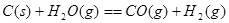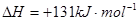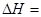��Ŀ����
��֪��Ӧ��Fe(s)+CO2(g) FeO(s)+CO(g)����H="a" kJ��mol-1,ƽ�ⳣ��ΪK;��Ӧ��CO(g)+1/2O2(g)
FeO(s)+CO(g)����H="a" kJ��mol-1,ƽ�ⳣ��ΪK;��Ӧ��CO(g)+1/2O2(g) CO2(g)����H="b" kJ��mol-1;��Ӧ��Fe2O3(s)+3CO(g)
CO2(g)����H="b" kJ��mol-1;��Ӧ��Fe2O3(s)+3CO(g) 2Fe(s)+3CO2(g)����H="c" kJ��mol-1������ڲ�ͬ�¶���,Kֵ����:
2Fe(s)+3CO2(g)����H="c" kJ��mol-1������ڲ�ͬ�¶���,Kֵ����:
| �¶�/�� | 500 | 700 | 900 |
| K | 1.00 | 1.47 | 2.40 |
(1)��500 ��ʱ���з�Ӧ��,CO2����ʼŨ��Ϊ2 mol��L-1,CO��ƽ��Ũ��Ϊ����������
(2)��Ӧ��Ϊ��������(ѡ����ȡ����ȡ�)��Ӧ��
(3)700 ��ʱ��Ӧ�ٴﵽƽ��״̬,Ҫʹ��ƽ�������ƶ�,������������ʱ,���Բ�ȡ�Ĵ�ʩ����������(�����)��
A.��С��Ӧ����� B.ͨ��CO2 C.�¶����ߵ�900 �� D.ʹ�ú��ʵĴ���
E.����Fe����
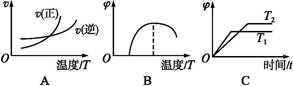
(4)����ͼ����Ϸ�Ӧ�ٵ�����������(�����)(ͼ��vΪ����,��Ϊ�������CO����,TΪ�¶���T1>T2)��
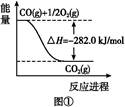
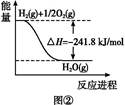
(5)�ɷ�Ӧ�ٺ͢ڿ����,��Ӧ2Fe(s)+O2(g)
 2FeO(s)�Ħ�H=����������
2FeO(s)�Ħ�H=���������� (6)�����ø�˹����д��Fe(����)��O2(����)�����õ�Fe2O3(����)���Ȼ�ѧ����ʽ:����
(1)1 mol��L-1��(2)���ȡ�(3)BC��(4)A
(5)2(a+b) kJ��mol-1��(6)2Fe(s)+3/2O2(g) Fe2O3(s)����H="(3b-c)" kJ��mol-1
Fe2O3(s)����H="(3b-c)" kJ��mol-1
����

(1)�µġ�����������������(GB 3095 2012)����2016��1��1�����ҹ�ȫ��ʵʩ���ݴ�,������������ָ��(AQI)�ձ���ʵʱ���������SO2��NO2��CO��O3��PM10��PM2.5��ָ��,Ϊ�����ṩ����ָ��,�������ؾ���������ų��к����
2012)����2016��1��1�����ҹ�ȫ��ʵʩ���ݴ�,������������ָ��(AQI)�ձ���ʵʱ���������SO2��NO2��CO��O3��PM10��PM2.5��ָ��,Ϊ�����ṩ����ָ��,�������ؾ���������ų��к����
�������ų���β���к���CO��NO������,�û�ѧ����ʽ���Ͳ���NO��ԭ�� ��
�������������ڰ�װ�Ĵ�ת����,��ʹ����β���е���Ҫ��Ⱦ��ת��Ϊ���Ĵ���ѭ�����ʡ���֪:
N2(g)+O2(g)="2NO(g)" ��H="+180.5" kJ/mol
2C(s)+O2(g)="2CO(g)" ��H="-221.0" kJ/mol
C(s)+O2(g)=CO2(g) ��H="-393.5" kJ/mol
��Ӧ2NO(g)+2CO(g)=N2(g)+2CO2(g)�Ħ�H= kJ/mol��
(2)ֱ���ŷŵ���������γ����ꡢ����,����ԭ�����������շ��dz��õĴ�������������NH3��CH4�������ȥ�����еĵ��������֪:CH4(g)+2O2(g)=CO2(g)+2H2O(l) ��H1="a" kJ/mol;�����㷴ӦCH4(g)+4NO(g)=CO2(g)+2H2O(l)+2N2(g)���ʱ䦤H2����Ҫ��ѯij��Ӧ���ʱ䦤H3,����Ӧ�и����ʵĻ�ѧ������֮��Ϊ���������ʱ,��H3="b" kJ/mol,�÷�Ӧ���Ȼ�ѧ����ʽ�� ,�ݴ˼������H2= kJ/mol(�ú�a��b��ʽ�ӱ�ʾ)��
(3)�±��г��˹�ҵ������SO2�����ַ�����
| ������ | �ð�ˮ��SO2ת��(NH4)2SO3,��������(NH4)2SO4 |
| ������ | ���������Ƚ���(��Ҫ�ɷ�CO��CH4��H2)��SO2�ڸ����»�ԭ�ɵ����� |
| ������ | ��Na2SO3��Һ����SO2,�پ����ת��ΪH2SO4 |
2CO(g)+SO2(g)=S(g)+2CO2(g) ��H="+8.0" kJ/mol
2H2(g)+SO2(g)=S(g)+2H2O(g)��H="+90.4" kJ/mol
2CO(g)+O2(g)=2CO2(g) ��H="-566.0" kJ/mol
2H2(g)+O2(g)=2H2O(g) ��H="-483.6" kJ/mol
��S(g)��O2(g)��Ӧ����SO2(g)���Ȼ�ѧ����ʽ�ɱ�ʾΪ ��
(4)�ϳɰ��õ��������Լ���Ϊԭ���Ƶá��йػ�ѧ��Ӧ�������仯��ͼ��ʾ,��CH4(g)��H2O(g)��Ӧ����CO(g)��H2(g)���Ȼ�ѧ����ʽΪ ��
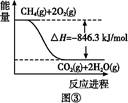
ij������ȼ���ɼס��������л����϶��ɣ��ס����������ʺ���C��H��O����Ԫ���е����ֻ����֡���֪�ס��Ҽ�CO��H2��ȼ�������£�
| ���� | �� | �� | CO | H2 |
| ȼ����/(kJ��mol��1) | 1 366 | 5 518 | 283 | 286 |
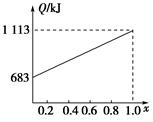
ȡ�ס��Ұ���ͬ������ϵ�ȼ��23 g����������O2��ȼ��ʱ���ų�������Q���������ҵ����ʵ�������x�Ĺ�ϵ��ͼ��ʾ������
(1)�ҵ���Է�������Mr(��)��________��
(2)160 g�ɼס����Ե����ʵ�����϶��ɵ�ȼ����347.2 L O2��ǡ����ȫȼ�գ���492.8 L���壬��ȴ������ʱ����ʣ��224 L(����������ڱ�״���²ⶨ)���ɴ˿���û�����У�C��H��O��ԭ�Ӹ�����Ϊ________���ס��ҵķ���ʽΪ����________����________��
(3)1 mol�ɼס����Ե����ʵ�����϶��ɵ�ȼ����һ������O2��ȼ�գ��ų�����2 876 kJ����Ӧ������CO________mol��
��15�֣����Ŵ�����Ⱦ���������أ��������ڡ�ʮ���塱�ڼ䣬����������(SO2)�ŷ�������8%����������(NOx)�ŷ�������10%��Ŀǰ������������Ⱦ�ж��ַ�����
(1)��CH4����ԭ��������������������������Ⱦ����֪��
��CH4(g)+4NO2(g)��4NO(g) + CO2(g) +2H2O(g) �SH�� -574 kJ��mol��1
��CH4(g) +4NO(g)��2N2(g) + CO2(g) + 2H2O(g) �SH��-1160 kJ��mol��1
��H2O(g)��H2O(l) ��H��-44.0 kJ��mol��1
д��CH4(g)��NO2(g)��Ӧ����N2 (g)��CO2 (g)��H2O(1)���Ȼ�ѧ����ʽ ��
��2������Fe2+��Fe3+�Ĵ����ã������¿ɽ�SO2ת��ΪSO42-���Ӷ�ʵ�ֶ�SO2����������֪��SO2�ķ���ͨ�뺬Fe2+��Fe3+����Һʱ������һ����Ӧ�����ӷ���ʽΪ4Fe2+ + O2+ 4H+ ��4Fe3+ + 2H2O������һ��Ӧ�����ӷ���ʽΪ ��
(3)�û���̿��ԭ��������������йط�ӦΪ��C(s)+2NO(g) N2(g)+CO2(g)��ij�о�С�����ܱյ���������У���������������䣬��������������Բ��ƣ�����NO�������Ļ���̿������(T1��)�����·�Ӧ����Ӧ���е���ͬʱ���ø����ʵ�Ũ�����£�
N2(g)+CO2(g)��ij�о�С�����ܱյ���������У���������������䣬��������������Բ��ƣ�����NO�������Ļ���̿������(T1��)�����·�Ӧ����Ӧ���е���ͬʱ���ø����ʵ�Ũ�����£�
Ũ��/mol��L��1
| NO | N2 | CO2 | ||
| 0 | 1.00 | 0 | 0 | ||
| 10 | 0.58 | 0.21 | 0.21 | ||
| 20 | 0.40 | 0.30 | 0.30 | ||
| 30 | 0.40 | 0.30 | 0.30 | ||
| 40 | 0.32 | 0.34 | 0.17 | ||
| 50 | 0.32 | 0.34 | 0.17 |
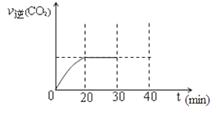
��10min��20min��v(CO2) ��ʾ�ķ�Ӧ����Ϊ ��
�ڸ��ݱ������ݣ�����T1��ʱ�÷�Ӧ��ƽ�ⳣ��Ϊ ���� ��������λС������
��һ���¶��£�����NO����ʼŨ��������NO��ƽ��ת���� ������������䡱��С���� ��
�����и�������Ϊ�жϸ÷�Ӧ�ﵽƽ����� ������ ���������ĸ����
A��������ѹǿ���ֲ��䡡�� B��2v��(NO)��v��(N2)
C��������CO2������������� D�����������ܶȱ��ֲ���
��30minĩ�ı�ijһ��������һ��ʱ�䷴Ӧ���´ﵽƽ�⣬��ı������������ ��������ͼ�л���30min��40min�ı仯���ߡ�
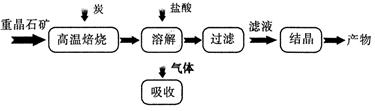
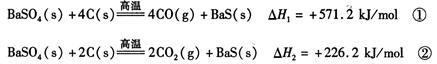
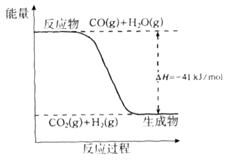
 2CO(g)�ġ�H = kJ/mol
2CO(g)�ġ�H = kJ/mol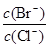 = ��[��֪��
= ��[��֪��