��Ŀ����
 �����ᣨH3PO2����һ�־�ϸ������Ʒ�����н�ǿ�Ļ�ԭ�ԣ��ش��������⣺
�����ᣨH3PO2����һ�־�ϸ������Ʒ�����н�ǿ�Ļ�ԭ�ԣ��ش��������⣺��1��H3PO2��һԪ��ǿ�ᣬд������뷽��ʽ
��2��H3PO2��NaH2PO2���ɽ���Һ�е�Ag+��ԭΪAg���Ӷ������ڻ�ѧ������
��H3PO2�У�PԪ�صĻ��ϼ�Ϊ
������H3PO2���л�ѧ������Ӧ�У��������뻹ԭ�������ʵ���֮��Ϊ4��1������������Ϊ
��NaH2PO2Ϊ
��3��H3PO2�Ĺ�ҵ�Ʒ��ǣ������ף�P4����Ba��OH��2��Һ��Ӧ����PH3�����Ba��H2PO2��2����������H2SO4��Ӧ��д��������Ba��OH��2��Һ��Ӧ�Ļ�ѧ����ʽ
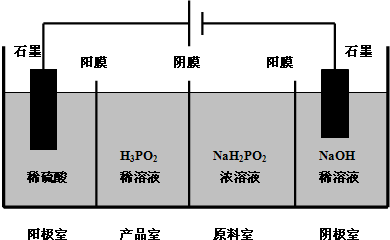
��4��H3PO2Ҳ���õ��������Ʊ��������ҵ�������������ԭ����ͼ��ʾ����Ĥ����Ĥ�ֱ�ֻ���������ӡ�������ͨ������д�������ĵ缫��Ӧ
���㣺���뷽��ʽ����д,ԭ��غ͵��صĹ���ԭ��,���������ˮ��Һ�еĵ���ƽ��
ר�⣺
��������1��H3PO2��һԪ���ᣬ��ˮ��Һ�ﲿ�ֵ������������Ӻ�������ӣ�
��2���ٸ��ݻ������л��ϼ۵Ĵ�����Ϊ0ȷ��PԪ�ػ��ϼۣ�
�ڸ���ת�Ƶ����غ��ж��������
��H3PO2��һԪ���ᣬ��NaH2PO2Ϊ���Σ����������ˮ��Һ��ˮ�����Һ�ʼ��ԣ�
��3�����ݷ�Ӧ�����������ԭ���غ�͵����غ���д����ʽ��
��4������������������Ϊ��������ӡ����������Ӻ�H2PO2-���жϷŵ�����ǿ����Ȼ��д�������ĵ缫��Ӧʽ��
��2���ٸ��ݻ������л��ϼ۵Ĵ�����Ϊ0ȷ��PԪ�ػ��ϼۣ�
�ڸ���ת�Ƶ����غ��ж��������
��H3PO2��һԪ���ᣬ��NaH2PO2Ϊ���Σ����������ˮ��Һ��ˮ�����Һ�ʼ��ԣ�
��3�����ݷ�Ӧ�����������ԭ���غ�͵����غ���д����ʽ��
��4������������������Ϊ��������ӡ����������Ӻ�H2PO2-���жϷŵ�����ǿ����Ȼ��д�������ĵ缫��Ӧʽ��
���
�⣺��1��H3PO2��һԪ��ǿ�ᣬ��ˮ��Һ�ﲿ�ֵ������������Ӻ�������ӣ����뷽��ʽΪH3PO2?H++H2PO2-��
�ʴ�Ϊ��H3PO2?H++H2PO2-��
��2���ٸ��ݻ������л��ϼ۵Ĵ�����Ϊ0ȷ��PԪ�ػ��ϼۣ�OԪ��Ϊ-2�ۡ�HԪ��Ϊ+1�ۣ�����PԪ��Ϊ+1�ۣ��ʴ�Ϊ��+1��
�ڸ÷�Ӧ������������������H3PO2�ǻ�ԭ�����������뻹ԭ�������ʵ���֮��Ϊ4��1������ת�Ƶ������֪��1molH3PO2ʧȥ4mol���ӣ�����PԪ��Ϊ+5�ۣ�������������H3PO4��
�ʴ�Ϊ��H3PO4��
��H3PO2��һԪ���ᣬ��NaH2PO2Ϊ���Σ����������ˮ��Һ��ˮ�����Һ�ʼ��ԣ�
�ʴ�Ϊ�����Σ������ԣ�
��3�����ף�P4����Ba��OH��2��Һ��Ӧ����PH3�����Ba��H2PO2��2����Ӧ����ʽΪ2P4+3Ba��OH��2+6H2O=3Ba��H2PO2��2+2PH3����
�ʴ�Ϊ��2P4+3Ba��OH��2+6H2O=3Ba��H2PO2��2+2PH3����
��4������������������Ϊ��������ӡ����������Ӻ�H2PO2-�����зŵ�������ǿ�������������ӣ������������ĵ缫��ӦΪ��2H2O-4e-=O2��+4H+��
�ʴ�Ϊ��2H2O-4e-=O2��+4H+��
�ʴ�Ϊ��H3PO2?H++H2PO2-��
��2���ٸ��ݻ������л��ϼ۵Ĵ�����Ϊ0ȷ��PԪ�ػ��ϼۣ�OԪ��Ϊ-2�ۡ�HԪ��Ϊ+1�ۣ�����PԪ��Ϊ+1�ۣ��ʴ�Ϊ��+1��
�ڸ÷�Ӧ������������������H3PO2�ǻ�ԭ�����������뻹ԭ�������ʵ���֮��Ϊ4��1������ת�Ƶ������֪��1molH3PO2ʧȥ4mol���ӣ�����PԪ��Ϊ+5�ۣ�������������H3PO4��
�ʴ�Ϊ��H3PO4��
��H3PO2��һԪ���ᣬ��NaH2PO2Ϊ���Σ����������ˮ��Һ��ˮ�����Һ�ʼ��ԣ�
�ʴ�Ϊ�����Σ������ԣ�
��3�����ף�P4����Ba��OH��2��Һ��Ӧ����PH3�����Ba��H2PO2��2����Ӧ����ʽΪ2P4+3Ba��OH��2+6H2O=3Ba��H2PO2��2+2PH3����
�ʴ�Ϊ��2P4+3Ba��OH��2+6H2O=3Ba��H2PO2��2+2PH3����
��4������������������Ϊ��������ӡ����������Ӻ�H2PO2-�����зŵ�������ǿ�������������ӣ������������ĵ缫��ӦΪ��2H2O-4e-=O2��+4H+��
�ʴ�Ϊ��2H2O-4e-=O2��+4H+��
���������⿼�����ε�ˮ��ԭ����������ʵĵ��롢������ԭ��Ӧ������֪ʶ����Ŀ�Ѷ��еȣ������漰��֪ʶ��϶࣬��ֿ�����ѧ������ѧ֪ʶ�����������

��ϰ��ϵ�д�
�����Ŀ
����������Ԫ��A��B��C��D��ԭ��������������A��C��ԭ�������IJ�Ϊ8��A��B��C����Ԫ��ԭ�ӵ�����������֮��Ϊ15��Bԭ����������������Aԭ��������������һ�룮������������ȷ���ǣ�������
| A��ԭ�Ӱ뾶��B��C��D��A |
| B������B����������������ʢ��Ũ���� |
| C��A��C�γɵ������������������� |
| D��Ԫ��D�����ڱ���λ�ڵ������ڡ�VIIA�� |
VmL Al2��SO4��3��Һ�к�Al3+ ag��ȡV/4mL��Һϡ�͵�4VmL����ϡ�ͺ���Һ��SO42-�����ʵ���Ũ���ǣ�������
A��
| ||
B��
| ||
C��
| ||
D��
|
�������������г������ʵ���Ҫ�ɷ��뻯ѧʽ���Ӧ���ǣ�������
| A�����ά-Si |
| B��������-Al��OH��3 |
| C��ˮ��-Na2SiO3 |
| D������-KAl��SO4��2?12H2O |
�����л���Ӧ�ķ���ʽ��д������ǣ�������
A��CH4+Cl2
| |||
B��2CH3CH2OH+O2
| |||
C�� +HNO3 +HNO3
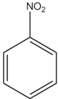 +H2O +H2O | |||
D��nCH2=CH2
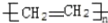 |
ij�����ˮ��Һ�п��ܺ������������е������֣�K+��NH4+��Cl-��Ca2+��CO32-��SO42-����ÿ��ȡ100mL��Һ�ֱ��������ʵ�飺����˵����ȷ���ǣ�������
| ʵ���� | ʵ�鲽�� | ʵ������ |
| �� | ����AgNO3��Һ | �г������� |
| �� | ��������NaOH��Һ����� | �ռ�����0.896L����״���� |
| �� | ����������BaCl2 ��Һ | �õ�����6.27g����������������ϴ�ӡ������ʣ��2.33g |
| A��һ�������ڵ����ӣ�Ca2+��Cl- |
| B��c��K+��=0.2 mol/L |
| C��c��K+��+c��NH4+��=c��CO32-��+c��SO42-�� |
| D��һ�����ڵ����ӣ�K+��NH4+��CO32-��SO42- |
������ʵ��������������ԭ�����͵��ǣ�������
| A����ˮ��������ƽ��Br2+H2O?HBr+HBrO����������������Һ����Һ��ɫ��dz |
| B���ϳɰ���Ӧ��Ϊ��߰��IJ��ʣ�������Ӧ��ȡ�����¶ȵĴ�ʩ |
| C����ӦCO��g��+NO2��g��?CO2��g��+NO��g��������ӦΪ���ȷ�Ӧ������ƽ��������¶���ϵ��ɫ���� |
| D������2NO2��g��?N2O4��g������ƽ�����С���������ʹ��ϵ��ɫ���� |