��Ŀ����
6���Ȼ����dz�����ˮ��������ij�Ȼ�����FeCl3•6H2O����Ʒ��������FeCl2���ʣ���Ҫ�ⶨ����FeCl3•6H2O������������ʵ�鰴���²�����У�
��֪�й����ӷ���ʽΪ��2Fe3++2I-��2Fe2++I2��I2+2S2O32-��2I-+S4O62һ
��1��ȡ�����Ȼ�����Ʒ����50mL��ˮ�У�����Ƭ�̣�Һ����ֺ��ɫ����Ӧ�����ӷ���ʽΪ��Fe3++3H2O?Fe��OH��3+3H+��
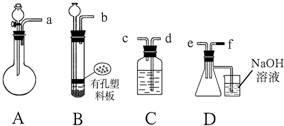
��2������I���õ��IJ����������ձ����������⣬��������100mL����ƿ����ͷ�ιܣ����������ƣ���
��3������II�����õ���������d��ѡ���ţ���
a��50mL�ձ� b��10mL��Ͳ c��20mL��Ͳ d��25mL�ζ���
ָʾ���ǵ�����Һ����ﵽ�ζ��յ�����������һ�α�Һ����ʱ����ƿ����Һ����ɫ��Ϊ��ɫ���Ұ�����ڲ���ɫ��
��4���ζ�ʱ������Ũ��Ϊ0.1000mol/L�ı�Na2S2O3��Һ18.00mL������Ʒ��FeCl3•6H2O��ʽ��Ϊ270.5������������Ϊ97.38%��
��5��Ҫ����Ʒ�Ȼ����е�����FeCl2���ʳ�ȥ�����õ��Լ���bd��ѡ���ţ���
a������ b����ˮ c����ˮ d��˫��ˮ
��6������������²���ⶨ�Ȼ�����ƷԪ�صĺ��������������գ�
�ٳ�����Ʒ �ڼ�ˮ�ܽ� �ۼ�������ˮ������ �ܹ��� ������ ���������к��ز�����
��ȱ�ٵ�һ��������ϴ�ӣ��ڹ���ǰ����Ҫ�����Ƿ������ȫ����������������ڲ���Һ�У����백ˮ��Һ�۲����������ɣ�����������֤��������ȫ��
�ж��Ƿ���صı������γ�����������Ȼ�������0.001g��
���� ��1���Ȼ���������Һ�����ˮ�м������������������壻
��2����������һ�����ʵ���Ũ�ȵ���Һ����Ҫ�������У���Ͳ����ͷ�ιܡ��ձ�����������һ����������ƿ��
��3��������������Һ����ľ�ȷ�ȿ�֪��100.00mL����Һ��Ҫ����������ȡ���������۱���ɫ�����Na2S2O3��Һ����͵ⵥ�ʷ�Ӧ����Һ��ɫ��Ϊ��ɫ�Ұ���Ӳ���ɫ��
��4�����ݷ�Ӧ�Ķ�����ϵ����õ���ע����Һ����ı仯��
��5��Ҫ����Ʒ�Ȼ����е�����FeCl2���ʳ�ȥ����Ҫ����������������������Ϊ�����ӣ�����������������������µ����ʣ�
��6������ʵ��������̷���������Ҫ���˺�ϴ�ӳ�ȥ��������ʣ������Ƿ������ȫ���������ϲ���Һ�м��백ˮ�۲��Ƿ��г������ɣ������������صı������γ���������ͬ��������0.001g��
��� �⣺��1��ȡ�����Ȼ�����Ʒ����50mL��ˮ�У�����Ƭ�̣�Һ����ֺ��ɫ�����ɵ��������������壬��Ӧ�����ӷ���ʽΪFe3++3H2O?Fe��OH��3+3H+��
�ʴ�Ϊ��Fe3++3H2O?Fe��OH��3+3H+��
��2��������һ�����ʵ���Ũ�ȵ���Һ����Ҫ�������У���Ͳ����ͷ�ιܡ��ձ�����������һ����������ƿ��
�ʴ�Ϊ��100mL����ƿ����ͷ�ιܣ�
��3��100.00mL����Һ��Ҫ����������ȡ���ձ��Ǵ�����ȡ����Ͳֻ�ܾ�ȷ��0.1mL�������õζ��ܾ�ȷ��0.01mL��ѡ�õζ�����ȡ��Һ100.00mL����Һ���������۱���ɫ�����Na2S2O3��Һ����͵ⵥ�ʷ�Ӧ�����һ�α�Һ����ʱ����ƿ����Һ����ɫ��Ϊ��ɫ���Ұ�����ڲ���ɫ��˵���ﵽ��Ӧ�յ㣻
�ʴ�Ϊ��d�����һ�α�Һ����ʱ����ƿ����Һ����ɫ��Ϊ��ɫ���Ұ�����ڲ���ɫ��
��4��2Fe3++2I-��2Fe2++I2��I2+2S2O32-��2I-+S4O62-��2FeCl3-6H2O��2Fe3+��I2��2S2O32-���ζ�ʱ��10.00ml��Һ�еⵥ������Ũ��Ϊ0.1000mol/L�ı�Na2S2O3��Һ18mL��FeCl3-6H2O�����ʵ���=0.1000mol/L��0.018L=0.0018mol������Ʒ��100.00mL��Һ������FeCl3•6H2O�����ʵ���Ϊ0.018mol����������=$\frac{0.018mol��270.5g/mol}{5.0g}$��100%=97.38%��
�ʴ�Ϊ��97.38%��
��5��Ҫ����Ʒ�Ȼ����е�����FeCl2���ʳ�ȥ����Ҫ����������������������Ϊ�����ӣ�����������������������µ����ʣ�
a�����ۺ������ӷ�Ӧ�����ܺ� �������ӷ�Ӧ����a�����ϣ�
b����ˮ����������������Ϊ�����ӣ��Ҳ������µ����ʣ���b���ϣ�
c����ˮ�������������ӣ��������������ӣ���c�����ϣ�
d��˫��ˮ����������������Ϊ�����ӣ��������ⱻ��ԭΪˮ�����������ʣ���d���ϣ�
��ѡbd��
��6��ʵ��������̷���������Ҫ���˺�ϴ�ӳ�ȥ��������ʣ������Ƿ������ȫ���������ϲ���Һ�м��백ˮ�۲��Ƿ��г������ɣ������������صı������γ���������ͬ��������0.001g��
�ʴ�Ϊ��ϴ�ӣ����������ڲ���Һ�У����백ˮ��Һ�۲����������ɣ�����������֤��������ȫ�����γ�����������Ȼ�������0.001g��
���� ���⿼����������ɺ����ʵ�ʵ����֤��ʵ��̽�������������仯�������ʵķ���Ӧ�ã����ʳ��ӣ��ζ�ʵ��ⶨ���ʺ����ļ���Ӧ���жϣ���Ŀ�Ѷ��еȣ�

| A�� | ����ˮ�ĵ���̶ȵ��Ǣٵ�8�� | |
| B�� | �ٺ͢���ˮ�ĵ���̶���� | |
| C�� | ������Ģٺܻ͢�Ϻ�������Һ�У�c��NH3•H2O����c��NH4+�� | |
| D�� | ����ˮ�ĵ���̶ȵ��ڢ� |
| A�� | m=2 | |
| B�� | ����ƽ���ƽ�ⳣ����ͬ��ƽ�ⳣ��ֵΪ2 | |
| C�� | X��Y��ƽ��ת����֮��Ϊ1��1 | |
| D�� | �ڶ���ƽ��ʱ��Z��Ũ��Ϊ1.0 mol•L-1 |