��Ŀ����
9��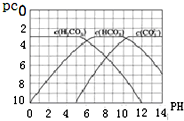 pC��ָ��ϡ��Һ���������ʵ���Ũ�ȵij��ö�����ֵ������pH����ij��Һ���ʵ�Ũ��Ϊ1��10-3 mol•L-1�������Һ�и����ʵ�pC=-lg10-3=3����֪H2CO3��Һ�д�������ƽ�⣺CO2+H2O?H2CO3��H2CO3?H++HCO3-��HCO3-?H++CO32-����ͼΪH2CO3��HCO3-��CO32-�ڼ���ǿ���ǿ����Һ�ﵽƽ��ʱ��Һ�����ֳɷֵ�pCpHͼ��
pC��ָ��ϡ��Һ���������ʵ���Ũ�ȵij��ö�����ֵ������pH����ij��Һ���ʵ�Ũ��Ϊ1��10-3 mol•L-1�������Һ�и����ʵ�pC=-lg10-3=3����֪H2CO3��Һ�д�������ƽ�⣺CO2+H2O?H2CO3��H2CO3?H++HCO3-��HCO3-?H++CO32-����ͼΪH2CO3��HCO3-��CO32-�ڼ���ǿ���ǿ����Һ�ﵽƽ��ʱ��Һ�����ֳɷֵ�pCpHͼ����ش��������⣺
��1����pH=9ʱ��H2CO3��Һ��Ũ�����ĺ�̼Ԫ�ص�����ΪHCO3-��
��2��pH��4ʱ����Һ��H2CO3��pC����Լ����3��ԭ����c��H+�������H2CO3
 H++HCO3-ƽ�������ƶ��ų�CO2��̼��Ũ�ȱ��ֲ��䣮
H++HCO3-ƽ�������ƶ��ų�CO2��̼��Ũ�ȱ��ֲ��䣮��3����֪M2CO3Ϊ���������Ksp�ı���ʽΪKsp=c2��M+��•c��CO32-����������ij��Һ�е�M+��̼���Σ�Ksp=1��10-12������ʽ������ȫ���������Һ�е�CO32-��pC���ֵΪ2����Һ�е�����Ũ��С��1��10-5 mol•L-1ʱ��������ȫ����
���� ��1������pC����֪��pCֵԽ������Ũ��ԽС����֮��pCֵԽС������Ũ��Խ����ͼ������жϣ�
��2��������Ũ��Խ��Խ����̼����룻�����ж�����̼���ɣ�
��3��Ksp=c2��M+��•c��CO32-���������ܶȻ�����������Һ��c��CO32-�����ٸ���pC�������pCֵ
��� �⣺��1������pC����֪��pCֵԽ������Ũ��ԽС����֮��pCֵԽС������Ũ��Խ����ͼ��֪��pH=9ʱ��pC��С����HCO3-������HCO3-Ũ�����
�ʴ�Ϊ��HCO3-��
��2��pH��4ʱ����Һ�У�c��H+�������H2CO3?H++HCO3-ƽ�������ƶ��ų�CO2������̼��Ũ�Ȳ��䣬̼��Ϊ������Һ��������Һ��H2CO3��pC����Լ����3��
�ʴ�Ϊ��c��H+�������H2CO3?H++HCO3-ƽ�������ƶ��ų�CO2��̼��Ũ�ȱ��ֲ��䣻
��3���ܶȻ�����Ϊ����Ũ��ϵ������֮������M2CO3���ܶȻ�ΪKsp=c2��M+��•c��CO32-������Һ��c��CO32-��=$\frac{{K}_{sp}}{{c}^{2}��{M}^{+}��}$=$\frac{1.0��1{0}^{-12}}{��1.0��1{0}^{-5}��^{2}}$mol/L=0.01mol/L����pC=-lg0.01=2��
�ʴ�Ϊ��Ksp=c2��M+��•c��CO32-����2��
���� ���⿼����������ʵĵ��롢pC�ļ��㣬��ȷpC�ĸ����ǽⱾ��ؼ������Բ���֪ʶǨ�Ƶķ�������pC����������ܶȻ�������֪ʶ�������������Ŀ�Ѷ��еȣ�

 һŵ��ҵ�����ҵ���ּ�����������������ϵ�д�
һŵ��ҵ�����ҵ���ּ�����������������ϵ�д�| A�� | NaCl | B�� | MgCl2 | C�� | K2SO4 | D�� | NH3 |
| A�� | ������ڷֱ�ϡ����ͬ��������Һ��pH���٣��� | |
| B�� | ������ۻ�Ϻ���Һ�����ԣ����ڳ�����Ka��CH3COOH��=Kb��NH3��NH3�� | |
| C�� | ��������ѵ����ˮ���ӵ���Ŀ��� | |
| D�� | ����ܻ��������Һ�����ԣ���c��CH3COO-��+c��OH-����c��CH3COOH��+c��H+�� |
| A�� | 0.01molMg�ڿ�������ȫȼ������MgO��Mg3N2��ת�Ƶ�����ĿΪ0.02NA | |
| B�� | ���³�ѹ�£�18.00����ˮ��D2O��������������ԼΪ10NA | |
| C�� | ������״̬��1molNaHSO4������������Ϊ2NA | |
| D�� | 1mol FeCl3��ȫˮ��ת��Ϊ�������������������NA������ |
| A | B | C | D | |
| ���� | AOH��BOH | AOH��BOH | AOH��BOH | AOH��BOH |
| ���� | HC��HD | HC��HD | HC��HD | HC��HD |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ��ơ��ƿ��ƿ���������������ĭ | |
| B�� | ��ҵ��ȡ�����أ�Na��l��+KCl��l��?NaCl��l��+K��g����ѡȡ���˵��¶ȣ�ʹK�������ӷ�Ӧ������з������ | |
| C�� | ����ˮ�д�������ƽ�⣺Br2+H2O?HBr+HBrO��������NaOH��Һ����ɫ��dz | |
| D�� | ����2HI��g��?H2��g��+I2��g��ƽ����ϵ����ѹǿʹ��ɫ���� |
| A�� | CuSO4��Һ | B�� | NaOH��Һ | C�� | Fe2O3 | D�� | Ũ���� |
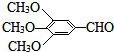 ���Ϳ����ҩ��H��
���Ϳ����ҩ��H��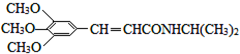 ����·����ͼ�����ַ�Ӧ��ȥ�������Լ�����
����·����ͼ�����ַ�Ӧ��ȥ�������Լ�����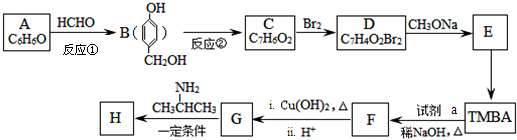
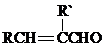 +H2O��R��R���ʾ�������⣩
+H2O��R��R���ʾ�������⣩ ���Լ�a�ķ���ʽ��C2H4O��
���Լ�a�ķ���ʽ��C2H4O�� +2Br2��
+2Br2��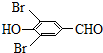 +2HBr��
+2HBr��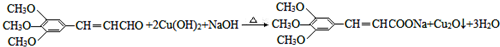 ��
�� ������������ˮ������ʣ�д������ﲡҩ��H������NaOH��Һ������Ӧ�Ļ�ѧ����ʽ
������������ˮ������ʣ�д������ﲡҩ��H������NaOH��Һ������Ӧ�Ļ�ѧ����ʽ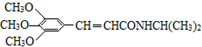 +NaOH��
+NaOH��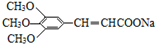 +H2NCH��CH3����
+H2NCH��CH3����