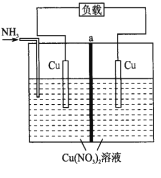
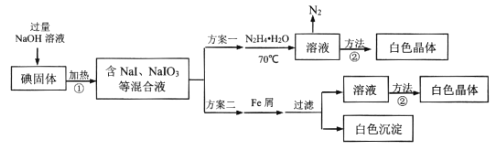
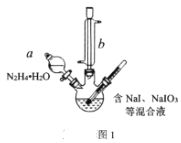
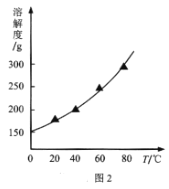
��Ŀ����
����Ŀ��ʵ��������NaOH��������1.0mol��L-1��NaOH��Һ480mL��
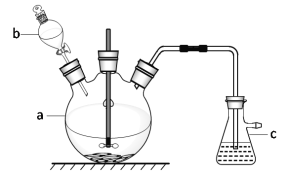
��1������ʱ������ʹ�õIJ���������___��
��2��Ҫ��ɱ�ʵ���ͬѧӦ�Ƴ�NaOH__g��
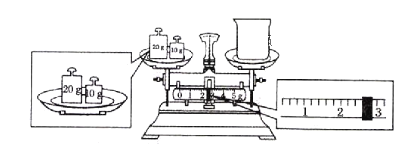
��3��ijͬѧ������NaOH��������������������ƽ�����ձ�����������ƽƽ����״̬��ͼ���ձ���ʵ������Ϊ__g��(����һ��20�ˡ�һ��10��)
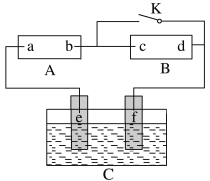
��4��ʹ������ƿǰ������е�һ��������__��
��5�������ƹ����У���������������ȷ�ģ����в������������ƫ�ߵ���__��
��û��ϴ���ձ��Ͳ�����
�ڶ���ʱ���ӿ̶���
��δ��ȴ�����¾ͽ���Һת�Ƶ�����ƿ������
�ܶ��ݺ�����ƿ������ҡ�ȣ����ú�Һ����ڿ̶��ߣ��ټ�ˮ���̶���
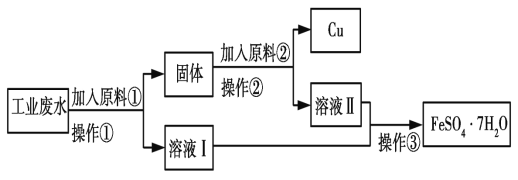
���𰸡�500mL����ƿ����ͷ�ιܡ��ձ��������� 20.0 27.4 ��© �ڢ�
��������
��1������ʵ������IJ����Լ�ÿ��������Ҫ����ȷ����Ӧ����������
��2������n=cV������������Ƶ����ʵ������ٸ���m=nM���������������Ƶ�������
��3��������ƽƽ��ԭ��Ϊ����������=����������+������ֵ��
��4�������ߵ�ҡ�ȣ���������ƿ��ʹ��ǰ�������Ƿ�©ˮ��
��5���������������ʵ����ʵ��������Һ�������Ӱ�죬����c=n/V�����жϡ�
��1��û��480mL����ƿ������ѡ��500mL����ƿ�����������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣬�ò��������裬�����ܽ⣬�ָ����º�ת�Ƶ�500mL����ƿ�У����ò�����������ϴ��2��3�Σ�����ϴ��Һ��������ƿ�У�����ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�����ݵߵ�ҡ�ȡ�������Ҫ��������������ƽ���ձ���500ml������ƿ����ͷ�ιܡ���������
��2�����������Ƶ�����Ϊm=0.5L��1molL-1��40g/mol=20.0g��
��3����ͼ��֪���ձ�������λ�õߵ��ˣ������ձ���ʵ������Ϊ30.0g-2.6g=27.4g��
��4�������ߵ�ҡ�ȣ���������ƿ��ʹ��ǰ�������Ƿ�©ˮ��
��5����δϴ���ձ�����������������������մ���ձ����벣�����ϣ��������Ƶ�ʵ��������С����ҺŨ��ƫ�ͣ�
�ڶ���ʱ���ӿ̶��ߣ�������Һ���ƫС��������ҺŨ��ƫ�ߣ�
��Һ������������������ʣ����������ܽ���ȣ�δ��ȴ�����£����Ƚ���Һ��������ƿ���������Һ���¶Ȼָ����ºᵼ����Һ���ƫС����ҺŨ��ƫ�ߣ�
��ҡ�Ⱥ�Һ���½���һ������Һ����ƿ����ƿ��֮�䣬�ټ�����ˮ���̶��ߣ�������Һ���ƫ��������ҺŨ��ƫ�͡�
��ѡ���ڢۡ�

 ABC����ȫ�ž�ϵ�д�
ABC����ȫ�ž�ϵ�д�����Ŀ����![]() ��
��![]() ��
��![]() ������ȣ��ֱ�ͨ�뵽���Ϊ2L�ĺ����ܱ������н��з�Ӧ��
������ȣ��ֱ�ͨ�뵽���Ϊ2L�ĺ����ܱ������н��з�Ӧ��![]() ���õ������������ݣ�����ʵ��١���û��ʹ�ô���������˵������ȷ���ǣ� ��
���õ������������ݣ�����ʵ��١���û��ʹ�ô���������˵������ȷ���ǣ� ��
ʵ�� | �¶�/�� | ��ʼ | ƽ�� | ��ƽ������ʱ��/min |
�� | 500 | 5.00 | 2.00 | 40 |
�� | 800 | 3.00 | 1.00 | 15 |
�� | 500 | 3.00 | 1.20 | 9 |
A. ʵ����У���![]() �ڣ���
�ڣ���![]() ��ʾ�ķ�Ӧ���ʴ���
��ʾ�ķ�Ӧ���ʴ���![]()
B. �Ƚ�ʵ��ٺ͢ڣ�����˵��Ũ�ȶԷ�Ӧ���ʵ�Ӱ��
C. �Ƚ�ʵ��ٺ͢ڣ�����˵����Ӧ�������¶����߶�����
D. �Ƚ�ʵ��ںۣ͢�˵��ʵ���ʹ���˴���