��Ŀ����
18�� �����仯�������������������м�����Ҫ�����ã�
�����仯�������������������м�����Ҫ�����ã���1���ҹ�����ϵ�����ػ�����£�N2H4����ȼ�ϣ�N2H4��NH3�����ƵĻ�ѧ���ʣ�
��д���������ᷴӦ�����ӷ���ʽ��N2H4+2H+=N2H62+��
���ڻ���ƽ�����װ��Һ̬�º�˫��ˮ�������ǻ��ʱѸ�ٷ�Ӧ���ɵ�����ˮ������д����Ӧ�Ļ�ѧ����ʽ��N2H4+2H2O2�TN2+4H2O��
�ۻ������ʱ����Ϊȼ�ϣ�Ҳ������һ������������������Ӧ��������ת��2mol���ӣ�������ȼ���µ�����Ϊ16g
��2������β���еĵ����������γ����ꡢ�������ж�����֮һ��Ϊ�˼�����Ⱦ���ɳ���ʹ������β������װ�ã���ԭ����ͼ��ʾ��д�������������ܷ�Ӧ�Ļ�ѧ����ʽ��2NO+O2+4CO$\frac{\underline{\;����\;}}{\;}$4CO2+N2��
���� ��1�����������ᷴӦ����N2H62+��
�ڸ��������Ϣ�ҳ���Ӧ��������Ӧ���������ݻ�ѧ����ʽ����д���������������д��
�����ݵ�ʧ�����غ���㣺һ������ת��Ϊ�����õ��ĵ�������=��ʧȥ����������
��2������ͼת����ϵ��֪��Ӧ��Ϊһ��������������һ����̼��������Ϊ�����Ͷ�����̼�����ԭ�Ӹ����غ㡢��ʧ�����غ���д����ʽ��
��� �⣺��1�����������ᷴӦ����N2H62+�����ӷ�ӦΪN2H4+2H+=N2H62+��
�ʴ�Ϊ��N2H4+2H+=N2H62+��
�ڻ���ƽ����У���Ӧ�N2H4����˫��ˮ��H2O2�����������ǵ�����ˮ����Ӧ�Ļ�ѧ����ʽΪ��N2H4+2H2O2�TN2+4H2O��
�ʴ�Ϊ��N2H4+2H2O2�TN2+4H2O��
��N2H4���л�ԭ�ԣ�NO���������ԣ����߷�Ӧ����N2��ˮ�����������غ㶨�ɿ�д����ѧ����ʽΪN2H4+2NO=2N2+2H2O���÷�Ӧ�л�ԭ����N2H4��N��-2��0����������ΪNO��N��+2��0����ת��4e-������ȼ����1����ת��2mol���ӣ�������ȼ���µ�0.5mol������Ϊm=nM=0.5mol��32g/mol=16g��
�ʴ�Ϊ��16g��
��2������������һ��������һ����̼��������Ӧ���ɶ�����̼�͵�������ѧ����ʽΪ2NO+O2+4CO$\frac{\underline{\;����\;}}{\;}$4CO2+N2��
�ʴ�Ϊ��2NO+O2+4CO$\frac{\underline{\;����\;}}{\;}$4CO2+N2��
���� ���⿼�鵪���仯���������ԭ��Ӧ��֪ʶ��Ϊ��Ƶ���㣬���շ�Ӧ��Ԫ�صĻ��ϼ۱仯Ϊ���Ĺؼ����������ӷ�Ӧ����ѧ��Ӧ����ѧ��Ӧ��д�������Ӧ����Ŀ��飬�ۺ��Խ�ǿ����Ŀ�Ѷ��еȣ�

 ������ȫ�̼����ĩ���100��ϵ�д�
������ȫ�̼����ĩ���100��ϵ�д�| �� | ��ˮ�����c��H+��=l��l0-14mol/L����Һ�У�Ca2+��K+��Cl-��HCO3- | |
| �� | ��ʹpH��ֽ�ʺ�ɫ����Һ��Na+��NH4+��I-��NO3- | |
| �� | Kw/c��H+��=0.1mol/L����Һ��Na+��K+��SiO32-��NO3- | |
| �� | FeCl2��Һ��K+��Na+��SO42-��AlO2- |
| A�� | �� | B�� | �� | C�� | �� | D�� | �� |
| A�� | 1 mol H2������ֻ���ڱ�״���²�ԼΪ2g | |
| B�� | �ڱ�״����ij����������22.4L������Ϊ����������ʵ���Լ��1mol | |
| C�� | ��20��ʱ��1mol�κ����������ܱ�22.4L�� | |
| D�� | 1mol H2��O2�Ļ���������������Ϊ34g |
 �����ᣨ��ͼ����������������ҩ����Ұ���ܲ�����Ҷˮ�ա�������У���������ֹѪ���ã����� ���ڿ������˵������ȷ���ǣ�������
�����ᣨ��ͼ����������������ҩ����Ұ���ܲ�����Ҷˮ�ա�������У���������ֹѪ���ã����� ���ڿ������˵������ȷ���ǣ�������| A�� | ������ķ���ʽΪC9H8O4 | |
| B�� | 1 mol��������Ժͺ�4 molBr2��Ũ��ˮ��Ӧ | |
| C�� | 1 mol������������NaHCO3��Һ��Ӧ�ܲ��������Լ67.2L��CO2 | |
| D�� | �������ø�����ؼ��鿧��������е�̼̼˫�� |
| A�� | ��������������һ�� | B�� | �����Ũ������һ������������ | ||
| C�� | �¶Ƚ��͵�25�� | D�� | ����KHCO3��ĩ���� |




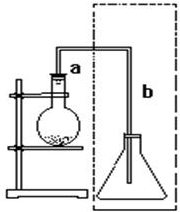 ��ͼ��ʵ������ȡ�屽��װ�ã�˵����
��ͼ��ʵ������ȡ�屽��װ�ã�˵����