��Ŀ����
���������ƷA����(NH4)2SO4��NH4HSO4�Ļ���Ϊȷ��A�и��ɷֵĺ�����ij�о���ѧϰС���ͬѧȡ��������ͬ��������ƷA����ˮ��Ȼ��ֱ���벻ͬ�����1 mol/L��NaOH��Һ��ˮԡ����������ȫ���ݳ�(���¶��£���β��ֽ�)��������������������Ũ������ȫ���ա�Ũ�������ص����������NaOH��Һ������Ĺ�ϵ��ͼ��ʾ��������ͼ�ش��������⣺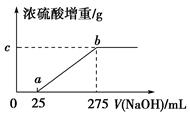
(1)д��ab���漰�����ӷ���ʽ��_____________________________��
(2)c���Ӧ����ֵ��________����ƷA��(NH4)2SO4��NH4HSO4�����ʵ���֮��Ϊ________��
��(1)NH4+��OH��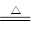 NH3����H2O
NH3����H2O
(2)4.25��9��2
����

 ��������ϵ�д�
��������ϵ�д� ����˼ά����ѵ����ʱ��ѧ��ϵ�д�
����˼ά����ѵ����ʱ��ѧ��ϵ�д�����500 mL 0.5 mol/L��NaOH��Һ,�Իش���������:
(1)����:��ҪNaOH���������Ϊ ��
(2)���Ʒ���:�������������衣
����ʢ��NaOH������ձ��м���200 mL����ˮʹ���ܽ�,����ȴ������;
�ڼ���������ƿ�м�����ˮ��Һ��ӽ��̶���1��2 cm;
�۽�NaOH��Һ�ز�����ע��500 mL����ƿ��;
��������������ˮϴ���ձ��Ͳ�����2��3��,Ȼ��ϴ��Һ��������ƿ;
�ݸ��ý�ͷ�ιܼ�����ˮ���̶���,�Ӹ�ҡ�ȡ�
�Խ�����������ȷ���� ��
(3)ijѧ��ʵ������NaOH��Һ��Ũ��Ϊ0.48 mol/L,ԭ������� ��
| A��ʹ����ֽ�����������ƹ��� |
| B������ƿ��ԭ��������������ˮ |
| C���ܽ����ձ�δ�����ϴ�� |
| D����ͷ�ιܼ�ˮ����ʱ���ӿ̶��� |

 2CuSO4��2H2O��������6.4 gͭ��ȫ�ܽ⣬��ˮ���200 mL��Һ�����㣺
2CuSO4��2H2O��������6.4 gͭ��ȫ�ܽ⣬��ˮ���200 mL��Һ�����㣺