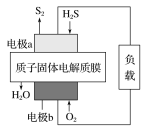
��Ŀ����
����Ŀ��ͭ����������粢�㷺ʹ�õ�һ�ֽ������ش��������⣺
��1��ʵ����ʹ��ϡ�����H2O2�ܽ�ͭƬ���÷�Ӧ�����ӷ���ʽΪ___��
��2�����ӹ�ҵʹ��FeCl3��Һ��ʴӡˢ��·��ͭ����д���÷�Ӧ�Ļ�ѧ����ʽ___������FeCl3��Һ��FeCl2��Һ��Ӧ����___(����������������������������)��ԭ����___������FeCl2��Һʱ��Ӧ����������м��ԭ����___��
��3����Һ��Cu2����Ũ�ȿɲ��õ�������ã�
��2Cu2����5I-��2CuI����I3- ��I3-��2S2O32-��3I-��S2O62-
��Ӧ���еĻ�ԭ����Ϊ___����ȡ10.00mL��Cu2������Һ����������KI��ַ�Ӧ����0.1000mol��L��1Na2S2O3����Һ�ζ����յ㣬����Na2S2O3��Һ12.50mL������Һ��Cu2����Ũ��Ϊ___mol��L��1��
���𰸡�Cu+H2O2+2H+=Cu2++2H2O 2FeCl3+Cu=2FeCl2+CuCl2 ���� ����Fe3+��Fe2+ˮ�� ��ֹFe2+�������е��������� CuI 0.125
��������
��1��ϡ������������ԣ�ʵ����ʹ��ϡ�����H2O2�ܽ�ͭƬ��������ͭ��ˮ��
��2�������Ӿ��������ԣ����ӹ�ҵʹ��FeCl3��Һ��ʴӡˢ��·��ͭ���������Ȼ��������Ȼ�ͭ�� ����Fe3+��Fe2+����ˮ�������Fe2+���л�ԭ�ԣ��ױ�����������
��3������Ԫ�ػ��ϼ������жϻ�ԭ������ݹ�ϵʽ2S2O32-������I3-������2Cu2������Cu2����Ũ�ȡ�
��1��ʵ����ʹ��ϡ�����H2O2�ܽ�ͭƬ��������ͭ��ˮ����Ӧ�����ӷ���ʽ��Cu+H2O2+2H+=Cu2++2H2O��
��2�����ӹ�ҵʹ��FeCl3��Һ��ʴӡˢ��·��ͭ���������Ȼ��������Ȼ�ͭ����Ӧ�Ļ�ѧ����ʽ��2FeCl3+Cu=2FeCl2+CuCl2��Fe3+��Fe2+����ˮ�⣬������Һ�����ԣ���������Fe3+��Fe2+ˮ�⣻Fe2+���л�ԭ�ԣ��ױ�����������Ϊ��ֹFe2+�������е���������������FeCl2��Һʱ��Ӧ����������м��
��3��2Cu2����5I-��2CuI����I3-��Ӧ��ͭԪ�ػ��ϼ���+2����Ϊ+1����ԭ������CuI��
����Һ��Cu2����Ũ��Ϊc��
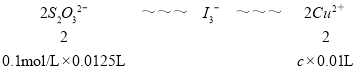
![]() =0.125mol/L��
=0.125mol/L��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�����Ŀ��2.3g�Ҵ���һ������������ϵ�ȼ��ǡ����ȫȼ�գ��ų�����Ϊ68.35kJ��
��1���÷�Ӧ���Ȼ�ѧ����ʽΪ___��
��2����֪��
��ѧ�� | Si-Cl | H-H | H-Cl | Si-Si |
����/kJ��mol-1 | 360 | 436 | 431 | 176 |
�ҹ辧����ÿ����ԭ�Ӻ�����4����ԭ���γ�4�����ۼ���
��ҵ�����õĸߴ����ͨ�����з�Ӧ��ȡ��SiCl4(g)��2H2(g)![]() Si(s)��4HCl(g)���÷�Ӧ�ķ�Ӧ��Ϊ___kJ��mol-1��
Si(s)��4HCl(g)���÷�Ӧ�ķ�Ӧ��Ϊ___kJ��mol-1��
��3����֪ˮ�ı�����Ϊ4.2��10-3kJ��g-1����-1��8g�����O2����ȫȼ��������̬SO2���ų���������ʹ500g H2O���¶���18������58�棬�����ȼ���ȵ��Ȼ�ѧ����ʽΪ___��