��Ŀ����
19���廯��ͭ��һ�ְ�ɫ��ĩ����������ˮ������ˮ�л���ⶼ��ֽ⣬�ڿ����л�������������ɫ��ĩ���Ʊ�CuBr��ʵ�鲽�����£�����1������ͼ��ʾ��������ƿ�м���45gCuSO4•5H2O��19gNaBr��150mL��й�������ˮ��60��ʱ���Ͻ��裬���ʵ�����ͨ��SO22Сʱ��
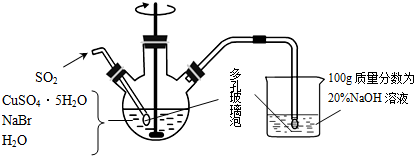
����2����Һ��ȴ����ȥ�ϲ���Һ���ڱܹ�������¹��ˣ�
����3����������������SO2��ˮ����������SO2���Ҵ���������ϴ�ӣ�
����4����˫����������ֱ�װ��Ũ������������ƣ��и���3��4h���پ������������������ո��
��1��ʵ����������ˮ�辭��У����Ŀ���dz�ȥ����ˮ�е�O2��д��ѧʽ����
��2������1�У���������ƿ�з�Ӧ����CuBr�����ӷ���ʽΪ2Cu2++2Br-+SO2+2H2O=2CuBr��+SO42-+4H+��
�ڿ��Ʒ�Ӧ��60����У�ʵ���пɲ�ȡ�Ĵ�ʩ��60��ˮԡ���ȣ�
��3������2������Ҫ�ܹ��ԭ���Ƿ�ֹCuBr����ֽ⣮
��4������3��ϴ�Ӽ��衰����SO2����ԭ���Ƿ�ֹCuBr������������ܼ��������ѵ�Ŀ���dz�ȥ�����Ҵ�����ʹ������ٸ��
���� ��1���廯��ͭ�ܱ���������������Ҫ�ų������ĸ��ţ�
��2����������ƿ��ͭ���ӱ���������ԭ����ͭ���ӣ��������ӷ�Ӧ����CuBr������
�ڿ��Ʒ�Ӧ��60����У�������60���ˮԡ���ȣ�
��3���廯��ͭ�����ֽ⣻
��4���廯��ͭ�ڿ����л�������������ϴ�Ӽ��衰����SO2�����Է�ֹCuBr���������ܼ��������ѿ��Գ�ȥ�����Ҵ�����ʹ������ٸ��
��� �⣺��1���廯��ͭ�ܱ����������������ö�������ԭͭ���������廯��ͭҪ�ų������ĸ��ţ�����ͨ����еķ�����ȥ����ˮ�е�O2��
�ʴ�Ϊ��O2��
��2����������ƿ��ͭ���ӱ���������ԭ����ͭ���ӣ��������ӷ�Ӧ����CuBr��������Ӧ�����ӷ���ʽΪ2Cu2++2Br-+SO2+2H2O=2CuBr��+SO42-+4H+��
�ʴ�Ϊ��2Cu2++2Br-+SO2+2H2O=2CuBr��+SO42-+4H+��
�ڿ��Ʒ�Ӧ��60����У�������60���ˮԡ���ȣ�
�ʴ�Ϊ��60��ˮԡ���ȣ�
��3���廯��ͭ�����ֽ⣬���Բ���2������Ҫ�ܹ⣬��ֹCuBr����ֽ⣬
�ʴ�Ϊ����ֹCuBr����ֽ⣻
��4���ڿ����л�����������������ϴ�Ӽ��衰����SO2�����Է�ֹCuBr������������ܼ��������ѿ��Գ�ȥ�����Ҵ�����ʹ������ٸ��
�ʴ�Ϊ����ֹCuBr����������ȥ�����Ҵ�����ʹ������ٸ��
���� ���⿼����CuCl�������������̣�Ϊ�߿��ȵ���ѵ㣬������ʵķ��롢���ʵ��Ʊ�����ȷ���ʵ������ǽ���ؼ���ע���������ʵ�����Ӧ�ü�������Ϣ��������ã���Ŀ�Ѷ��еȣ�

 ��У����ϵ�д�
��У����ϵ�д� ����ͼ��ʾװ�ü�����ϩʱ����Ҫ���ӵ��ǣ�������
����ͼ��ʾװ�ü�����ϩʱ����Ҫ���ӵ��ǣ�������| ��ϩ���Ʊ� | �Լ�X | �Լ�Y | |
| A | CH3CH2Br��NaOH�Ҵ���Һ���� | ˮ | KMnO4������Һ |
| B | CH3CH2Br��NaOH�Ҵ���Һ���� | ˮ | Br2��CCl4��Һ |
| C | C2H5OH��ŨH2SO4������170�� | NaOH��Һ | KMnO4������Һ |
| D | C2H5OH��ŨH2SO4������170�� | NaOH��Һ | Br2��CCl4��Һ |
| A�� | A | B�� | B | C�� | C | D�� | D |

| A�� | ��ع���ʱ��Li+ͨ�����ӵ�������b�� | |
| B�� | ������X��ͨ����·����Y�� | |
| C�� | ���������ķ�ӦΪ2H++2e-�TH2�� | |
| D�� | Y��ÿ����1molCl2��a���õ�2molLiCl |

| A�� | ��a�Թ����ȼ���Ũ���ᣬȻ���ҡ���Թܱ����������Ҵ����ټӱ����� | |
| B�� | �Թ�b�е������¶˹ܿڲ��ܽ���Һ���ԭ���Ƿ�ֹʵ������з����������� | |
| C�� | ʵ��ʱ�����Թ�a��Ŀ���Ǽ�ʱ�����������������ӿ췴Ӧ���� | |
| D�� | �Թ�b��Na2CO3�������dz�ȥ��������������������������Ҵ�������������������Һ�е��ܽ�� |
��̫������ֽ�ˮ���⣺2H2O��l��=2H2��g��+O2��g����H1=+571.6kJ•mol-1
�ڼ�����ˮ��Ӧ���⣺CH4��g��+H2O��g��=CO��g��+3H2��g����H2=+206.1kJ•mol-1
�۽�̿��ˮ��Ӧ���⣺C��s��+H2O��g��=CO��g��+H2��g����H3=+131.3kJ•mol-1 ��
| A�� | ��Ӧ���е���ת��Ϊ��ѧ�� | |
| B�� | ��Ӧ��ʹ�ô�������H2��С | |
| C�� | ��Ӧ���з�Ӧ�������������������������� | |
| D�� | ��ӦCH4��g��=C��s��+2H2��g���ġ�H=+74.8kJ•mol-1 |
| A�� | ${\;}_{48}^{112}$Cdԭ�ӵ�������Ϊ64 | |
| B�� | ���Ȼ����ܽ�������Ϊ����ǿ������ | |
| C�� | ���Ȼ����ǻ���� | |
| D�� | �ӵĵ����е����Ժ���չ�� |