0.025 mol•L-1�İ�ˮʱ��Һ��pH�仯������ V1��20.00 |