��Ŀ����
3�������أ�NiMH��Ŀǰ�Ѿ���Ϊ��϶���������һ����Ҫ������ͣ�NiMH�е�M��ʾ���������Ͻ𣮸õ���ڳ������е��ܷ�Ӧ����ʽ�ǣ�Ni��OH��2+M�TNiOOH+MH ��֪��6NiOOH+NH3+H2O+OH-�T6Ni��OH��2+NO2-
����˵����ȷ���ǣ�������
| A�� | ��طŵ�����У������缫��ӦʽΪ��NiOOH+H2O+e-�TNi��OH��2+OH- | |
| B�� | �������������ĵ缫��Ӧʽ��H2O+M+e-�TMH+OH-��H2O�е�H��M��ԭ | |
| C�� | ��������OH-���Ӵ�����������Ǩ�� | |
| D�� | NiMH����п�����KOH��Һ����ˮ����Ϊ�������Һ |
���� ����������ҪΪKOH�����Һ���ʱ��������Ӧ��Ni��OH��2+OH--e-=NiOOH+H2O��������Ӧ��M+H2O+e-=MH+OH-���ܷ�Ӧ��M+Ni��OH��2=MH+NiOOH��
�ŵ�ʱ��������NiOOH+H2O+e-=Ni��OH��2+OH-��������MH+OH--e-=M+H2O���ܷ�Ӧ��MH+NiOOH=M+Ni��OH��2������ʽ��MΪ����Ͻ�MHΪ��������ԭ�ӵĴ���Ͻ�
A������������ԭ��Ӧ��
B��H2O�е�H�õ��ӣ����DZ�M��ԭ��
C�����ʱ�������������ƶ���
D������6NiOOH+NH3+H2O+OH-=6Ni��OH��2+NO2-�������ð�ˮ���������Һ��
��� �⣺����������ҪΪKOH�����Һ ���ʱ��������Ӧ��Ni��OH��2+OH-=NiOOH+H2O+e-��������Ӧ��M+H2O+e-=MH+OH-���ܷ�Ӧ��M+Ni��OH��2=MH+NiOOH��
�ŵ�ʱ��������NiOOH+H2O+e-=Ni��OH��2+OH-��������MH+OH-=M+H2O+e-���ܷ�Ӧ��MH+NiOOH=M+Ni��OH��2������ʽ��MΪ����Ͻ�MHΪ��������ԭ�ӵĴ���Ͻ�
A������������ԭ��Ӧ���缫��ӦʽΪ��NiOOH+H2O+e-�TNi��OH��2+OH-����A��ȷ��
B��H2O�е�H�õ��ӣ����DZ�M��ԭ����B����
C�����ʱ�������������ƶ���������Ӧ��Ni��OH��2+OH-=NiOOH+H2O+e-������OH-���Ӵ���������������C����
D������6NiOOH+NH3+H2O+OH-=6Ni��OH��2+NO2-�������ð�ˮ���������Һ����D����
��ѡA��
���� ���⿼����ԭ��غ͵���ԭ����Ϊ��Ƶ���㣬������ѧ���ķ��������Ŀ��飬��ȷ�����Ϣ�ĺ����ǽⱾ��ؼ����ѵ�ĵ缫��Ӧʽ����д����Ŀ�Ѷ��еȣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | KNO3��Һ | B�� | FeCl3��Һ | C�� | Al2��SO4��3��Һ | D�� | FeSO4��Һ |
| A�� | ClO3-��BrO3-��Cl2��Fe3+��I2 | B�� | BrO3-��Cl 2��ClO3-��I2��Fe3+ | ||
| C�� | BrO3-��ClO3-��Cl2��Fe3+��I2 | D�� | BrO3-��ClO3-��Fe3+��Cl2��I2 |
| A�� | �Թܡ������� | B�� | �Թܡ��ձ� | C�� | �Թܡ�ƽ����ƿ | D�� | ������Բ����ƿ |
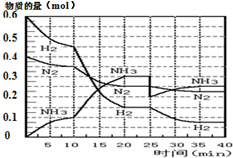 ���ĺϳ�ԭ��Ϊ��N2��g��+3H2��g��?2NH3��g������H=-92.4KJ•mol-1������500�桢20MPaʱ����N2��H2����һ���ݻ�Ϊ2L���ܱ������з�����Ӧ����Ӧ�����и����ʵ����ʵ����仯��ͼ��
���ĺϳ�ԭ��Ϊ��N2��g��+3H2��g��?2NH3��g������H=-92.4KJ•mol-1������500�桢20MPaʱ����N2��H2����һ���ݻ�Ϊ2L���ܱ������з�����Ӧ����Ӧ�����и����ʵ����ʵ����仯��ͼ���ش��������⣺
��1��10min����NH3��ʾ��ƽ����Ӧ����0.005mol/��L��min����
��2����10��20min�ڣ�NH3Ũ�ȱ仯��ԭ�������A��
A�����˴��� B����С�������
C�������¶� D������NH3���ʵ���
��3����1��ƽ���ʱ�䷶ΧΪ��20-25min����1��ƽ�⣺ƽ�ⳣ��K1=$\frac{��\frac{0.3mol}{2L}��^{2}}{��\frac{0.25mol}{2L}����{\frac{0.15mol}{2L}��}^{3}}$�������ݵı���ʽ����
��4���ڷ�Ӧ������25minʱ��
�����߷����仯��ԭ�����0.1molNH3
�ڴ�ڶ���ƽ��ʱ����ƽ���ƽ�ⳣ��K2���� K1������ڡ��������ڡ�����С�ڡ�����
��5���������¡��˹��̵������о��������ڳ��¡���ѹ�����������£�N2�ڴ�������������Fe2O3��TiO2��������ˮ�������з�Ӧ��
N2��g��+3H2O��1��?2NH3��g��+$\frac{3}{2}$O2��g������H=a kJ•mol-1
��һ���о�NH3���������¶ȵĹ�ϵ����ѹ�´ﵽƽ��ʱ��ò���ʵ���������±���
| T/K | 303 | 313 | 323 |
| NH3������/��10-6mol�� | 4.8 | 5.9 | 6.0 |
����֪��N2��g��+3H2��g��?2NH3��g����H=-92.4kJ•mol-1
2H2��g��+O2��g��=2H2O��l��=-571.6kJ•mol-1
�����µ�����ˮ��Ӧ���ɰ������������Ȼ�ѧ����ʽΪ��2N2��g��+6H2O��l��=4NH3��g��+3O2��g����H=+1536kJ•mol-1��
| A�� | һ����������ӵ�����ΪM/NA | |
| B�� | һ����������ӵ����ΪVm/NA | |
| C�� | ����Ħ������Ĵ�Сȡ����������ӱ����Ĵ�С | |
| D�� | ����˵��������ȷ |
 ������һ����Ҫ�Ļ���ԭ�ϣ���ҵ������������̼��Ӧ�õ����ƣ���Ӧ����ʽ���£�
������һ����Ҫ�Ļ���ԭ�ϣ���ҵ������������̼��Ӧ�õ����ƣ���Ӧ����ʽ���£� H2S��
H2S�� Na2O2��
Na2O2�� ��
��