��Ŀ����
 ����������д�����з�Ӧ���Ȼ�ѧ����ʽ��
����������д�����з�Ӧ���Ȼ�ѧ����ʽ����1����������N2��O2��ȫ��Ӧ��ÿ����23g NO2��Ҫ����16.95kJ������
���Ȼ�ѧ����ʽΪ
��2����֪��1mol H-H����1mol N-H����1mol N��N���ֱ���Ҫ��������436kJ��391kJ��946kJ����N2��H2��Ӧ����NH3���Ȼ�ѧ����ʽΪ
�ⶨϡ�����ϡ���������к��ȵ�ʵ��װ����ͼ��ʾ��
��1������ĭ���ϵ�������ʲô��
��2��ʵ����ֵ�����57.3kJ?mol-1��ƫ�����ƫ���ԭ���ǣ�����ĸ��˫ѡ��
A��ʵ��װ�ñ��¡�����Ч����
B���ⶨʹ�õ�����ʹ��ǿ��ǿ��
C��һ����NaOH��Һ����ʢ�������С�ձ���
D�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�ӲⶨH2SO4��Һ���¶ȣ�
���㣺�к��ȵIJⶨ
ר�⣺��ѧ��Ӧ�е������仯
��������1��������֪��ÿ����23g NO2��Ҫ����16.95kJ���������Ӧ�ȣ��ٸ����Ȼ�ѧ����ʽ����д����д�����Ȼ�ѧ����ʽ��
��2����ѧ��Ӧ�У���ѧ�����������������γ��»�ѧ���ų����������ݷ���ʽ����ֱ����պͷų����������Դ˼��㷴Ӧ�Ȳ��ж����Ȼ��Ƿ��ȣ�
��1�������к��ȵ�ʵ��Ĺؼ��Ǿ����ܼ�������ɢʧ���н��
��2��A��װ�ñ��¡�����Ч�����õ�����ƫС��
B���ⶨ�к���ʱ����ʹ��ϡ��ǿ���ǿ����Һ��
C�����ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ��У���õ�����ƫС������Ӧ��һ���Ե�������������Һ��
D���¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�Ӳ���ϡH2SO4���¶ȣ��������ʼ�¶�ƫ�ߣ�
��2����ѧ��Ӧ�У���ѧ�����������������γ��»�ѧ���ų����������ݷ���ʽ����ֱ����պͷų����������Դ˼��㷴Ӧ�Ȳ��ж����Ȼ��Ƿ��ȣ�
��1�������к��ȵ�ʵ��Ĺؼ��Ǿ����ܼ�������ɢʧ���н��
��2��A��װ�ñ��¡�����Ч�����õ�����ƫС��
B���ⶨ�к���ʱ����ʹ��ϡ��ǿ���ǿ����Һ��
C�����ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ��У���õ�����ƫС������Ӧ��һ���Ե�������������Һ��
D���¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�Ӳ���ϡH2SO4���¶ȣ��������ʼ�¶�ƫ�ߣ�
���
�⣺��1��������N2��O2��ȫ��Ӧ��ÿ����23��NO2��Ҫ����16.95kJ����������ÿ����92��NO2��Ҫ����67.8kJ���������Ȼ�ѧ����ʽΪN2��g��+2O2��g��=2NO2��g����H=+67.8kJ?mol-1��
�ʴ�Ϊ��N2��g��+2O2��g��=2NO2��g����H=+67.8kJ?mol-1��
��2���ڷ�ӦN2+3H2?2NH3�У�����3molH-H����1molN��N�������յ�����Ϊ3��436kJ+946kJ=2254kJ������2molNH3�����γ�6molN-H�����ų�������Ϊ6��391kJ=2346kJ�����յ������٣��ų��������࣬�÷�ӦΪ���ȷ�Ӧ���ų�������Ϊ2346kJ-2254kJ=92kJ��N2��H2��Ӧ����NH3���Ȼ�ѧ����ʽΪ��N2��g��+3H2��g��?2NH3��g����H=-92kJ?mol-1��
�ʴ�Ϊ��N2��g��+3H2��g��?2NH3��g����H=-92kJ?mol-1��
��1���к��ȵ�ʵ��Ĺؼ��DZ��£���������ĭ���ϵ������DZ��¡����ȣ���ֹ�кͷ�Ӧʱ������ʧ��
�ʴ�Ϊ�����¡����ȣ�����������ʧ��
��2��A��װ�ñ��¡�����Ч�����õ�����ƫС���к��ȵ���ֵƫС����A��ȷ��
B���к��ȵIJⶨ�б���ʹ��ϡ��ǿ���ǿ����Һ������ʹ����������������Ӱ��ⶨ��������Ը���������������Ӱ��ⶨ�������B����
C������һ�ο��ٽ�NaOH��Һ����ʢ�������С�ձ��У��������ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ��У��������������ɢʧ�����Ըò�����������C����
D���¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�Ӳ���ϡH2SO4���¶ȣ������¶ȼ��ϻ����������ƣ��������������ᷴӦ���ȣ������������ʼ�¶�ƫ�ߣ���ô������¶��������ͱ�ʵ��ҪС����D��ȷ��
�ʴ�Ϊ��AD��
�ʴ�Ϊ��N2��g��+2O2��g��=2NO2��g����H=+67.8kJ?mol-1��
��2���ڷ�ӦN2+3H2?2NH3�У�����3molH-H����1molN��N�������յ�����Ϊ3��436kJ+946kJ=2254kJ������2molNH3�����γ�6molN-H�����ų�������Ϊ6��391kJ=2346kJ�����յ������٣��ų��������࣬�÷�ӦΪ���ȷ�Ӧ���ų�������Ϊ2346kJ-2254kJ=92kJ��N2��H2��Ӧ����NH3���Ȼ�ѧ����ʽΪ��N2��g��+3H2��g��?2NH3��g����H=-92kJ?mol-1��
�ʴ�Ϊ��N2��g��+3H2��g��?2NH3��g����H=-92kJ?mol-1��
��1���к��ȵ�ʵ��Ĺؼ��DZ��£���������ĭ���ϵ������DZ��¡����ȣ���ֹ�кͷ�Ӧʱ������ʧ��
�ʴ�Ϊ�����¡����ȣ�����������ʧ��
��2��A��װ�ñ��¡�����Ч�����õ�����ƫС���к��ȵ���ֵƫС����A��ȷ��
B���к��ȵIJⶨ�б���ʹ��ϡ��ǿ���ǿ����Һ������ʹ����������������Ӱ��ⶨ��������Ը���������������Ӱ��ⶨ�������B����
C������һ�ο��ٽ�NaOH��Һ����ʢ�������С�ձ��У��������ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ��У��������������ɢʧ�����Ըò�����������C����
D���¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�Ӳ���ϡH2SO4���¶ȣ������¶ȼ��ϻ����������ƣ��������������ᷴӦ���ȣ������������ʼ�¶�ƫ�ߣ���ô������¶��������ͱ�ʵ��ҪС����D��ȷ��
�ʴ�Ϊ��AD��
���������⿼���Ȼ�ѧ����ʽ�Լ��к��ȵIJⶨ����Ŀ�Ѷȴ�ע�������к��ȵĸ�������Ȼ�ѧ����ʽ����д�����Լ��ⶨ��Ӧ�ȵ��������⣮

��ϰ��ϵ�д�
�����Ŀ
����ʱ����V1mLc1mol?L-1�İ�ˮ�μӵ�V2mLc2mol?L-1�������У���������һ����ȷ���ǣ�������
| A����V1��V2��c1=c2������Һ��pH��7 |
| B���������Һ��pH=7����c1V1��c2V2 |
| C����c1=c2�����Һ��c��NH4+���Tc��Cl-������V1��V2 |
| D���������Һ��pH��7������Һ��c��NH4+����c��OH-����c��Cl-����c��H+�� |
����˵����ȷ���ǣ�������
| A��0.1mol/LCH3COOH��Һ��ˮϡ�ͣ���Һ��c��OH-������ | ||
B����NaOH�Ͱ�ˮ��Һ��ϡ��һ�������ߵ�OH-Ũ�Ⱦ����ٵ�ԭ����
| ||
| C����ͬŨ�ȵ�HCl��CH3COOH������Һ��c��H+����ͬ | ||
| D����HA��HB��Ϊ���ᣬ����HA��HB������ͬ�����£���Һ��pH��СΪNaA��NaB |
�������ӷ���ʽ�У�������ǣ�������
| A������ͨ�������У�NH3+H+=NH4+ |
| B������ͭм�ŷ�ϡ�����У�3Cu+8H++2NO3-=3Cu2++2NO��+4H2O |
| C������SO2ͨ��������ռ���Һ�У�SO2+2OH-=SO32-+H2O |
| D������������ĩ�е��백ˮ��Al��OH��3+OH-=AlO2-+H2O |
��һ�����ļ���ȼ�պ�õ�CO��CO2��ˮ��������������������49.6gͨ����ˮ�Ȼ���ʱ����ˮ�Ȼ�������25.2g����CO2�������ǣ�������
| A��12.5g |
| B��13.2g |
| C��19.7g |
| D��24.4g |

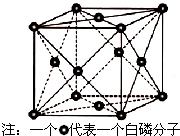 VA��ĵ����ס��飨As����Ԫ�صĻ������ڿ��к���������������Ҫ��;����ش��������⣮
VA��ĵ����ס��飨As����Ԫ�صĻ������ڿ��к���������������Ҫ��;����ش��������⣮ �Ĵ�ʢ���屶�ӣ����屶��Ϊԭ�Ͽ����Ƶû�����A��A�Ľṹ��ʽ��ͼ��ʾ����ش��������⣺
�Ĵ�ʢ���屶�ӣ����屶��Ϊԭ�Ͽ����Ƶû�����A��A�Ľṹ��ʽ��ͼ��ʾ����ش��������⣺