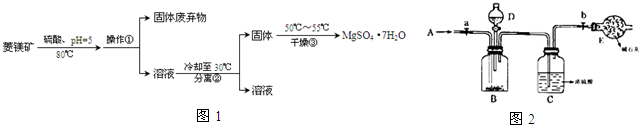
��Ŀ����
9��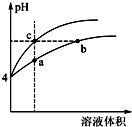 ���ᡢ�����̼���ǻ�ѧʵ����о��г��õļ����ᣮ
���ᡢ�����̼���ǻ�ѧʵ����о��г��õļ����ᣮ��֪�����£�Ka��CH3COOH��=1.7��10-5 mol•L-1��H2CO3�ĵ��볣��Ka1=4.2��10-7mol•L-1��Ka2=5.6��10-11mol•L-1
��1�������£����ʵ���Ũ����ͬ������������Һ��a������������Һb������������Һc��̼������Һd��̼��������Һe����������Һ����PH�ɴ�С��˳���ǣ�a��b��c��d��e������ţ���
��2��ij�¶��£�pH��Ϊ4������ʹ�����Һ�ֱ��ˮϡ�ͣ���pH����Һ����仯������ͼ��a��b��c�����Ӧ����Һ��ˮ�ĵ���̶��ɴ�С��˳����b=c��a���ô�����Һϡ�����У����и���һ����С����ad��
a��c��H+�� b��c��OH-�� c��$\frac{c��C{H}_{3}COOH��c��O{H}^{-}��}{c��C{H}_{3}CO{O}^{-}��}$ d��$\frac{c��C{H}_{3}COOH��}{c��{H}^{+}��}$
��3����0.10mol•L-1NaOHΪ��Һ���ⶨij�����Ũ�ȣ�ȡ20.00mL����������Һ������ƿ�У����μ�2��3�η�̪��ָʾ������NaOH����Һ���еζ����ظ������ζ�����2��3�Σ���¼�������£�
| ʵ���� | ����������Һ��Ũ�ȣ�mol•L-1�� | �ζ����ʱ������������Һ����������mL�� | ��������������mL�� |
| 1 | 0.10 | 24.12 | 20.00 |
| 2 | 0.10 | 23.88 | 20.00 |
| 3 | 0.10 | 24.00 | 20.00 |
�ڸ����������ݣ��ɼ�����������Ũ��ԼΪ0.12mol/L��
��4����t��ʱ��ijNaOHϡ��Һ��c��H+��=10-a mol•L-1��c��OH-��=10-b mol•L-1����֪a+b=12����
�ٸ��¶���ˮ�����ӻ�����Kw=10-12mol2•L-2��
���ڸ��¶��£���100mL0.1mol•L-1��ϡH2SO4��100mL0.4mol•L-1��NaOH��Һ��Ϻ���Һ��pH=11��
���� ��1������������Ũ��Խ����Һ��pHԽ��ˮ��̶ȴ�����Ӷ�Ӧ�ļ���Խǿ���ݴ˻ش�
��2��pH��ȵĴ����������Һ�У���ˮϡ�ʹٽ�������룬�Ȼ�����ȫ���룬����ϡ����ͬ�ı�����pH�仯��������ᣬ�仯С���Ǵ��
������ˮ���룬�������Խǿ��ˮ�ĵ���̶�ԽС��
��ˮϡ�ʹ��ᣬ�ٽ�������룬��Һ�г������������ӡ�ˮ�����⣬������Ũ�ȶ���С��
��3��������Һ��ɫ�仯�Ұ�����ڲ���ɫ����˵���ﵽ�ζ��յ㣻
�����ж����ݵĺ����ԣ������NaOH��Һ�����Ȼ������c�����⣩=$\frac{c������V������}{V�����⣩}$������ɣ�
��4������Һ�е����ӻ�Kw=C��H+����c��OH-��=10-a��10-b=10-��a+b��=10-12��
�ڸ��ݻ����Һ������Լ���������Ũ�ȣ��Ӷ�������Һ��pH��
��� �⣺��1�����ʵ���Ũ����ͬ��������Һ��c��d��ǿ���������������������Ũ�ȴ�����pH��ab��������ˮ�⣬������Һ��ʾ���ԣ���������ӵ�ˮ��̶�С��̼��������ӣ�̼��������ӵ�ˮ��̶�С��̼������ӣ�����̼���Ƶļ���ǿ��̼�����ƣ�̼�����Ƶļ���С�ڴ����ƣ���pH��С˳���ǣ�a��b��c��d��e��
�ʴ�Ϊ��a��b��c��d��e��
��2��pH��ȵĴ����������Һ�У���ˮϡ�ʹٽ�������룬�Ȼ�����ȫ���룬����ϡ����ͬ�ı�����pH�仯��������ᣬ�仯С���Ǵ��ᣬ������ˮ���룬�������Խǿ��ˮ�ĵ���̶�ԽС��������Һ���Խ��ˮ�ĵ���̶�Խ����ˮ�ĵ���̶���a��b��c������Һ��ˮ�ĵ���̶��ɴ�С��˳����b=c��a����ˮϡ�ʹ��ᣬ�ٽ�������룬��Һ�г������������ӡ�ˮ�����⣬������Ũ�ȶ���С��
a����Һ��c��H+����С������ȷ��
b���¶Ȳ��䣬ˮ�����ӻ��������䣬������Ũ�ȼ�С����c��OH-�����ʴ���
c��c.$\frac{c��C{H}_{3}COOH��c��O{H}^{-}��}{c��C{H}_{3}CO{O}^{-}��}$=$\frac{{K}_{W}}{{K}_{a}}$���¶Ȳ��䣬ˮ�����ӻ��������䡢����ĵ���ƽ�ⳣ�����䣬����$\frac{c��C{H}_{3}COOH��c��O{H}^{-}��}{c��C{H}_{3}CO{O}^{-}��}$ ���䣬�ʴ���
d����ˮϡ�ʹٽ�������룬�������Ӹ�����������Ӹ�����С������$\frac{c��C{H}_{3}COOH��}{c��{H}^{+}��}$��С������ȷ��
�ʴ�Ϊ��b=c��a��a d��
��3���ٵζ�ʱ������Һ��ɫ�仯�Ұ�����ڲ���ɫ����˵���ﵽ�ζ��յ㣬����̪������Ϊ��ɫ���ڼ�����Һ����dz��ɫ��
�ʴ�Ϊ�����һ��NaOH��Һ���룬��Һ����ɫǡ�ñ��dz��ɫ�Ұ�����ڲ���ɫ��
����ȥ���ϴ�������ó�V������=$\frac{23.88+24.00}{2}$=23.94mL��c�����⣩=$\frac{0.1mol/L��23.94��L}{20.00mL}$=0.12mol/L���ʴ�Ϊ��0.12mol/L��
��4������Һ�е����ӻ�Kw=C��H+����c��OH-��=10-a��10-b=10-��a+b��=10-12���ʴ�Ϊ��10-12��
�����������ӵ����ʵ���=0.1mol/L��2��0.1L=0.02mol���������������ӵ����ʵ���=0.4mol/L��0.1L=0.04mol/L�������Һ������������Ũ��=$\frac{0.04mol-0.02mol}{0.2L}$=0.1mol/L����Һ��������Ũ��Ϊ10-11mol/L������Һ��pH=11��
�ʴ�Ϊ��11��
���� ������Ҫ����������ʵĵ��롢�ζ�������pH���йؼ����֪ʶ���ѶȲ���

 ȫ�Ų��Ծ�ϵ�д�
ȫ�Ų��Ծ�ϵ�д� ���100��1�ž�ϵ�д�
���100��1�ž�ϵ�д�| A�� | ʹȼ�ϳ��ȼ�� | B�� | �Ӵ���Դ�������� | ||
| C�� | ��ǿ��Դ���õĿƼ�Ͷ�� | D�� | ����ʹ����Դ |
| A�� | V${\;}_{N{H}_{3}}$=$\frac{4}{5}$V${\;}_{{O}_{2}}$ | B�� | V${\;}_{N{H}_{3}}$=V${\;}_{{O}_{2}}$=V${\;}_{{H}_{2}O}$ | ||
| C�� | V${\;}_{{O}_{2}}$=$\frac{4}{5}$VNO | D�� | V${\;}_{{O}_{2}}$=$\frac{6}{5}$V${\;}_{{H}_{2}O}$ |
| A�� | ϡ�������þƬ�ϣ�Mg+2H+�TMg2++H2�� | |
| B�� | ����þ��Һ������������Һ��Ӧ��Ba2++SO42-�TBaSO4�� | |
| C�� | ��������Һ�����ᷴӦ��Ag++Cl-�TAgCl�� | |
| D�� | ����ʯ��ˮ��̼������Һ��Ӧ��Ca��OH��2+CO32-�TCaCO3��+H2O |
| A�� | 뭡�뮡�� | B�� | ���ס����� | C�� | ���ʯ��ʯī��C60 | D�� | O2��O3 |