��Ŀ����
18������ƽ�����ʢ��ǿ��ԭ��Һ̬�£�N2H4����ǿ������Һ̬˫��ˮ������0.4molҺ̬�º�0.8mol H2O2��Ϸ�Ӧ�����ɵ�����ˮ�������ų�256.7kJ���������൱��25�桢101kPa�²�õ�����������1����Ӧ���Ȼ�ѧ����ʽΪN2H4��l��+2H2O2��l��=N2��g��+4H2O��g����H=-641.75kJ/mol��
��2���˷�Ӧ���ڻ���ƽ������ͷŴ����ȺͿ��ٲ������������⣬����һ���ܴ���ŵ��Dz��ﲻ����ɻ�����Ⱦ��
���� ��1�����ݷ�Ӧ�����������ƽ��д��ѧ����ʽ�����ݶ��ɹ�ϵ�жϣ�0.4molҺ̬�º�0.8mol H2O2���ǡ�÷�Ӧ������1molҺ̬����ȫ��Ӧ�ų�641.75kJ��������
��2�����ݷ�Ӧ����Ի�����Ӱ�������
��� �⣺��1����Ӧ����ʽΪ��N2H4+2H2O2=N2+4H2O��0.4molҺ̬�·ų�256.7KJ����������1molҺ̬�·ų�������Ϊ��$\frac{256.7kJ}{0.4}$=641.75kJ���÷�Ӧ���Ȼ�ѧ����ʽΪ��N2H4��l��+2H2O2��l��=N2��g��+4H2O��g����H=-641.75kJ/mol��
�ʴ�Ϊ��N2H4��g��+2H2O2��l��=N2��g��+4H2O��g����H=-641.75kJ/mol��
��2���˷�Ӧ���ڻ���ƽ������ͷŴ����ȺͿ��ٲ������������⣬����һ���ܴ���ŵ��Dz���Ϊ������ˮ���ǿ����ɷֲ�����ɻ�����Ⱦ��
�ʴ�Ϊ�����ﲻ����ɻ�����Ⱦ��
���� ���⿼�鷴Ӧ�����ʱ��Ӧ�ã��Ǹ߿��еij������ͣ���ȷ�Ȼ�ѧ����ʽ����дԭ��Ϊ���ؼ������������ǿ���������У������ϻ�����䣬����������ѧ��ѧϰ��ѧ����Ȥ�������ڵ���ѧ����ѧϰ�����ԣ�

��ϰ��ϵ�д�
 Сѧ��10���ӿ������100��ϵ�д�
Сѧ��10���ӿ������100��ϵ�д�
�����Ŀ
7����֪��2Fe2++Br2=2Fe3++2Br-��2Br-+Cl2=Br2+2Cl-��������amolFeBr2����Һ�У�ͨ��xmolCl2�����и���Ϊͨ��Cl2�����У���Һ����������Ӧ�����ӷ���ʽ�����в���ȷ���ǣ�������
| A�� | x=0.4a��2Fe2++Cl2=2Fe3++2Cl- | |
| B�� | x=1.5a��2Fe2++4Br-+3Cl2=2Br2+2Fe3++6Cl- | |
| C�� | x=a��2Fe2++2Br-+2Cl2=Br2+2Fe3++4Cl- | |
| D�� | x=0.6a��2Br-+Cl2=Br2+2Cl- |
6������β���к���CO��NO2���ж����壬��������װβ������װ�ã���ʹ�ж��������Ӧת��Ϊ������CO2��N2�ȣ�
��1������β����CO��NO2������һ�������¿��Է�����Ӧ��
4CO��g��+2NO2��g��?4CO2��g��+N2��g����H=-1200kJ•mol-1�����º��������£�����˵���÷�Ӧ�Ѵﵽƽ��״̬����CD������ţ���
A�������ڻ��������ɫ���ٱ仯 B�������ڵ�ѹǿ���ֲ���
C��2v����NO2��=v����N2�� D�������ڻ�������ܶȱ��ֲ���
���ڸ÷�Ӧ���¶Ȳ�ͬ��T2��T1��������������ͬʱ������ͼ����ȷ�����ң�����ţ���
����β����CO��H2O��g����һ�������¿��Է�����Ӧ��CO��g��+H2O��g��?CO2��g��+H2��g����H��0�� ij�¶�ʱ�ڼס��ҡ������������ܱ������У���ʼʱ�����ұ�����Ͷ�ϣ��ﵽƽ��״̬ʱK=81��
��2��ƽ��ʱ����������CO��ת������90%��ƽ��ʱ���Ƚ�������H2O��ת���ʣ��ң��ף����������������=������ͬ������=�ף�
��3����֪�¶�ΪTʱ��CH4��g��+2H2O��g��=CO2��g��+4H2��g����H=+165KJ•mol
CO��g��+H2O��g��=CO2��g��+H2��g����H=-41KJ•mol������Ͻ�ThNi5�ɴ���CO��H2�ϳ�CH4�ķ�Ӧ���¶�ΪTʱ���÷�Ӧ���Ȼ�ѧ����ʽ��CO��g��+3H2��g��=CH4��g��+H2O��g����H=-206kJ•mol?1��
��1������β����CO��NO2������һ�������¿��Է�����Ӧ��
4CO��g��+2NO2��g��?4CO2��g��+N2��g����H=-1200kJ•mol-1�����º��������£�����˵���÷�Ӧ�Ѵﵽƽ��״̬����CD������ţ���
A�������ڻ��������ɫ���ٱ仯 B�������ڵ�ѹǿ���ֲ���
C��2v����NO2��=v����N2�� D�������ڻ�������ܶȱ��ֲ���
���ڸ÷�Ӧ���¶Ȳ�ͬ��T2��T1��������������ͬʱ������ͼ����ȷ�����ң�����ţ���

����β����CO��H2O��g����һ�������¿��Է�����Ӧ��CO��g��+H2O��g��?CO2��g��+H2��g����H��0�� ij�¶�ʱ�ڼס��ҡ������������ܱ������У���ʼʱ�����ұ�����Ͷ�ϣ��ﵽƽ��״̬ʱK=81��
| ��ʼ���ʵ��� | �� | �� | �� |
| n��H2O��/mol | 0.10 | 0.20 | 0.20 |
| n��CO��/mol | 0.10 | 0.10 | 0.20 |
��3����֪�¶�ΪTʱ��CH4��g��+2H2O��g��=CO2��g��+4H2��g����H=+165KJ•mol
CO��g��+H2O��g��=CO2��g��+H2��g����H=-41KJ•mol������Ͻ�ThNi5�ɴ���CO��H2�ϳ�CH4�ķ�Ӧ���¶�ΪTʱ���÷�Ӧ���Ȼ�ѧ����ʽ��CO��g��+3H2��g��=CH4��g��+H2O��g����H=-206kJ•mol?1��
10�������йس��ӵ�ʵ������������ǣ�������
| A�� | �ü��������ˮ�����˵ķ�����ȥNaCl��Һ�л��е�AlCl3 | |
| B�� | ��ȥNaCl��Һ��������Na2S������AgCl���ٹ��� | |
| C�� | ��MgCl2��Һ�м���CaCO3��ĩ����ֽ������˿ɳ�ȥ����FeCl3 | |
| D�� | ��ȥFeCl3��Һ��������CuCl2�������������ۣ����� |
7�������ܱ������зֱ����H2��O2��CO2�������壬���¸��������������ȷ���ǣ�������
| A�� | �����ǵ�ѹǿ��������¶Ⱦ���ͬʱ�����������������m��H2����m��O2����m��CO2�� | |
| B�� | �����ǵ��¶Ⱥ�ѹǿ����ͬʱ������������ܶȣ��ѣ�H2�����ѣ�02�����ѣ�C02�� | |
| C�� | �����ǵ��������¶ȡ�ѹǿ����ͬʱ����������������V��CO2����V��O2����V��H2�� | |
| D�� | �����ǵ��¶Ⱥ��ܶȶ���ͬʱ�����������ѹǿ��P��H2����P��O2����P��CO2�� |
8����֪��C��s��+$\frac{1}{2}$O2��g���TCO��g����H1=-110.35kJ/mol
CO��g��+$\frac{1}{2}$O2��g���TCO2��g����H2=-282.57kJ/mol
��C��s��+O2��g���TCO2��g����H3��������
CO��g��+$\frac{1}{2}$O2��g���TCO2��g����H2=-282.57kJ/mol
��C��s��+O2��g���TCO2��g����H3��������
| A�� | +172.22kJ/mol | B�� | -172.22kJ/mol | C�� | -392.92kJ/mol | D�� | +392.92kJ/mol |
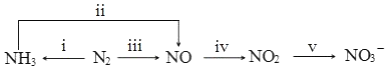
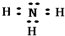 ��
��
 ��100mL��NaOH��Һ��ͨ��CO2��ַ�Ӧ���ڼ�ѹ�ͽϵ��¶��£�С�ĵؽ���Һ���ɣ��õ���ɫ����M��ͨ��CO2���������״������M������W�Ĺ�ϵ��ͼ��ʾ���Իش��������⣺
��100mL��NaOH��Һ��ͨ��CO2��ַ�Ӧ���ڼ�ѹ�ͽϵ��¶��£�С�ĵؽ���Һ���ɣ��õ���ɫ����M��ͨ��CO2���������״������M������W�Ĺ�ϵ��ͼ��ʾ���Իش��������⣺