��Ŀ����
11������ǹ�ҵ�����ij��÷�����ij�о���С������������̽����ʵ�����ͼ1װ�õ��CuCl2��Һ��ȡ����Ư��Һ��

��1��������W��Ӧ�������X���ӣ��X����Y������
��2��ʵ���������̼���ϳ��˸����к�ɫ���ʣ���������������ɫ���ʣ�
����������ʾ��
| �������Ƽ���ѧʽ | �Ȼ���ͭCuCl | ��ʽ�Ȼ�ͭCu2��OH��3Cl |
| ���� | ��ɫ���塢����ˮ | ��ɫ���塢����ˮ |
�ں�ɫ���ʿ�����Cu������Cu2O�����߶�����
ʵ���Ϊ̽������̼���ϸ��ŵĺ�ɫ����ɫ���ʣ����������ʵ�飺ȡ������̼����ϴ�ӡ������������ΪW1g�����������ͼ2��ʾװ��b�У�����ʵ�飮
ʵ���У�̼���ϵİ�ɫ������ȫ��Ϊ��ɫ����ˮ����ͭ����ɫ��d�г��ְ�ɫ������ʵ�����ʱ������ͨH2ֱ��̼����ȴ����������ΪW2g��
��3����ˮ����ͭ�������Ǽ����ɫ����������Cu2O��
��4��װ��b�з�����Ӧ�Ļ�ѧ����ʽ��2CuCl+H2$\frac{\underline{\;\;��\;\;}}{\;}$2Cu+2HCl��
��5�����CuCl2��Һʱ�������ϲ�����ɫ���ʵ�ԭ��Ϊ���õ缫��Ӧʽ���ͣ�Cu2++e-+Cl-=CuCl���������ϲ�����ɫ���ʵ����ʵ�����$\frac{{W}_{1}-{W}_{2}}{35.5}$mol��
ʵ���ⶨƯ��Һ��NaClO��Ũ�ȣ�ȷ��ȡ20.00mLϴ��ƿ�ڻ����Һ������������H2O2��Һ��ҡ�ȣ��μ�2��3�η�̪��Һ���� n mol•L-1����ζ����յ㣬��������V mL��
��6���û�ѧ����ʽ��ʾ����H2O2��Һ������NaClO+H2O2=O2��+NaCl+H2O��
��7��������ϴ��ƿ�ڵĸ���Ӧ������仯��Ư��Һ��NaClO��Ũ��Ϊ$\frac{20m-nV}{40}$mol•L-1��
���� ��1��������������ʧ������������������������������Һ���գ�
��2�����ݿ��ܺͼ����ɫ���ʿ���Ϊͭ��������ͭ��
��3����ˮ����ͭ����ɫ˵������̼���ϵIJ�����������Ӧ������û��ˮ��
��4��װ��b�з�����Ӧ�Ļ�ѧ����ʽΪ�Ȼ�������������Ӧ����ͭ���Ȼ��⣻
��5���缫�Ȼ�ͭ��Һ����ͭ���ӵõ���������ͭ���Ӻ��Ȼ���ͭ���ݴ�д�ɵ缫��Ӧ�����ӷ���ʽ�����ݷ�Ӧ2CuCl+H2=2Cu+2HCl����Ӧǰ����������仯Ϊ��Ԫ�������������㣻
��6��NaClO��H2O2��Ӧ����������NaCl��ˮ����ֹ����ζ�ʱ��NaClO��Ӧ��
��7��������Ԫ���غ����Ԫ���غ㣺n��NaOH��=2n��NaClO��+n��NaCl����n��NaCl���ǵζ�����������NaOH��Ӧ���ɵģ���ϵζ�ʱ���ĵ�������㣮
��� �⣺��1��������������ʧ������������������������������Һ���գ�����������X����������W���ʴ�Ϊ��X��
��2�������ݼ�������������������ɫ���ʿ���ΪCu��Cu2O������߶��У��ʴ�Ϊ��Cu��
��3��������ˮ����ͭ����ɫ��֤��b�з�Ӧ����û��ˮ���ɣ�̼���ϵĺ�ɫ������û����Ԫ�أ�һ�����Ậ��������ͭ��һ������ͭ������ˮ����ͭ�������ɫ����������Cu2O��
�ʴ�Ϊ�������ɫ����������Cu2O��
��4��װ��b�з�����ӦΪCuCl��������Ӧ����Ӧ�Ļ�ѧ����ʽΪ��2CuCl+H2=2Cu+2HCl��
�ʴ�Ϊ��2CuCl+H2=2Cu+2HCl��
��5�����CuCl2��Һʱ�������Ϸ����ķ�ӦΪͭ���ӵõ���������ͭ��Cu2++2e-=Cu��ͭ����ʧȥ���������Ȼ���ͭ��Cu2++e-+Cl-=CuCl��2CuCl+H2=2Cu+2HCl����Ӧǰ����������仯Ϊ��Ԫ����������ɫ����ΪCuCl������n��CuCl��=n��Cl��=$\frac{{W}_{1}-{W}_{2}}{35.5}$mol
�ʴ�Ϊ��Cu2++e-+Cl-=CuCl����$\frac{{W}_{1}-{W}_{2}}{35.5}$mol��
��6��NaClO��H2O2��Ӧ����������NaCl��ˮ����ֹ����ζ�ʱ��NaClO��Ӧ���䷴Ӧ�ķ���ʽΪ��NaClO+H2O2=O2��+NaCl+H2O��
�ʴ�Ϊ��NaClO+H2O2=O2��+NaCl+H2O��
��7��������Ԫ���غ����Ԫ���غ㣺n��NaOH��=2n��NaClO��+n��NaCl����n��NaCl���ǵζ�����������NaOH��Ӧ���ɵģ�
��0.1m��$\frac{20mL}{100mL}$=2n��NaClO��+nV��10-3����n��NaClO��=$\frac{1}{2}$��0.02m-nV��10-3��mol��
����c��NaClO��=$\frac{\frac{1}{2}����0.02m-nV��1{0}^{-3}��}{0.02}$=$\frac{20m-nV}{40}$mol/L��
�ʴ�Ϊ��$\frac{20m-nV}{40}$��
���� ���⿼�����ʵļ����������Ŀ�Ѷ��еȣ������ۺ���ǿ������������ѧ������˼ά�����ͷ�ɢ˼ά���������ѧ����Ӧ��������ѧϰЧ�ʣ�����������Ҫע����ǽ������ʵļ���ʱ��Ҫ�������ʵ��������ʺ�������Ӧ��ѡ���ʵ����Լ��ͷ�����

 ϰ�⾫ѡϵ�д�
ϰ�⾫ѡϵ�д�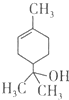
| A�� | 1mol����������ܺ�lmol���������ӳɷ�Ӧ | |
| B�� | ������������ | |
| C�� | ��������ʹ��ˮ��ɫ | |
| D�� | ����ʽΪC9H16O |
��1��ѡȡ��Ҫ��ʵ��װ�ã���ȷ������˳��ΪCAB������ţ���
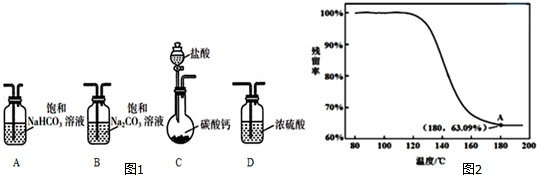
��2��Ϊȷ���ƵõĹ�����Ʒ�Ǵ�����NaHCO3��С��ͬѧ�������ʵ�鷽����
����������Ʒ��Һ�뱥�ͳ���ʯ��ˮ��Ӧ���۲�����
�ҷ���������Ʒ��Һ��BaCl2��Һ��Ӧ���۲�����
���������ⶨpH����
�����������ط�������
����������������������������
��Ϊ�ж��ҷ����Ŀ����ԣ�ijͬѧ�ô�����NaHCO3���Ƶ���Һ����BaCl2��Һ�������Ͻ���ʵ�飬������£�
NaHCO3��Һ BaCl2Ũ�� | 0.2mol•L-1 | 0.1mol•L-1 | 0.02mol•L-1 |
| 0.2mol•L-1 | ���� | ���� | �������� |
| 0.1mol•L-1 | ���� | �������� | ������ |
| 0.02mol•L-1 | �������� | ������ | ������ |
[��֪��0��l mol•L-1 NaHCO3��Һ�������c��CO32-��Ϊ0.001l mol•L-1��Ksp��BaCO3��=5.1��10-9]
��Qc=c��Ba2+����c��CO32-��=$\frac{0.2}{2}$��0.0011=1.1��10-4��5.1��10-9��
��ii���������ǣ�������������������ӷ���ʽBa2++2HCO3-=BaCO3��+CO2��+H2O��
����pH�Ʋⶨ�ı��������£�
ȡm�˵Ĺ��������ܽ���ˮ���V mL����Һ����pH�Ʋ�pH��
��Ӧ�����ʵ���ǣ���ȡ�������ķ�����NaHC03����ˮ�����V mL����Һ����pH�Ʋ�pH
�ܽ��ж�����ʵ�飬�õ�������������¶ȱ仯��������ͼ2��ʾ������A������õ��Ľ������ƵõĹ�����Ʒ�Ǵ�����NaHCO3��
��������=$\frac{ʣ����������}{ԭʼ���������}$��100%��
| A�� | ������ | B�� | 6.72L | C�� | 33.6L | D�� | 22.4L |
| A�� | ���³�ѹ�£�0.1mol D216O�к�������������������������ΪNA | |
| B�� | �Ȼƽ��18O2����ͨ��16O2�����ֲ�ͬ�ĺ��� | |
| C�� | ��״���£�2.24LCl2����ˮ��ת�Ƶĵ�����ĿΪ0.1NA | |
| D�� | 1L2mol•L-1��Al��NO3��3��Һ�к�Al3+����Ϊ2NA |
| A�� | Na��Al��Fe��Cu�ڿ����ж�����������Ϊ�������˶�Ӧ�������� | |
| B�� | �ڸ�������������ԭMgCl2����ȡ����þ | |
| C�� | ʵ����NH4HCO3��Һ�Լ��ԣ�CH3COONH4��Һ�����ԣ�˵������CH3COOH��H2CO3 | |
| D�� | ����ʱ��CuO��Cu2O�ȶ���Na2O2��Na2O�ȶ� |
| A�� | ����£�22.4L SO3����3NA��Oԭ�� | |
| B�� | ���³�ѹ�£�27g Al������NaOH ��Һ��Ӧ����1.5NA��H2 | |
| C�� | ���³�ѹ�£�32g�����������O3 ���Ļ�����к���NA����ԭ�� | |
| D�� | ���ʵ���Ũ��Ϊ1 mol•L-1 �� K2SO4 ��Һ�У���2NA��K+ |
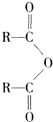 $\stackrel{H_{2}O}{��}$2RCOOH ��RCOOH$\stackrel{LiAlH_{4}}{��}$RCH2OH
$\stackrel{H_{2}O}{��}$2RCOOH ��RCOOH$\stackrel{LiAlH_{4}}{��}$RCH2OH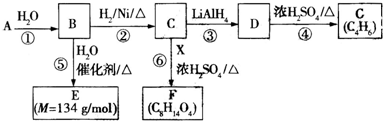
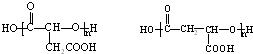 ��һ�ּ��ɣ���
��һ�ּ��ɣ���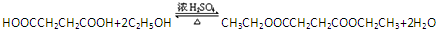 ��
�� ��Y����������������Һ��Ӧ���ò���֮һM���������ȵ�����ͭ��Ӧ��д��M�����ȵ�����ͭ��Ӧ�Ļ�ѧ����ʽ��
��Y����������������Һ��Ӧ���ò���֮һM���������ȵ�����ͭ��Ӧ��д��M�����ȵ�����ͭ��Ӧ�Ļ�ѧ����ʽ��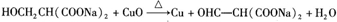 ��
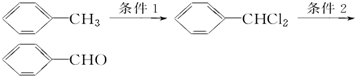
��
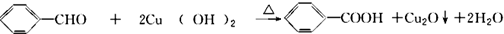 ��
�� ��
��