��Ŀ����
18�����л�ѧ��Ӧ�����ӷ���ʽ��ȷ���ǣ�������| A�� | ���Ṥҵβ���е�SO2�ù����İ�ˮ���գ�2NH3•H2O+SO2�T2NH4++SO32-+H2O | |
| B�� | FeO��ϡHNO3�ķ�Ӧ��FeO+2H+�TFe2++H2O | |
| C�� | ��NH4Cl��Һ�е����ռ���Һ�����ȣ�NH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3•H2O | |
| D�� | ��NaClO��Һ��ͨ������SO2��2ClO-+SO2+H2O�T2HClO+SO32- |
���� A����ˮ��������Ӧ����������泥�
B���������ӱ�ϡ���������������ӣ�
C������������笠����������������ӷ�Ӧ���ɰ�����ˮ��
D�����������ǿ�����ԣ��ܹ����������������������������ӣ�
��� �⣺A�����Ṥҵβ���е�SO2�ù����İ�ˮ���գ���Ӧ�����ӷ���ʽΪ��2NH3•H2O+SO2�T2NH4++SO32-+H2O����A��ȷ��
B��FeO��ϡHNO3����������ԭ��Ӧ����ȷ�����ӷ���ʽΪ��3FeO+10H++NO3-�T3Fe3++NO��+5H2O����B����
C����NH4Cl��Һ�е����ռ���Һ�����ȣ���Ӧ���ɰ������Ȼ��ƺ�ˮ����ȷ�����ӷ���ʽΪ��NH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O����C����
D����NaClO��Һ��ͨ������SO2�������������������ᣬ��ȷ�����ӷ���ʽΪ��3ClO-+SO2+H2O�T2HClO+Cl-+SO42-����D����
��ѡA��
���� ���⿼�������ӷ���ʽ���жϣ�Ϊ�߿��ĸ�Ƶ�⣬��Ŀ�ѶȲ���ע���������ӷ���ʽ�����жϳ��÷�������鷴Ӧ��������Ƿ���ȷ���������ʲ���Ƿ���ȷ���������������ʵ���Ҫ������ѧʽ������Ƿ����ԭ��ѧ����ʽ�ȣ�

��ϰ��ϵ�д�
�����Ŀ
6��ʳƷ�㾫����������������·�ߣ���Ӧ������ȥ�����£�������������ȷ���ǣ�������


| A�� | ��������ķ���ʽΪC8H8O3 | |
| B�� | ���ӡ���������Ͳ�����������NaOH��Һ������Ӧ | |
| C�� | ���裨1�������в����ı��ӿ���FeCl3��Һ���� | |
| D�� | ���裨2�������в�����ϩ����������ˮ���� |
3�����б仯�д��ڿ��淴Ӧ���ǣ�������
| A�� | ��ˮ���� | B�� | �����ϻ� | C�� | ţ�̱��� | D�� | ������ʴ |
10�����м��������л�Ϊͬ���칹����ǣ�������
| A�� | ˮ����ˮ | B�� | �����ͳ��� | ||
| C�� | 2-������������� | D�� | 뮺�� |
7��ijԪ��X����̬�⻯��ķ���ʽΪH2X����X��������ۺ�����ķ���ʽΪ��������
| A�� | H2S | B�� | HNO3 | C�� | H2SO4 | D�� | H2SO3 |
8��ijʵ������������������һ�ֻ����Һ����֪��Һ�У�c��K+��=c��Cl-��=$\frac{1}{3}$ c��Na+��=$\frac{2}{3}$c��SO42-�����������ʿ����ǣ�������
| A�� | MgCl2��Na2SO4��K2SO4 | B�� | KCl��Na2SO4��NaCl | ||
| C�� | NaCl��Na2SO4��K2SO4 | D�� | KCl��Na2SO4��K2SO4 |

 ��
��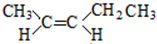 ��
��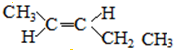 ��
�� ��
�� Ϊ��֤��ͭ��ϡ���ᷴӦ��������NO��ijУѧ��ʵ��С�������һ��ʵ�飬��װ����ͼ��ʾ������װ�ú̶�װ�þ�����ȥ����BΪһ���ý���˿�̶��ĸ���ܣ���װ��״̼��ƹ��壻EΪһ���յ�������ƿ��F�����ڹ�������Ĵ�����
Ϊ��֤��ͭ��ϡ���ᷴӦ��������NO��ijУѧ��ʵ��С�������һ��ʵ�飬��װ����ͼ��ʾ������װ�ú̶�װ�þ�����ȥ����BΪһ���ý���˿�̶��ĸ���ܣ���װ��״̼��ƹ��壻EΪһ���յ�������ƿ��F�����ڹ�������Ĵ�����