��Ŀ����
8��ͭ���ء��������Ԫ�صĻ�����������������̫���ܵ�ص���Ҫ���ϣ���ش���1����̬ͭԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p63d104s1��[Ar]3d104s1����֪������CuO��Cu2O+O2����ͭԭ�Ӽ۲���ӽṹ��3d��4s�����Ӧ���ĵ��������仯�Ƕ�������������Cu2O��ԭ����CuO��ͭ�ļ۲�����Ų�Ϊ3d 94s0��Cu2O��ͭ�ļ۲�����Ų�Ϊ3d10�����ߴ����ȶ���ȫ����״̬��ǰ�߲��ǣ�
��2���������������Ԫ���γ���̬�⻯�����Si-H���й��õ��Ӷ�ƫ����Ԫ�أ�����������Ӧʱ�����������������������ĵ縺����Դ�СΪSe��Si���������������������Siͬ���ڲ���Ԫ�صĵ�������ͼ1��ʾ������a��b��c�ֱ����B��
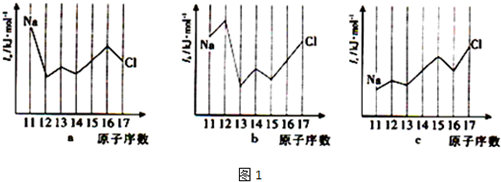
A��aΪIl��bΪI2��cΪI3 B��aΪI2��bΪI3��cΪI1
C��aΪI3��bΪI2��cΪI1 D��aΪIl��bΪI3��cΪI2
��3��SeO2�����°�ɫ���壬�۵�Ϊ340��350�棬315��ʱ��������SeO2����ľ�������Ϊ���Ӿ��壻��SeO2������SO2��V�ͷ��ӣ���Seԭ����������ӻ�����Ϊsp2��
��4������ij�л����γɵ�����������ͼ2����ͼ�ϻ��������е���λ����
��5�����ɰ��SiC����Ӳ��Ϊ9.5���侧���ṹ��ͼ3��ʾ����Siԭ�ӵ���λ��Ϊ4��ÿ��Cԭ����Χ�����Cԭ����ĿΪ12�����������ı߳�Ϊa pm������ɰ���ܶȱ���ʽΪ$\frac{4��40}{��a��10{\;}^{-10}��{\;}^{3}��6.02��10{\;}^{23}}$g/cm3��
���� ��1��CuΪ29��Ԫ�أ�ԭ�Ӻ�����29�����ӣ�����������ԭ����д��������Ų�ʽ��Cu+�ĺ�����28�����ӣ����ݹ���ԭ����д���̬���Ӻ�������Ų�ʽ��ԭ�ӹ������ȫ�ա�������ȫ��ʱ���ȶ���
��2������Si-H���м��ϵ���ƫ����ԭ�ӣ�˵���������ۣ�����������Ӧʱ�������������������Ը��ۣ��ڵ�������Ԫ���У���ʧȥ1�����Ӻ��Ѿ��ﵽ�ȶ��ṹ�������Ƶĵڶ����������þ�����Ϊ2�����ӣ�ʧȥ2�����Ӻ�Ϊ�ȶ��ṹ������þ�ĵڶ������ܽ�С�����������3�����ӣ�ʧȥ2�����Ӻ�δ���ȶ��ṹ�������Ľ����Ա�þ�������Եڶ������ܱ�þ�Ըߣ����������2p����2�����ӣ�ʧȥ������2s�������2�����ӣ���Խ��ȶ������Թ�ĵڶ������ܱ���Ҫ�ͣ��ס���ǽ����������ڶ�������Ҳ����������ʧȥһ�����Ӻ�3p�������3�����ӣ��ǽ��ȶ��ṹ��������ĵڶ�������Ҫ�����ȣ�
��3�����ݷ��Ӿ�����ۡ��е�ͣ����ݼ۲���ӶԻ�������ȷ���ӻ����ͣ�
��4������ij�л����γɵ��������������ɵIJ�������ԭ�Ӻ͵�ԭ�Ӽ��γ���λ�����ɵ�ԭ�ӵ������ṩ���Ӷԣ�����ԭ�Ӽ��γ��ĸ���λ����
��5��ÿ��̼ԭ������4����ԭ�ӣ�ÿ����ԭ������������3��̼ԭ�ӣ��ݴ��ж�ÿ��Cԭ����Χ�����Cԭ����Ŀ���þ�����Cԭ�Ӹ���=8��$\frac{1}{8}$+6��$\frac{1}{2}$=4��Siԭ�Ӹ���Ϊ4�����ݦ�=$\frac{m}{V}$�������ܶȣ�
��� �⣺��1��CuԪ��Ϊ29��Ԫ�أ�ԭ�Ӻ�����29�����ӣ����Ժ�������Ų�ʽΪ��1s22s22p63s23p63d104s1��[Ar]3d104s1��CuO��ͭ�ļ۲�����Ų�Ϊ3d94s0��Cu2O��ͭ�ļ۲�����Ų�Ϊ3d10��3d10Ϊ�ȶ��ṹ�������ڸ���ʱ��������Cu2O��
�ʴ�Ϊ��1s22s22p63s23p63d104s1��[Ar]3d104s1��CuO��ͭ�ļ۲�����Ų�Ϊ3d 94s0��Cu2O��ͭ�ļ۲�����Ų�Ϊ3d10�����ߴ����ȶ���ȫ����״̬��ǰ�߲��ǣ�
��2������Si-H���м��ϵ���ƫ����ԭ�ӣ�˵���������ۣ�����������Ӧʱ�������������������Ը��ۣ����������ĵ縺����Դ�СΪSe��Si��
�ڵ�������Ԫ���У���ʧȥ1�����Ӻ��Ѿ��ﵽ�ȶ��ṹ�������Ƶĵڶ����������þ�����Ϊ2�����ӣ�ʧȥ2�����Ӻ�Ϊ�ȶ��ṹ������þ�ĵڶ������ܽ�С�����������3�����ӣ�ʧȥ2�����Ӻ�δ���ȶ��ṹ�������Ľ����Ա�þ�������Եڶ������ܱ�þ�Ըߣ����������2p����2�����ӣ�ʧȥ������2s�������2�����ӣ���Խ��ȶ������Թ�ĵڶ������ܱ���Ҫ�ͣ��ס���ǽ����������ڶ�������Ҳ����������ʧȥһ�����Ӻ�3p�������3�����ӣ��ǽ��ȶ��ṹ��������ĵڶ�������Ҫ�����ȣ�aΪ�ڶ�������ΪI2��bΪ����������ΪI3��cΪ��һ������ΪI1��ѡB���ʴ�Ϊ������B��
��3��SeO2�����°�ɫ���壬�ۡ��е�ͣ�Ϊ���Ӿ��壻�������������м۲���Ӷ�=2+$\frac{1}{2}$��6-2��2��=3��Seԭ�ӵ��ӻ�����Ϊsp2���Һ���һ���µ��Ӷԣ���������V�Σ�
�ʴ�Ϊ�����Ӿ��壻sp2��
��4����ԭ�Ӻ͵�ԭ�Ӽ��γ���λ�����ɵ�ԭ�ӵ������ṩ���Ӷԣ�����ԭ�Ӽ��γ��ĸ���λ������������λ��ͼʾΪ��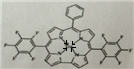
�ʴ�Ϊ�� ��
��
��5��ÿ��̼ԭ������4����ԭ�ӣ�����Siԭ�ӵ���λ��Ϊ4��ÿ����ԭ������������3��̼ԭ�ӣ�����ÿ��̼ԭ����Χ�����̼ԭ����ĿΪ3��4=12���þ�����Cԭ�Ӹ���=8��$\frac{1}{8}$+6��$\frac{1}{2}$=4��Siԭ�Ӹ���Ϊ4�������߳�=a��10-10cm�����V=��a��10-10cm��3����=$\frac{m}{V}$=$\frac{4��40}{��a��10{\;}^{-10}��{\;}^{3}��6.02��10{\;}^{23}}$g•cm3��
�ʴ�Ϊ��4��12��$\frac{4��40}{��a��10{\;}^{-10}��{\;}^{3}��6.02��10{\;}^{23}}$��
���� ���⿼�����ʽṹ�����ʣ��漰�������㡢ԭ���ӻ��жϡ�ԭ�Ӻ�������Ų�ʽ����д��֪ʶ�㣬�ѵ��Ǿ������㣬��Ŀ�Ѷ��еȣ�

| A�� | ���þ�����ϩ������ϩ����ʳƷ��װ�� | |
| B�� | ȥ����������Ag2S���ɽ���������ʢ��ʳ�����������Ƭ�� | |
| C�� | �������ܡ�̫���ܵ�����Դ���ƹ�״����ͣ�ʹ������ϴ�Ӽ������ϻ������� | |
| D�� | ��������ά���͡����ղ�����Ҫ����̼���衢�մɺ�̼��ά���϶��ɣ���һ���������ǽ������� |
| A�� | ���������ˮ����ļ��ԣ�Y��Z | B�� | ����̬�⻯������ȶ��ԣ�X��W | ||
| C�� | ����̬�⻯��ķе㣺X��W | D�� | �����ӵĻ�ԭ�ԣ�X��W |
| A�� | �Ȼ�ͭ��Һ�����۷�Ӧ��Cu2++Fe�TFe2++Cu | |
| B�� | ϡH2SO4�����۷�Ӧ��2Fe+6H+�T2Fe3++3H2�� | |
| C�� | ����������Һ��ϡH2SO4��Ӧ��Ba2++SO42-�TBaSO4�� | |
| D�� | ���Ȼ�����Һ��������ˮ��ӦFe3++3OH-�TFe��OH��3�� |
| A�� | ������KMnO4��Һ�ܼ���CH3CH�TCHCH2OH��CH3CH2CH2CHO | |
| B�� | ����������ˮ�⣬�������Ʊ����� | |
| C�� | ���ݺ˴Ź������ײ��ܼ���1-������2-����� | |
| D�� | ʳ�û����ͺͼ����嶼�ܷ���ˮ�ⷴӦ |
| A�� | 2NO2��g��?N2O4��g�� | B�� | 2HI��g��?H2��g��+I2��g�� | ||
| C�� | PCl5?PCl3��g��+Cl2��g�� | D�� | CaCO3��s��?CaO��g��+CO2 ��g�� |

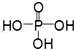 ��
��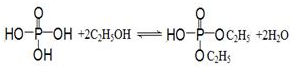 ��
�� ��
��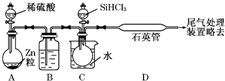 �������һ����Ҫ�ķǽ������ϣ��Ʊ��������Ҫ�������£�
�������һ����Ҫ�ķǽ������ϣ��Ʊ��������Ҫ�������£�