��Ŀ����
�������и��������������ݣ��ɷֱ�����䡰���ʵ����������������ʵ����ʵ���Ũ�ȡ������жϲ���⡣
��1����NA��ʾ�����ӵ���������ֵ����ij����������ҺV L�к���N��OH-������������Һ��______Ϊ______��
��2����֪ij����������Һ��Na����H2O�ĸ���֮��Ϊ1��a������������Һ��______Ϊ______��
��3����֪��״����1���ˮ���ܽ�500������Ȼ��⣬��������״�����Ȼ��ⱥ����Һ��______Ϊ______��
��4����֪��100 mL�Ȼ�����ˮ��Һ�����������գ��ɵõ���ɫ����b g��������ԭ�Ȼ�����Һ��______Ϊ______��
��1��NaOH�����ʵ���Ũ�� 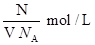
��2��NaOH���������� 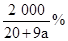
��3��HCl���������� 44.9%
��4��AlCl3�����ʵ���Ũ�� 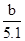 mol/L
mol/L
����

��.ʵ��������1mol/L Na2CO3��Һ250ml��
��1����Ҫ����Na2CO3 g����2������Һ�е���������ĿΪ ����
��3����Ҫ���ʵ���Ũ��Ϊ5mol/L ��Na2CO3��Һ ml��
��4��������Һ������ϡ���ᷴӦ�������������ڱ�״���µ����Ϊ L��
��5�����Ƹ���Һ�IJ���˳����(����ĸ��ʾ,���ظ�ʹ��) ��
| A������ | B��ϴ�� | C������ | D���ܽ� E��ҡ�� F��ת�� |
�ش��������⣨����ţ���
��1�����������У���©����������ƿ����������ƿ������ƽ���ݷ�Һ©��������Ͳ��
��ȼ�ճס����������ʷ������________________�����и������ʷе㲻ͬ���������ʵ�������____________�����������д��
��2��������NaOH��������220mL 0.2mol/L��NaOH��Һ������������գ�
| A�����Ƹ���ҺӦѡ�ò�������_________mL����ƿ�� |
| B����������ƽ��ȡ ________g NaOH���塣 |
| C�����ƺõ�NaOH��������ձ��У�����Լ25mL����ˮ����__________��������ȫ�ܽ⡣����ȴ�����º��ձ��е���Һ�ò���������ת��������ƿ�� |
| D������������ˮϴ���ձ��ڱںͲ�����2-3�Σ�����ÿ��ϴ�ӵ���Һ��ע������ƿ�� ����ҡ������ƿ��ʹ��Һ��;��ȡ� |
F�����ƺõ���Һ__________����ܡ����ܡ������ڴ��������ƿ�С�
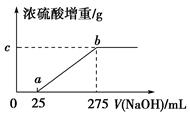
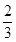 ʱ��b�� ���õ���CO��CO2�����ʵ���֮��n��CO����n��CO2���� ��
ʱ��b�� ���õ���CO��CO2�����ʵ���֮��n��CO����n��CO2���� ��