��Ŀ����
15��ij��ѧ��ȤС��ⶨijFeCl3��Ʒ��������FeCl2���ʣ�����Ԫ�ص�����������ʵ���Ұ����²�����У��ٳ���a g��Ʒ�������ձ��У��ڼ��������������������ˮ��ʹ��Ʒ�ܽ⣬Ȼ��ȷ���Ƴ�250mL��Һ��
��ȷ��ȡ25.00mL���������õ���Һ�������ձ��У�������������ˮʹ��Ӧ��ȫ��
�ܳ���Ѹ�ټ��������ˮ����ֽ��裬ʹ������ȫ�� �ݹ��ˣ�ϴ�ӳ�����
������ת�Ƶ������ڣ����ȡ����裬ֱ������ȫ���ɺ��ɫ��Ϊ����ɫ���ڸ���������ȴ�����º�����
���ظ����ϲ�������������γƵõ�����������0.1gΪֹ��
����������������ش�
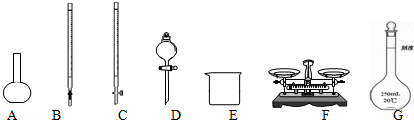
��1����ͼ��ʾ�����У���ʵ�鲽��٢ڢ��б����õ���������E��F��CG������ĸ����
��2������ڼ��������Ŀ���Ƿ�ֹFeCl3��FeCl2ˮ�⣻�ӿ���Ʒ�ܽ����������ò��������裮
��3��д��������з�����Ӧ�����ӷ���ʽ2Fe2++Cl2=2Fe3++2Cl-��
��4������ݵ�ϴ����ϴȥ�����ڳ����ϵ�NH4+��Cl-��OH-��д���ӷ��ţ���ϴ�ӳ����IJ�����©����ע������ˮ��ʹˮ��û����������ˮ��Ȼ������ظ�����2��3�Σ�
��5��������������W1����������Ⱥ������������W2������Ʒ����Ԫ�ص���������Ϊ$\frac{7��W{\;}_{2}-W{\;}_{1}��}{a}$��100%��
���� ��1�����ݳ������ܽ⡢����һ�����ʵ���Ũ�ȵ���Һ��ȷ��ȡ��Һȷ������������
��2������FeCl3��FeCl2�ܷ���ˮ�⣻�����ò���������ɼӿ���Ʒ�ܽ⣻
��3��������ˮ�ܽ�FeCl2��������FeCl3��
��4��������Һ�к��а�ˮ���Ȼ����ȷ�����ӣ�����ϴ�ӳ����ķ�����
��5��������Ԫ�������غ㣬������ɫ���壨Fe2O3���е���������Ʒ�������������������Ĺ�ʽ�����Ԫ�ص�����������
��� �⣺��1�������ʱ��Ҫ��ƽ��ҩ�ף��ܽ�ʱ��Ҫ�ձ���������������һ�����ʵ���Ũ�ȵ���Һʱ��Ҫ��Ͳ����ͷ�ιܡ��ձ�����������һ����������ƿ��ȷ��ȡ��Һȷ��������ʽ�ζ��ܡ�ϴ����
�ʴ�Ϊ��CG��
��2����FeCl3��FeCl2�ܷ���ˮ�⣻���ò���������ɼӿ���Ʒ�ܽ⣬
�ʴ�Ϊ����ֹFeCl3��FeCl2ˮ�⣻�ò��������裻
��3������ˮ�ܽ�FeCl2��������FeCl3��2Fe2++Cl2=2Fe3++2Cl-��
�ʴ�Ϊ��2Fe2++Cl2=2Fe3++2Cl-��
��4������Һ�к��а�ˮ���Ȼ�泥�����������NH4+��Cl-��OH-����ϴ�ӳ����ķ���Ϊ��©����ע������ˮ��ʹˮ��û����������ˮ��Ȼ������ظ�����2��3�Σ�
�ʴ�Ϊ��NH4+��Cl-��OH-����©����ע������ˮ��ʹˮ��û����������ˮ��Ȼ������ظ�����2��3�Σ�
��5������Ԫ�������غ㣬������ɫ�����е���������Ʒ������Fe2O3����Ԫ�ص�����Ϊ��W2-W1��g��$\frac{112}{160}$��$\frac{250}{25}$=7��W2-W1��g����Ʒ����Ԫ�ص���������Ϊ��$\frac{7��W{\;}_{2}-W{\;}_{1}��}{a}$��100%��
�ʴ�Ϊ��$\frac{7��W{\;}_{2}-W{\;}_{1}��}{a}$��100%��
���� ������Ҫ��������Ԫ�ص����������IJⶨ��ͬʱ������ʵ��֪ʶ���ѶȲ���

| A�� | 1mol MgCl2 | B�� | 2mol KClO3 | C�� | 1 mol AlCl3 | D�� | 1.5 mol CaCl2 |
| A�� | ����Ԫ�صĻ����ﶼ�������� | B�� | ����Ԫ�صĻ����ﶼ���� | ||
| C�� | ��ˮ����ﲻ�Ǵ����� | D�� | ֻ��һ��Ԫ�ص����ʲ�һ���ǵ��� |
| A�� | ��ȥˮ�к���������KNO3��ͨ��������ռ�ˮ | |
| B�� | ������Һ�е�笠����ӣ����ȣ�����ʪ��ĺ�ɫʯ����ֽ������������� | |
| C�� | ��ȥKI�����е������ⵥ�ʣ�I2�������������м��� | |
| D�� | SO42-�ļ��飺��Һ����ϡHNO3�ữ��Ȼ��μ�BaCl2��Һ�����Ƿ��г������� |
| A�� | 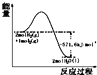 ��ʾH2��O2������Ӧ�����е������仯��H2��ȼ����Ϊ285.8 kJ/mol | |
| B�� |  ��ͼ�ó�����ȥCuSO4��Һ�е�Fe3+���ɲ�������Һ�м�������CuO��������Һ��pH��4���� | |
| C�� |  ��ʾ��ˮϡ��pH��ͬ������ʹ���ʱ��Һ��pH�仯���ߣ����Т��ʾ���ᣬ���ʾ���ᣬ����Һ�����ԣ�c��b��a | |
| D�� |  ��ʾ�����������Ϊ10 mL 0.1 mol/LNaOH��Һ����μ���0.1 mol/L CH3COOH��Һ����Һ��pH�仯���ߣ���b�㴦�У�c��CH3COOH��+c��H+��=c��OH-�� ��ʾ�����������Ϊ10 mL 0.1 mol/LNaOH��Һ����μ���0.1 mol/L CH3COOH��Һ����Һ��pH�仯���ߣ���b�㴦�У�c��CH3COOH��+c��H+��=c��OH-�� |
| A�� | 6g SiO2�������0.4NA��Si-O�� | |
| B�� | PH=12��Na2CO3����0.01NA��ˮ���ӷ����˵��� | |
| C�� | ����£�11.2LHF���е�HF����������0.5NA | |
| D�� | �Ѻ�1molFeCl3�ı�����Һ�����ˮ��������ɫ����ֹͣ�����С��1NA |

