��Ŀ����
8�� ��ͼ��ʵ������ȡ�屽��װ�ã�˵����
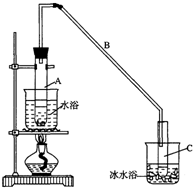
��ͼ��ʵ������ȡ�屽��װ�ã�˵������1������a�ε����õ��������������ӷ����壬����b��ĩ�˸���ƿ��Һ�汣��һ�ξ����Ŀ���Ƿ�ֹ����������ƿ��ʢ��������Һ��ͼ�����߷��ֵ�����֤��Ϊȡ����Ӧ��������β����
��2����Ӧ�õ���b���ڿɼ���ʲô�������������ƿ��ʢ��������Һ����Ӧ����ֵ���ɫ�����ܣ���ܡ����ܡ���֤�������嵥�ʷ���ȡ����Ӧ��������Ϊ���������ţ�
��3����Ӧ�õ����屽�ʺ�ɫ��������Ϊ�屽�����壻Ϊ��ȥ�����屽�е��壬�ɼ�������������Һ�Լ����÷�Һ�������룮
���� ���������廯���������·���ȡ����Ӧ�����屽��HBr�����ڷ�Ӧ���ȣ��塢���е�ͣ��ӷ����ӵ��ܵ����������п��ܺ���һ������ͱ��������ܵ�����Ϊ������������������Ӧ����ɵ�HBr����ˮ���գ���������Һ���飬�Դ˽����⣮
��� �⣺��1�����ӷ������Գ����ܵ����ó��˵���������ǻӷ��������������������ɵ�HBr������ˮ���������������Ե���b���¿ڲ��ܽ�û��Һ���У�����ƿ��ʢ��������Һ��ͼ�����߷��ֵ�������֤��Ϊȡ����Ӧ��������β����
�ʴ�Ϊ�����������������ӷ����壻��ֹ������֤��Ϊȡ����Ӧ��������β����
��2�������ɵ�HBr������ˮ���ɹ۲쵽����b�����а������֣������ɵ�HBr�л���������������֤�������嵥�ʷ���ȡ����Ӧ��
�ʴ�Ϊ�����������ܣ����������ţ�
��3���������������屽����Ӧ�õ����屽�ʺ�ɫ��Ϊ��ȥ�����屽�е��壬�ɼ�������������Һ��Ȼ���Һ���룬
�ʴ�Ϊ���֣��屽�����壻����������Һ����Һ��
���� �����Ա�������Ϊ���壬����ѧ���Ķ�װ�õ����⡢���ʵ��ᴿ�ȣ�����������ѧ�����õĿ�ѧ���������ʵ���������Ѷ��еȣ��Ƕ���ѧ֪ʶ���ۺ����ã�

| A�� | M+ | B�� | HA- | C�� | A2- | D�� | M+��HA- |
| A�� | PH=7����Һһ�������� | |
| B�� | ����Һc��H+��=c��OH-��ʱ����Һ������ | |
| C�� | ������Һһ�������ԣ���������Һ���������Ի���� | |
| D�� | ����Һ������ʱ��ˮ�����c��H+����c��OH-�� |
| A�� | 4m mol | B�� | 10m mol | C�� | $\frac{10m}{3}$mol | D�� | $\frac{2m}{3}$mol |
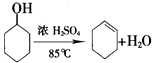
 ����ϩ��һ����Ҫ�Ļ���ԭ�ϣ�
����ϩ��һ����Ҫ�Ļ���ԭ�ϣ���1��ʵ���ҿ��ɻ������Ʊ�����ϩ����Ӧ�Ļ�ѧ����ʽ��
 ��
����2��ʵ��װ����ͼ��ʾ����10mL�����������Թ�A�У��ټ���1mLŨ���ᣬҡ�Ⱥ�������Ƭ��������������Ӧ��ȫ�����Թ�C�ڵõ�����ϩ��Ʒ��
�������ͻ���ϩ�IJ��������������£�
| �ܶ� ��g/cm3�� | �۵� ���棩 | �е� ���棩 | �ܽ��� | |
| ������ | 0.96 | 25 | 161 | ������ˮ |
| ����ϩ | 0.81 | -103 | 83 | ������ˮ |
���Թ�A����ˮԡ�е�Ŀ�������Ⱦ��ȣ����ڿ��£��Թ�C���ڱ�ˮԡ�е�Ŀ����ʹ����ϩҺ�������ٻӷ���
��3������ϩ��Ʒ�к����������������������ʣ����ƻ���ϩ�ķ����ǣ�
����ϩ��Ʒ�м���C�������ţ��������Һ����������ƣ���
A��Br2��CCl4��Һ B��ϡH2SO4 C��Na2CO3��Һ
���ٶԳ������Ӻ�Ļ���ϩ�������õ�����ϩ��Ʒ������ʱ��������ƿ��Ҫ����������ʯ�ң�Ŀ���dz�ȥ��Ʒ��������ˮ��
��ʵ���ƵõĻ���ϩ��Ʒ�����������۲��������ܵ�ԭ����C��
A������ʱ��70�濪ʼ�ռ���Ʒ
B��������ʵ����������
C���Ʊ���Ʒʱ���������Ʒһ������
��4���������ֻ���ϩ��Ʒ�ʹ�Ʒ�ķ�������������B��
A������ˮ�۲�ʵ������
B����������ƹ۲�ʵ������
C���������Ը��������Һ����۲�ʵ������
 �����仯�������������������м�����Ҫ�����ã�
�����仯�������������������м�����Ҫ�����ã� ��
��
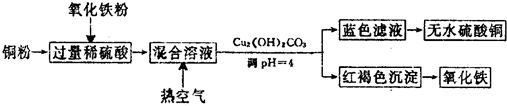
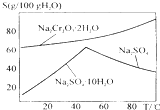 ��ҵ���Ը�������Ҫ�ɷ�FeO•Cr2O3����̼���ơ�����������Ϊԭ�������ظ����ƣ�Na2Cr2O7•2H2O������Ҫ��Ӧ���£�
��ҵ���Ը�������Ҫ�ɷ�FeO•Cr2O3����̼���ơ�����������Ϊԭ�������ظ����ƣ�Na2Cr2O7•2H2O������Ҫ��Ӧ���£�