��Ŀ����
���к�CaO���ʵ�CaC2�������������ʵ�飬�ⶨCaC2�����Ĵ��ȣ�����Ӧ����ʽΪ��CaC2+2H2O��Ca��OH��2+C2H2�����������ͼ��ѡ���ʵ���װ�ã���ɸ�ʵ�飮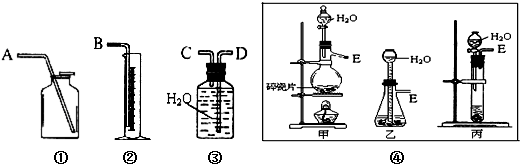
��1����ȡC2H2���ѡ���е� ����ѡ��װ�õ�����˳���ǣ�����ӿ�A��E��˳�� ��
��2����ʵ��ʱ��ȡ������Ϊ1.4g����������Ȳ�ڱ�״���µ����Ϊ448mL����������CaC2����������Ϊ ��������3λ��Ч���֣�

��1����ȡC2H2���ѡ���е�
��2����ʵ��ʱ��ȡ������Ϊ1.4g����������Ȳ�ڱ�״���µ����Ϊ448mL����������CaC2����������Ϊ
���㣺̽�����ʵ���ɻ�������ʵĺ���
ר�⣺ʵ��̽�������ݴ�����
��������1������ʵ������ȡ��Ȳ�ķ�Ӧԭ�Ϻͷ�Ӧ����ѡ��ʵ��װ�ã�����+Һ������壬��Ȳ����������ˮ��������ˮ���������ⶨ���������
��2�����ݷ�Ӧ����ʽ���㣬�������Ϊ���ʵ�������ϻ�ѧ����ʽ���㣬�õ���Ȳ�������ɽ�������Ĵ��ȣ�
��2�����ݷ�Ӧ����ʽ���㣬�������Ϊ���ʵ�������ϻ�ѧ����ʽ���㣬�õ���Ȳ�������ɽ�������Ĵ��ȣ�
���
�⣺��1��̼���ƺ�ˮ��Ӧʵ��Ϊ̼���Ƶ�ˮ�ⷴӦ�������������ƺ���Ȳ����ӦΪ��CaC2+2H2O��Ca��OH��2+C2H2�����ǹ���+Һ������壬��Ӧ�����ȣ�����Ӧ������Ҫ���Ʒ�Ӧ�����ʣ����ǹ����Һ������������װ�ã���װ�ò��ܿ��Ʒ�Ӧ���У���װ���ǹ����Һ�岻�����������װ�ã���Һ©�����Կ��Ʒ�Ӧ���е����ʣ�����ѡ��װ�ã���Ȳ��������ˮ��������ˮ�������ⶨ���������װ�õ�����˳����E��C��D��B��
�ʴ�Ϊ������E��C��D��B��
��2����ʵ��ʱ��ȡ������Ϊ1.4g����������Ȳ�ڱ�״���µ����Ϊ448mL�����ʵ���Ϊ0.02mol��
CaC2+2H2O��Ca��OH��2+C2H2��
64g 1mol
m 0.02mol
���m=1.28g
���Դ���Ϊ��
��100%=91.4%��
�ʴ�Ϊ��91.4%��
�ʴ�Ϊ������E��C��D��B��
��2����ʵ��ʱ��ȡ������Ϊ1.4g����������Ȳ�ڱ�״���µ����Ϊ448mL�����ʵ���Ϊ0.02mol��
CaC2+2H2O��Ca��OH��2+C2H2��
64g 1mol
m 0.02mol
���m=1.28g
���Դ���Ϊ��
| 1.28g |
| 1.40g |
�ʴ�Ϊ��91.4%��
���������⿼����Ȳ���Ʊ���������ƣ�����ѧ����ʵ�������������Ŀ��һ�����Ѷȣ�����ʱע�����ʵ���������֪ʶ��

��ϰ��ϵ�д�
�����Ŀ
�������Ż�����������������𣨡�����
| A��ˮ | B����ĭ����� |
| C�������ɳ�� | D���ɱ������ |
�������ձ��зֱ�ʢ�к�ˮ�����β��� ��1����Ƭ����2������������ͭ����Ƭ����3������������п����Ƭ��������ʴ���ٶ��ɿ쵽����˳���ǣ�������
| A����2����1����3�� |
| B����2����3����1�� |
| C����1����2����3�� |
| D����3����2����1�� |
�����й���Һ���������ʵ���Ũ�ȹ�ϵ��ȷ���ǣ�������
| A��ij�������ʽ��NaHA��Һ��һ���У�c��OH-��+2c��A2-��=c��H+��+c��H2A�� |
| B��0.1mol?L-1CH3COONa��Һ��0.05mol?L-1����������Ϻ��������Һ�У�c��CH3COO-����c��CH3COOH����c��Cl-����c��H+�� |
| C�����ʵ���Ũ����ȵ�HCN��Һ��NaCN��Һ�������Ϻ����Һ�У�c��CN-��+2c��OH-��=2c��H+��+c��HCN�� |
| D��0.1mol?L-1��NH4��2Fe��SO4��2��Һ�У�c��NH4+��+c��NH3?H2O��+c��Fe2+��=0.3mol?L-1 |
�������ӷ���ʽ��д��ȷ���ǣ�������
| A����Ư�۵���Һ��ͨ�����������������壺Ca2++2ClO-+SO2+H2O=CaSO3+��2HClO | ||||
B���ö��Ե缫��ⱥ��ʳ��ˮCl-+H2O
| ||||
| C���Ȼ�������Һ�е�������˫��ˮ2Fe2++2H++H2O2=2Fe3++2H2O | ||||
| D�������ʵ���Ũ�ȡ��������Ca��HCO3��2��Һ������������Һ���ϣ�Ca2++2HCO3-=CaCO3��+CO32-+2H2O |
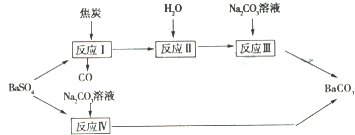



 ���Ͱ�˾ƥ�������ƵĽṹ������һ���Ʊ���������
���Ͱ�˾ƥ�������ƵĽṹ������һ���Ʊ���������
 ���������������ڼ��������·�Ӧ�Ļ�ѧ����ʽΪ
���������������ڼ��������·�Ӧ�Ļ�ѧ����ʽΪ