��Ŀ����
17��SO2����Ҫ�Ĵ�����Ⱦ���ҵ�����в����ĺ�SO2�����������������ŷţ���ȥ������SO2�ķ����ж��֣��ش��������⣺��1�����Ƚ�����ԭ������CO��ԭSO2����S4��CO2����Ӧ�Ļ�ѧ����ʽΪ8CO+4SO2$\frac{\underline{\;\;��\;\;}}{\;}$S4+8CO2
��2����ʪʽ���շ������������ռ���SO2������Ӧ�Ӷ�����
�������Լ����ʺ������÷����ռ�����c������ĸ����ͬ����
a������ʯ��ˮ b��Ũ���� c��Na2CO3��Һ
�ڡ��Ƽ����NaOH��Һ�����ռ�����100mL��0.2mol•L-1NaOH��Һͨ���״����0.448LSO2���壬��Ӧ������ҺpH��7������Һ�����и�����Ũ�ȹ�ϵ��ȷ����ab
a��c��HSO3-����c��SO42-����c��H2SO3��
b��c��OH-��+c��SO32-��=c��H+��+c��H2SO3��
c��c��Na+��+c��H+��=c��HSO3-��+c��SO32-��+c��OH-��
�۹�ҵ�ϳ���Na2SO4��Һ���������е�SO2��������ͨ��1.0mol•L-1��Na2SO4��Һ����ҺpH���ϼ�С���������������Һ������䣩������ҺpHԼΪ6ʱ������SO2�����������½���Ӧ�������ռ�����ʱ��Һ��c��SO32-��=0.2mol•L-1������Һ��c��HSO3-��=1.6mol/L
�ܡ�����������շ����ã�NH4��2SO3��Һ���������з��ѭ���ѹ죬��������Ҫ��ӦΪ��NH4��2SO3+SO2+H2O=2NH4HSO3�����25��ʱ��ҺpH���������ʵ��������ı仯��ϵ��ͼ1��ʾ��a��ʱn��HSO3-����n��H2SO3��=1��1��b��ʱ��ҺpH=7����n��NH4+����n��HSO3-��=3��1
��3��������Ĥ��ⷨ��������������Һ����SO2�����ö��Ե缫��⣬������X��Һ�����������˷�����Ǻ�ѭ���������ã�װ����ͼ2��ʾ���������ĵ缫��ӦʽΪSO2+4H++4e-�TS+2H2O����������������Ļ�ѧʽΪO2��

���� ��1��CO��ԭSO2����S4��CO2�����ݻ��ϼ����������ƽ�÷�Ӧ����ʽ��
��2���ٸ��������ԭ�����ʺ������÷����ռ���������SO2������Ӧ�����ʣ��ݴ˶Ը�ѡ������жϣ�ע�����ʯ��ˮ����������Ũ�Ƚ�С��
��n��SO2��=$\frac{0.488L}{22.4L/mol}$=0.02mol��n��NaOH��=0.1L��0.2mol/L=0.02mol�����߷�Ӧ�Ļ�ѧ����ʽΪ��SO2+NaOH=NaHSO3����Ӧ������ҺpH��7��NaHSO3��Һ�����ԣ�˵��HSO3-����̶ȴ���ˮ��̶ȣ������Һ�ĵ�����ԭ�������غ��Լ������غ�����⣻
�۽��Na2SO3+SO2+H2O�T2NaHSO3��������Ũ�ȣ���ҺpHԼΪ6ʱ������SO2�����������½�������ΪNaHSO3������������ˮ�⣻
�ܸ���ͼ�������ڶ��߱�ֵ�ı仯��������李���������淋Ļ�ѧʽ��ɽ��з��������㣻
��3�������Ƕ���������������Һ�еõ��������ɵ�������������������Ӧ����Һ������������ʧȥ��������������
��� �⣺��1��CO��ԭSO2����S4��CO2��COת���ɶ�����̼�����ϼ�����2�ۣ�SO2����S4�����ϼ۽���4��4=16�ۣ����ϼ۱仯����С������Ϊ16����CO�ļ�����Ϊ8����������ļ�����Ϊ4��Ȼ����ݹ۲취��ƽ�ɵã�8CO+4SO2$\frac{\underline{\;\;��\;\;}}{\;}$S4+8CO2��
�ʴ�Ϊ��8CO+4SO2$\frac{\underline{\;\;��\;\;}}{\;}$S4+8CO2��
��2����a��ʯ��ˮ���������Ƶ�Ũ�Ƚ�С�����ʺ������ռ�����a����
b������������Ũ���ᷴӦ���������ն�������b����
c�������������ǿ��̼�ᣬ���Զ�����������Na2CO3��Һ��Ӧ��SO2+Na2CO3=Na2SO3+CO2����c��ȷ��
�ʴ�Ϊ��c��
��n��SO2��=$\frac{0.488L}{22.4L/mol}$=0.02mol��n��NaOH��=0.1L��0.2mol/L=0.02mol�����߷�Ӧ�Ļ�ѧ����ʽΪ��SO2+NaOH=NaHSO3��
a�������������ᣬ��Ӧ������ҺpH��7��NaHSO3��Һ�����ԣ�˵��HSO3-����̶ȴ���ˮ��̶ȣ���c��SO32-����c��H2SO3����NaHSO3��Һ������Һ������������Ӻ�������������ӣ�HSO3-�����ˮ��̶�������������Ƶĵ��붼�Ƚ�С������c��HSO3-����c��SO32-����c��H2SO3������a��ȷ��
b������������������Һ�е������غ�ɵã�c��OH-��+c��SO32-��=c��H+��+c��H2SO3������b��ȷ��
C����Һ��һ�����ڵ���غ㣬����������������Һ�еĵ���غ�ɵã�c��Na+��+c��H+��=c��HSO3-��+2c��SO32-��+c��OH-������c����
�ʴ�Ϊ��ab��
����Na2SO3+SO2+H2O�T2NaHSO3��c��SO32-����Ũ����0.2mol•L-1����Ӧ��Na2SO3��ҺΪ1.0mol/L-0.2mol/L=0.8mol/L���ɷ�Ӧ�е����ʵ�����ϵ��֪�����ɵ�c��HSO3-��Ϊ��0.8mol/L��2=1.6mol/L��
�ʴ�Ϊ��1.6mol/L��
�ܡ�����������շ����ã�NH4��2SO3��Һ���������з��ѭ���ѹ죬��������Ҫ��ӦΪ��NH4��2SO3+SO2+H2O=2NH4HSO3�����25��ʱ��ҺpH���������ʵ��������ı仯��ϵ��ͼ1��ʾ������ͼ���֪a��ʱ������ֵ��ȣ���n��HSO3-����n��H2SO3��=1��1����b��ʱ��ҺpH=7�����ݵ���غ��֪n��NH4+��=n��HSO3-��+2n��SO32-�����ָ���ͼ�����߿�֪��n��HSO3-��=n��SO32-������n��NH4+����n��HSO3-��=��1��2����1=3��1��
�ʴ�Ϊ��1��1��3��1��
��3�������Ƕ���������������Һ�еõ��������ɵ����缫��ӦΪ��SO2+4H++4e-�TS+2H2O����������������ʧȥ��������O2��
�ʴ�Ϊ��SO2+4H++4e-�TS+2H2O��O2��
���� ���⿼��������Ũ�ȴ�С�Ƚϡ����ԭ������Ӧ�á���ѧ����ʽ�����ӷ���ʽ��д��֪ʶ����Ŀ�ѶȽϴ�����֪ʶ��϶ࡢ�����ϴ�ֿ�����ѧ���ķ���������������ע�����յ��ԭ������Ӧ�÷�������ȷ�ж�����Ũ�ȴ�С���÷�����

 �����й��ڸ�Ԫ�ص�˵���У�������ǣ�������
�����й��ڸ�Ԫ�ص�˵���У�������ǣ�������| A�� | ���������Ӵ�3����λ����� | B�� | ����һ�ֽ���Ԫ�� | ||
| C�� | ������������10������ | D�� | ��ԭ�Ӻ�����13������ |
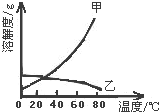 �������ֹ�����ܽ��������ͼ��ʾ����һ�����ձ���װ�ź��м��ҵ����ֱ�����Һ�����ұ��л����������ҵĹ�����ڣ��ֽ��¶���80�潵��20�森����˵��������ǣ�������
�������ֹ�����ܽ��������ͼ��ʾ����һ�����ձ���װ�ź��м��ҵ����ֱ�����Һ�����ұ��л����������ҵĹ�����ڣ��ֽ��¶���80�潵��20�森����˵��������ǣ�������| A�� | ���й�������ࡢ�����Ҽ��� | |
| B�� | ����һ���й������һ���й����� | |
| C�� | ��Һ�����ʼ��١����������� | |
| D�� | ���ҵ�������Һ��Ȼ�DZ�����Һ |
| A�� | 7.8g�������ƺ��еĹ��õ��Ӷ���Ϊ0.2NA | |
| B�� | 2H2O2��l��=2H2O��l��+O2��g����H=-98.2kJ•mol-1����S=70.5J•mol-1•K-1�÷�Ӧ�����¶��¶����Է����� | |
| C�� | ��������ʹ���������Һ��ɫ��1mol��������õ�2NA���� | |
| D�� | �ں���NH4+��Ba2+��Cl-��NO3-���ӵ���Һ�У��������������������ϸ����������������� |
| A�� | ��߸�����ص�������������Ũ�����ữ | |
| B�� | ����Fe2+ ��ˮ�⣬��ϡ�����ữ | |
| C�� | ����ij±�������Ƿ���Ԫ�أ��ȼ�HNO3�ữ���ټ�AgNO3��Һ���飮 | |
| D�� | ȷ����Һ�к���SO42- ʱ�����ù��������ữ���ټ�BaCl2��Һ���飮 |
| A�� | ���ڷ�ӦNa2SO3+H2SO4�TNa2SO4+SO2��+H2O��������ϵ��ѹǿ | |
| B�� | ����N2��g��+3H2��g��?2NH3��g������������̶�����ʱ������Ar | |
| C�� | ����N2��g��+3H2��g��?2NH3��g������������̶�����ʱ������N2 | |
| D�� | ����N2��g��+3H2��g��?2NH3��g������������ѹǿ����ʱ������Ar |
 �����������г�������Ʒ��a�������� b����Դ���� c���Ͼɸɵ�� d���ϱ�ֽ e�������� f����̥ g������
�����������г�������Ʒ��a�������� b����Դ���� c���Ͼɸɵ�� d���ϱ�ֽ e�������� f����̥ g������