��Ŀ����
Cl2��H2O2�Ǹ��н����������������������������Cl2��������ǿ��H2O2���ܽ�H2O2������Ϊ����֤�ý��ۣ�ѧ�����������ͼ��ʾ��ʵ��װ�ý���ʵ�飨�г�װ����ȥ����Բ����ƿA�еķ�Ӧ����ʽΪ2KMnO4+16HCl��Ũ��=2KCl+2MnCl2+5Cl2��+8H2O����ش���������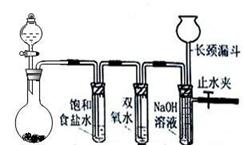
A B C D
��1���Թ�B�б���ʳ��ˮ������ ��
��2���Թ�C�м���5mL 30% ˫��ˮ��������Ӧ�Ļ�ѧ����ʽ____________________��
��3���Թ�D�г���10% NaOH��Һ��NaOH��Һ�������� ��
��4����Ũ����ע��Բ����ƿA����װ���еĿ��������ž���ر�ֹˮ�У���Ӧһ��ʱ����Թ�D�е�����Ϊ ���Թ�D�е�������鷽��Ϊ__________________��
��5���е�ͬѧ������ʵ�����Թ�D��������Դ�������ɣ�����Ϊ���ܵ���Դ�� ���û�ѧ����ʽ��ʾ�����������ɿ��Բ��öԱ�ʵ���������
��1����ȥCl2�л��е�HCl����2��Cl2+H2O2=2HCl+O2����3�����ն����Cl2����4���Թ�D��Һ���½�������©����Һ����������������ʹ�����ǵ�ľ����ȼ��֤������������5��2H2O2=2H2O+O2����Cl2+H2O="HCl+HClO" ��2HClO=2HCl+O2����
���������������1���Ȼ��⼫������ˮ������������ˮ������ˮ��Ӧ��Cl2+H2O=H++Cl-+HClO��ʳ��ˮ��Һ�е������������������ܽ⣬�����������ܽ�ȣ��ʱ���ʳ��ˮ�������dz�ȥCl2�л��е�HCl����2��Cl2��������ǿ��H2O2���ܽ�H2O2��������O2����ѧ����ʽCl2+H2O2=2HCl+O2��3����������Cl2û�з�����Ӧ�ų�����NaOH��Һ���ն����Cl2��4����Ũ����ע��Բ����ƿ���������������������˫��ˮ��Ӧ���ɴ�����������D��װ���п����Ž��ֹˮ�йرգ�Dװ����ѹǿ����ʹҺ���½�������©����Һ�����������������ķ�����ʹ�����ǵ�ľ����ȼ��5��2H2O2=2H2O+O2����Cl2+H2O=HCl+HClO��2HClO=2HCl+O2����
���㣺������ʵ�����Ʒ���������ǿ���ıȽϣ������ļ��鷽��

���Ȼ�����S2Cl2���ڹ�ҵ����������Ϊ��ʵ���Һϳ�S2Cl2��ij��ѧ�о���ѧϰС��������й����ϣ��õ�������Ϣ��
�ٽ������������110�桫140������Ӧ�����ɵ�S2Cl2��Ʒ��
���й����ʵIJ����������±���
| ���� | �۵�/�� | �е�/�� | ��ѧ���� | ||
| S | 112.8 | 444.6 | �� | ||
| S2Cl2 | ��77 | 137 |
|
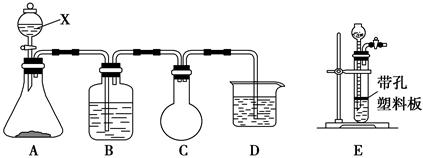
���ʵ��װ��ͼ���£�

��1����ͼ�����巢����β������װ�ò������ƣ���������Ľ���� �����øĽ������ȷװ�ý���ʵ�飬��ش��������⣺
��2��B�з�Ӧ�����ӷ���ʽ�� ��E�з�Ӧ�Ļ�ѧ����ʽ�� ��
��3��C�е��Լ��� ��
��4������B�������� ��F�������� ��
��5������ڼ���Eʱ�¶ȹ��ߣ���ʵ������Ӱ���� ��
��6��S2Cl2��Ʒ�п��ܻ��е������ǣ���д���֣� ��Ϊ�����S2Cl2�Ĵ��ȣ��ؼ��IJ����ǿ��ƺ��¶Ⱥ� ��
ѡ���ʵ�װ�á��Լ��ͷ������Ʊ���ѧ��ѧ�еļ��ֳ������塣����д���еĿոӢ١�����ѡ������ţ���
| ʵ�� | ���� | ����װ�� | �������� | ���������ѡ�õ��Լ� |
| ��1�� | | �� | ��ˮ��Һ�Լ��� | |
| ��2�� | | | 1mol�������2molH2��Ӧ | �� |

��Cl2 ��C2H2
��C2H4 ��NH3
���ʯ�� ��ŨH2SO4
����ʵ��������ܴﵽԤ��ʵ��Ŀ�ĵ���
| | ʵ��Ŀ�� | ʵ����� |
| A | ������������������ | �ֱ���뱥��Na2CO3��Һ |
| B | �Ƚ�Fe��Cu�Ľ������ | �ֱ����Ũ���� |
| C | �Ƚ�H2O���Ҵ����ǻ���Ļ����� | �ֱ��������Na |
| D | �Ƚ�I2��H2O��CCl4�е��ܽ�� | ��I2ˮ�м���CCl4���� |
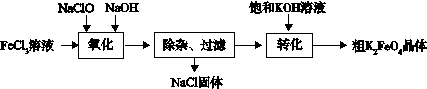
 3Zn(OH)2 + 2Fe(OH)3 + 4KOH
3Zn(OH)2 + 2Fe(OH)3 + 4KOH 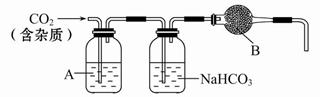
 FeSO4+H2��
FeSO4+H2�� +5C2
+5C2 +16H+
+16H+