��Ŀ����
8����������ƣ�Na2S2O3���������մ���������������ҵ����Ӱ����Ҳ������ֽ��Ư�������ȼ��ȣ�ʵ���ҳ������ƣ�Na2S��Ϊԭ����ȡNa2S2O3��ʵ����ĿI����ҵ�����ƵĴ�����
��ʵ���Na2S�Ĵ���Ҫ��ϸߣ�����ͼ1��ʾ��װ�ý���ҵ����Na2S�ᴿ��

��֪��Na2S���������ھƾ�������ʱ�ܽ��Ѹ���������ʲ����ھƾ���
�ᴿ��������Ϊ��
�ٽ��ѳ����õĹ�ҵNa2S����Բ����ƿ�У�������һ�������ľƾ�������ˮ��
�ڰ�ͼ1��ʾ����������ˮԡ���ȣ������ܵ���������������������������ͨ����ȴˮ�ķ����Ǵ�b�ڽ�ˮ���a����b������
�۴���ƿ�й��岻�ټ���ʱ��ֹͣ���ȣ�����ƿȡ�£����ȹ��ˣ���ȥ�����
�ܽ���Һת�����ձ��У���ȴ�ᾧ�����ˣ�
�ݽ����ù����������Ҵ������Լ����ƣ�ϴ�ӣ������õ�Na2S•9H2O���壮
ʵ����Ŀ����������Ƶ��Ʊ���
�Ʊ���������Ϊ��
�ٳ�ȡһ�����������ƾ����̼���ƹ��壬����ˮ��ת��������ƿ�У�
�ڰ�ͼ2��ʾ�������������ڸ������м�����Ӧ���Լ�����Һ©���Ļ�����ʹ��Ӧ���ɵ�����Ͼ��ȵ�ͨ������ƿ�У����õ�Ž��������Ͻ��裻
����A������Ϊ������ƿ��
д������ƿ�з����ķ�Ӧ�Ļ�ѧ����ʽ��Na2CO3+2Na2S+4SO2=3Na2S2O3+CO2��
�����������ͨ�룬����dz��ɫ��������������ͨ����������ҺpH�ӽ�7��ֹͣͨ�����壬ȡ��������ƿ�����ˣ�ʵ���������δ��ʱֹͣͨ��������ܲ����ĺ��ΪNa2S2O3�����Ի����в��ȶ������ʽ��ͣ�
�ܽ���Һ�����������м��ȣ���������������ʱ��ֹͣ���ȣ���ȴ�����ˣ�����Na2S2O3•5H2O���壮
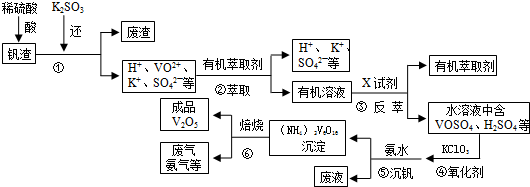
���� ʵ����ĿI���ᴿ���裺�ھƾ��ӷ������������ƾ�����ȡ����ԭ��ͨ������ˮ��
��Na2S������ˮ�����������ھƾ���
ʵ����Ŀ���Ʊ����裺���������ṹ��������֪AΪ������ƿ��
������ƿ��SO2��Na2S��Na2CO3��Ӧ����Na2S2O3��ͬʱ���ɶ�����̼��
��Na2S2O3�����Ի����в��ȶ������ֽ�õ�S���������
�ܽ���Һ�����������м��ȣ���������������ʱ��ֹͣ���ȡ���ȴ�����ˣ�����Na2S2O3•5H2O���壮
��� �⣺ʵ����ĿI���ᴿ���裺�ھƾ��ӷ��������ܿ������������ƾ������ٻӷ�����ȡ����ԭ��ͨ������ˮ�������ȴ������b��ͨ������ˮ��
�ʴ�Ϊ������������b��
��Na2S������ˮ�����������ھƾ������ù����������Ҵ�ϴ�ӣ��������ܽ�µ���ʧ��
�ʴ�Ϊ���Ҵ���
ʵ����Ŀ���Ʊ����裺���������ṹ��������֪AΪ������ƿ��
������ƿ��SO2��Na2S��Na2CO3��Ӧ����Na2S2O3��ͬʱ���ɶ�����̼����Ӧ����ʽΪ��Na2CO3+2Na2S+4SO2=3Na2S2O3+CO2��
�ʴ�Ϊ��������ƿ��Na2CO3+2Na2S+4SO2=3Na2S2O3+CO2��
��δ��ʱֹͣͨ�����壬���������������Һ�����ԣ�Na2S2O3�����Ի����в��ȶ������ֽ⣬���²��ʼ��ͣ�
�ʴ�Ϊ��Na2S2O3�����Ի����в��ȶ������ʽ��ͣ�
�ܽ���Һ�����������м��ȣ���������������ʱ��ֹͣ���ȡ���ȴ�����ˣ�����Na2S2O3•5H2O���壬
�ʴ�Ϊ����������������
���� ���⿼�����ʵ��Ʊ�ʵ�飬���ؿ���ѧ�����Ʊ�ԭ���ķ��������ʵķ����ᴿ����ѧʵ�����������ּ�ڿ���ѧ��������û���֪ʶ���ʵ��������������Ѷ��еȣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | 1��1 | B�� | 1��2 | C�� | 3��2 | D�� | 4��3 |
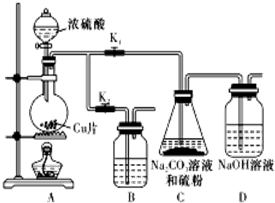 ��������ƣ�Na2S2O3�������������ƺ����ͨ�����Ϸ�Ӧ�Ƶã���֪��Na2S2O3��������Һ�в����ȶ����ڣ�
��������ƣ�Na2S2O3�������������ƺ����ͨ�����Ϸ�Ӧ�Ƶã���֪��Na2S2O3��������Һ�в����ȶ����ڣ���1��ij�о�С��������Ʊ�Na2S2O3•5H2O��װ�úͲ��ֲ����������£�
��Kl������K2����Բ����ƿ�м�������Ũ���ᣬ���ȣ�
��C�еĻ��Һ��������������Ӧһ��ʱ�����۵������٣���C����Һ��pH�ӽ�7ʱ��ֹͣC�еķ�Ӧ��
����C�еĻ��Һ��
��������Һ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ���ɣ��õ���Ʒ��
��I�У�Բ����ƿ�з�����Ӧ�Ļ�ѧ����ʽ�ǣ�Cu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��
�ڢ��С�ֹͣC�еķ�Ӧ���IJ����Ǵ�K2���ر�K1��
�ۢ��н���Һ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ���ɣ��õ���Ʒ��Na2S2O3•5H2O���ܽ�����¶����������������ò�Ʒͨ���ؽᾧ�����ᴿ��
��װ��B����������C�еķ�Ӧֹͣ������A�в����Ķ���SO2��ֹ������Ⱦ��
��2�����ݷ�Ӧ2S2O32-+I2�TS4O62-+2I-������I2�ı���Һ�ⶨ��Ʒ�Ĵ��ȣ�ȡ5.5g��Ʒ�����Ƴ�100mL��Һ��ȡ10mL��Һ���Ե�����ҺΪָʾ������Ũ��Ϊ0.050mol•L-1I2�ı���Һ���еζ���������ݼ�¼���±���ʾ��
| ��� | 1 | 2 | 3 | 4 |
| ��Һ�����/mL | 10.00 | 10.00 | 10.00 | 10.00 |
| ����I2����Һ�����/mL | 19.99 | 19.98 | 17.13 | 20.03 |
��Na2S2O3•5H2O�ڲ�Ʒ�е�����������90.2%��Na2S2O3•5H2O��ʽ��Ϊ248������������1λС������
| A�� | �� c��HCO3-��=0.1 mol•L-1 ����Һ�У�NH4+��AlO2-��Cl-��NO3- | |
| B�� | ����ˮ������� c��H+��=l��l0-12 mol•L-1 ����Һ�У�Cu2+��ClO-��Na+��SO42- | |
| C�� | �ڼ������۲��� H2 ����Һ�У�SO42-��NO3-��Na+��NH4+ | |
| D�� | ��ʹ��ɫʯ����ֽ��������Һ�У�SiO32-��CO32-��Na+��F- |
| A�� | pH=4.3��CH3COOH��CH3COONa�����Һ�У�c��Na+����c��CH3COO-�� | |
| B�� | Ũ��Ϊ0.2 mol/L��CH3COOH��Һ��Ũ��Ϊ0.1mol/L��NaOH��Һ�������Ϻ�c��CH3COO -��-c��CH3COOH��=2[c��H+��-c��OH-��] | |
| C�� | ����Ũ��Һ������ˮϡ�ͣ�$\frac{c��C{H}_{3}COOH��}{{c}^{2}��{H}^{+}��}$�������� | |
| D�� | �������ʵ�����NaHC2O4��Na2C2O4����Һ��3c��Na+��=2[c��HC2O4-��+c��C2O42-��+c��H2C2O4��] |