��Ŀ����
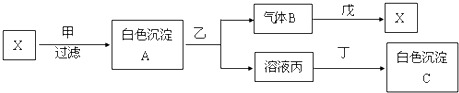
19��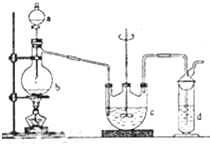 ��������ƣ�Na2S2O3���������մ����������Լ�����������ˮ�������ھƾ������ȡ������ֽ⣮��ҵ�Ͽ�����Ʊ�����Ӧԭ����
��������ƣ�Na2S2O3���������մ����������Լ�����������ˮ�������ھƾ������ȡ������ֽ⣮��ҵ�Ͽ�����Ʊ�����Ӧԭ����2Na2S+Na2CO3+4SO2�T3Na2S2O3+CO2��ʵ����ģ��ù�ҵ���̵�װ����ͼ��ʾ���ش��������⣺
������Һ©��a��ʹ�����������£��ʵ����������У�ʹ��Ӧ������SO2����Ͼ��ȵ�ͨ��Na2S��Na2CO3�Ļ����Һ�У�ͬʱ������Ž�����������
��1��bװ�õ�������������ƿ��b�з�Ӧ�����ӷ���ʽΪSO32-+2H+=SO2��+H2O��
��2����Ӧ��ʼ��c�����л��Dz��������ֱ�Ϊ���壬�˻�������S��
��3������b�е���Һ��pH�ӽ�7��ֹͣͨ��SO2����δ���ƺ�pH��7����Ӱ����ʣ�ԭ���ǣ������ӷ���ʽ��ʾ��S2O32-+2H+=S��+H2O+SO2����
��4��ֹͣͨ��SO2��c�е���Һ����d�У�d�е��Լ�ΪNaOH��Һ��
��5����d����Һ��ת�Ƶ��������У�ˮԡ����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӣ�ϴ�Ӿ������õ��Լ�Ϊ���ѧʽ��C2H5OH��
��6��Ϊ�����ƵõIJ�Ʒ�Ĵ��ȣ���ʵ��С���ȡ5.0�˵IJ�Ʒ���Ƴ�250mL�����������Һ�����ü�ӵ������궨����Һ��Ũ�ȣ�������ƿ�м���25mL 0.01mol/L KIO3��Һ�������������KI�ữ���������з�Ӧ��5I-+IO3-+6H+=3I2+3H2O���ټ��뼸�ε�����Һ������������Na2S2O3��Һ�ζ���������Ӧ��I2+2S2O32-=2I-+S4O62-�����ﵽ�ζ��յ�ʱ������Na2S2O3��Һ20.00mL����ò�Ʒ�Ĵ�����59.3%�����ðٷ�����ʾ������1λС����
���� ��һ��װ��Ϊ�����������ȡװ�ã���ȡ���������ԭ��Ϊ���������ƺ�70%��Ũ�����Ӧ�����ӷ���ʽΪ��SO32-+2H+=SO2��+H2O��cװ��ΪNa2S2O3������װ�ã�2Na2S+Na2CO3+4SO2�T3Na2S2O3+CO2��dװ��Ϊβ������װ�ã����ն��������������������壻��Ӧ��ʼʱ�����ķ�ӦΪ��Na2S+4SO2+H2O=2H2S+Na2SO3��SO2+2H2S=3S��+2H2O��
��1��װ���е�����b��������ƿ��b�з�Ӧ���Ʊ�������������ķ�Ӧ��װ��bΪ�����������ȡ����ȡ���������ԭ��Ϊ���������ƺ�70%��Ũ���cװ��ΪNa2S2O3������װ�ã���2����Ӧ��ʼ��c�����л��Dz��������ֱ���壬����SO2���������ԣ��������Ʒ���������ԭ��Ӧ���ɵ���S���Ӷ�ʹ��Һ����ǣ���˷�Ӧ��ʼ�����Ļ�������S��
��3�������������������Һ�лᷢ��������ԭ��Ӧ����S��SO2��ˮ��
��4���������������������ˮ�������ھƾ������ȡ������ֽ⣬����d�е���ҺӦ���Լ��ԣ�
��5���������������������ˮ�������ھƾ�������ϴ�Ӿ������õ��Լ�Ϊ�ƾ���
��6�����ݷ�Ӧʽ5I-+IO3-+6H+=3I2+3H2O��I2+2S2O32-=2I-+S4O62-������ϵʽ��KIO3��6Na2S2O3�����ݵ�������ӵ����ʵ������Na2S2O3�����ʵ���Ũ�ȣ����250mL��Һ��Na2S2O3�������������������5g��ȵô��ȣ�
��� �⣺��1��װ����b�����Ǵ�֧�ܵ���ƿΪ������ƿ������b��������������ƿ����һ��װ��Ϊ�����������ȡװ�ã���ȡ���������ԭ��Ϊ���������ƺ�70%��Ũ���ᣬ��Ӧ�����ӷ���ʽΪ��SO32-+2H+=SO2��+H2O��
�ʴ�Ϊ��������ƿ��SO32-+2H+=SO2��+H2O��
��2����Ӧ��ʼʱ�����ķ�ӦΪ��Na2S+SO2+H2O=H2S+Na2SO3��SO2+2H2S=3S��+2H2O���ʸû�������S��
�ʴ�Ϊ��S��
��3�����������������������Һ�лᷢ��������ԭ��Ӧ����S��SO2��ˮ�����ӷ���ʽΪ��S2O32-+2H+=S��+H2O+SO2����������δ���ƺã�pH��7ʱ��Ӱ����ʣ�
�ʴ�Ϊ��S2O32-+2H+=S��+H2O+SO2����
��4���������������������ˮ�������ھƾ������ȡ������ֽ⣬ֹͣͨ��SO2��c�е���Һ����d�У�d�е��Լ�Ӧ���Լ��ԣ�Ϊ����������Һ��
�ʴ�Ϊ��NaOH��Һ��
��5���������������������ˮ�������ھƾ�������ϴ�Ӿ������õ��Լ�Ϊ�ƾ�����ѧʽΪC2H5OH��
�ʴ�Ϊ��C2H5OH��
��6������ƿ�м���25mL 0.0lmol•L-1KIO3��Һ�������������KI���ữ���������з�Ӧ��5I-+IO3-+6H+�T3I2+3H2O���ټ��뼸�ε�����Һ������������Na2S2O3��Һ�ζ���������Ӧ��I2+2S2O32-�T2I-+S4O62-��
��ɵù�ϵʽ��IO3-��6S2O32-��
1mol 6mol
0.025L��0.0lmol•L-1 n��S2O32-��
��n��S2O32-��=0.0015mol����ԭ��Һ��Na2S2O3�����ʵ�����0.0015mol��250/20=0.01875mol��������0.01875mol��158g/mol=2.9625g����˸ò�Ʒ�Ĵ�����$\frac{2.9625g}{5.0g}$��100%=59.3%��
�ʴ�Ϊ��59.3%��
���� ���⿼���������Ʊ�ʵ�鷽������ƺ��Ʊ����̷���Ӧ�ã���ȷʵ���������Ƽ�������ʵ������ǽ����Ĺؼ�����Ŀ�Ѷ��еȣ������ֿ�����ѧ���ķ������������������Ӧ����ѧ֪ʶ��������

 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д�| A�� | ���м���ʯ���ƵÿɿڵĶ��� | |
| B�� | ��������ʳ��ˮ�����Ȼ��ƾ��� | |
| C�� | ���ˮ����μ��뱥�͵�FeCl3��Һ���õ����ɫ���ķ�ɢϵ | |
| D�� | ���������ܽ���������Һ�������Ⱦ����ж�������� |
| A�� | ��ϵͳ���������л��� 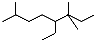 ������Ϊ3��3��7-����-4-�һ����� ������Ϊ3��3��7-����-4-�һ����� | |
| B�� | ʯ������ϩ��ú��Һ���������ʵ�����������ȥ���۾��漰��ѧ�仯 | |
| C�� | ���������Ҵ�����ϩ��ȫȼ�գ�ǰ�����ɸ����ˮ | |
| D�� | ȫ�������� 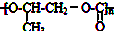 ���ɻ������飨 ���ɻ������飨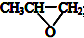 ����CO2��Ӧ�Ƶ� ����CO2��Ӧ�Ƶ� |
| A�� | x��y | B�� | x��y | C�� | x��y | D�� | ���ж� |
| A�� | �����ʵ�ˮ������ڷ�����ᴿ������ | |
| B�� | ��ϩ�������ͨ�����۷�Ӧ���ɸ߷��ӻ����� | |
| C�� | �л���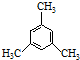 �ĺ˴Ź��������������壬�ҷ����֮��Ϊ3��1 �ĺ˴Ź��������������壬�ҷ����֮��Ϊ3��1 | |
| D�� | ���Ҵ���Ũ�����Ʊ���ϩʱ������ˮԡ���ȿ��Ʒ�Ӧ���¶� |
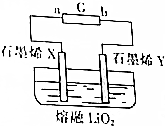 ��Ӣ��������ʱ������վ���������Ӣ����ѧ�����Ƴ�һ�ֳ�������﮵�أ������ع���ԭ����ͼ��ʾ��X��Y����ʯīϩ�Ƴɵ����Լ��õġ����ɡ��缫������ʯīϩX������ﮣ��õ�صĹ���ԭ����Li��s��+O2��g��$?_{���}^{�ŵ�}$LiO2��s��������˵������ȷ���ǣ�������
��Ӣ��������ʱ������վ���������Ӣ����ѧ�����Ƴ�һ�ֳ�������﮵�أ������ع���ԭ����ͼ��ʾ��X��Y����ʯīϩ�Ƴɵ����Լ��õġ����ɡ��缫������ʯīϩX������ﮣ��õ�صĹ���ԭ����Li��s��+O2��g��$?_{���}^{�ŵ�}$LiO2��s��������˵������ȷ���ǣ�������| A�� | �����̣���ԴG��b��Ϊ���� | |
| B�� | �ŵ���̣�Y��������ԭ��Ӧ | |
| C�� | �ŵ���̣�X���ĵ缫��ӦʽΪLi-e-+O2-�TLiO2 | |
| D�� | �����̣�ÿת��2 mol���ӣ�Y���ų�44.8L����״����O2 |
 ��
��