��Ŀ����
20��Ϊ�ⶨij�л�������A�Ľṹ����������ʵ�飺��һ������ʽ��ȷ��
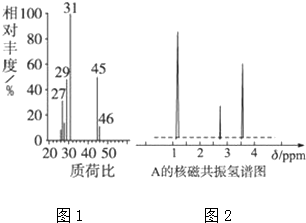
��1�����л���A�����������г��ȼ��ʵ���ã�̼����������Ϊ52.16%�������������Ϊ13��.14%����������и�Ԫ�ص�ԭ�Ӹ�������N��C����N��H����N��O��=2��6��1
��2���������Dzⶨ����Է����������õ���ͼ1��ʾ������ͼ������л���A����Է�������Ϊ46������ʽΪC2H6O
��3�����ݼۼ����ۣ�Ԥ��A�Ŀ��ܽṹ��д���ṹ��ʽCH3OCH3��CH3CH2OH
�������ṹʽ��ȷ��
��4���˴Ź��������ܶ��л�������в�ͬλ�õ���ԭ�Ӹ�����ͬ�ķ�ֵ���źţ������ݷ�ֵ���źţ�����ȷ����������ԭ�ӵ��������Ŀ���ⶨ���л���A�ĺ˴Ź�������ʾ��ͼ��ͼ2��ʾ����A�Ľṹ��ʽΪCH3CH2OH��
���� ��1��A��̼����������Ϊ52.16%�������������Ϊ13.14%������Ԫ����������=1-52.16%-13.14%=34.7%��Ȼ����ԭ�Ӹ���֮�ȣ�
��2�������Dzⶨ�л����������Է�������Ϊ46��������ʽ�жϷ���ʽΪC2H6O��
��3��A����Ϊ�Ҵ�������ѣ�
��4���л���A�����������ֲ�ͬ��ѧ��������ԭ�ӣ�ӦΪ�Ҵ���������ֻ��һ�ֲ�ͬ��ѧ��������ԭ�ӣ�
��� �⣺��1��A��̼����������Ϊ52.16%�������������Ϊ13.14%������Ԫ����������=1-52.16%-13.14%=34.7%��
���л��������N��C����N��H����N��O��=$\frac{52.16%}{12}$��$\frac{13.14%}{1}$��$\frac{34.7%}{16}$=2��6��1���ʴ�Ϊ��N��C����N��H����N��O��=2��6��1��
��2��ʵ��ʽΪC2H6O���л����������Է�������Ϊ46��ʵ��ʽ��Ϊ����ʽ��������ʵķ���ʽ��C2H6O��
�ʴ�Ϊ��46��C2H6O��
��3��A����Ϊ�Ҵ�������ѣ��ṹ��ʽΪCH3CH2OH��CH3OCH3���ʴ�Ϊ��CH3OCH3��CH3CH2OH��
��4���л���A�����������ֲ�ͬ��ѧ��������ԭ�ӣ�ӦΪ�Ҵ�����CH3CH2OH��������ֻ��һ�ֲ�ͬ��ѧ��������ԭ�ӣ��ʴ�Ϊ��CH3CH2OH��
���� ���⿼���л���ṹȷ������Ŀ�Ѷ��еȣ����غ�Ƕ��ж��л������ʽΪ������Ĺؼ���ע����������ͼ���˴Ź������ס�����������л���ṹȷ���е����ã�

 ��ѧ����ͬ����ϰϵ�д�
��ѧ����ͬ����ϰϵ�д� ��ǰ�κ�ͬ����ϰϵ�д�
��ǰ�κ�ͬ����ϰϵ�д� ����С��ҵϵ�д�
����С��ҵϵ�д� �Ƹ�С״Ԫ����������ϰ��ϵ�д�
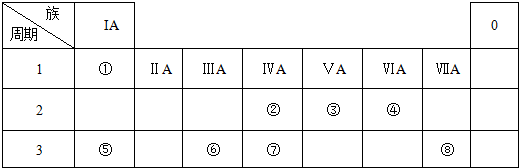
�Ƹ�С״Ԫ����������ϰ��ϵ�д� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | �� | �� | ||||||
| 3 | �� | �� | �� | �� | �� | �� | �� | |
| 4 | �� | ⑪ | ⑫ |
��2������ЩԪ���У�����õĽ���Ԫ�غ�����õķǽ���Ԫ�طֱ���K��F����Ԫ�ط������𣩣�
��3������ЩԪ�ص�����������Ӧˮ�����У���һ�ֳ����Ե���������θ����෴Ӧ�Ļ�ѧ����ʽ��Al��OH��3+3HCl=AlCl3+3H2O��
��4������ЩԪ���У�ijԪ�ص�����������Ӧ��ˮ������Ժ�����̬�⻯�ﷴӦ��д���÷���ʽNH3+HNO3=NH4NO3�� H2S+3H2SO4��Ũ��=4SO2+4H2O��
��5���ڢ�����У���Ƽ�ʵ�飨ֻҪд��ʵ�鷽�����ɣ���֤��������Ե�ǿ���������ֱ�ȡ��С����״��ͬ��Mg��Al��Ũ����ͬ�����ᷴӦ���۲�������ݵ����ʣ��ȽϿ����Mg����Mg������ǿ��
��6�����Ԫ�آ����ķǽ�����ǿ����д��������֤�ý��۵�һ�����ӷ�Ӧ����ʽS2-+Cl2=S��+2Cl-��
| A�� | H2O��g��=H2O��l����H=-44kJ/mol�����Ըù����Ƿ��ȷ�Ӧ | |
| B�� | �������õ���Դ����ͨ����ѧ��Ӧ��õ� | |
| C�� | 2CO��g��+O2$\frac{\underline{\;��ȼ\;}}{\;}$2CO2��g����H��0����56gCO��32gO2�����е����������88g�����е������� | |
| D�� | ��Ҫ���ȵķ�Ӧһ�������ȷ�Ӧ |
| A�� | ԭ�� | B�� | ���� | C�� | ���� | D�� | Ԫ�� |
| A�� | KOH��CaCl2�Ⱥ������Ӽ����ֺ��й��ۼ� | |
| B�� | PH3��ԭ�Ӿ�����8�����ȶ��ṹ | |
| C�� | �����ӵĽṹʾ��ͼ�� | |
| D�� | NH4Cl�ĵ���ʽ��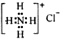 |
 ����ϩ���������Ϻͺϳ�����Ҫ�����л�ԭ�ϣ��ұ������ⷨ��Ŀǰ��������������ϩ����Ҫ�������仯ѧ����ʽΪ��
����ϩ���������Ϻͺϳ�����Ҫ�����л�ԭ�ϣ��ұ������ⷨ��Ŀǰ��������������ϩ����Ҫ�������仯ѧ����ʽΪ��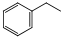 ��g��$?_{���£�T��}^{Fe_{2}O_{3}}$
��g��$?_{���£�T��}^{Fe_{2}O_{3}}$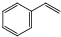 ��g��+H2��g����H=+120kJ•mol-1
��g��+H2��g����H=+120kJ•mol-1
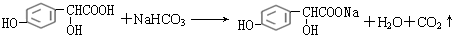 ��
�� ��
��
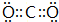 ��
��