��Ŀ����
��1����֪1gH2S������ȫȼ�գ�����Һ̬ˮ�Ͷ����������壬�ų�17.24kJ��������д��H2S����ȼ���ȵ��Ȼ�ѧ����ʽ��
��2������ƽ�����ʢ��ǿ��ԭ��Һ̬�£�N2H4����ǿ������Һ̬˫��ˮ�������ǻ�Ϸ�Ӧʱ������������������ˮ���������ų��������ȣ���֪0.4molҺ̬����������Һ̬˫��ˮ��Ӧ�����ɵ�����ˮ�������ų�256.652KJ����������Ӧ���Ȼ�ѧ����ʽΪ ��
��2������ƽ�����ʢ��ǿ��ԭ��Һ̬�£�N2H4����ǿ������Һ̬˫��ˮ�������ǻ�Ϸ�Ӧʱ������������������ˮ���������ų��������ȣ���֪0.4molҺ̬����������Һ̬˫��ˮ��Ӧ�����ɵ�����ˮ�������ų�256.652KJ����������Ӧ���Ȼ�ѧ����ʽΪ
���㣺�Ȼ�ѧ����ʽ
ר�⣺
��������1���Ȼ�ѧ����ʽ����д����Ҫע����У����ʵ�״̬����Ӧ�ȵ���ֵ�뵥λ����Ӧ�ȵ���ֵ�뻯ѧ����ʽǰ���ϵ�������ȣ��ݴ˽��
��2����Ӧ����ʽΪ��N2H4+2H2O2=N2+4H2O������0.4molҺ̬�·ų�������������1molҺ̬�·ų�������������д���Ȼ�ѧ����ʽ��
��2����Ӧ����ʽΪ��N2H4+2H2O2=N2+4H2O������0.4molҺ̬�·ų�������������1molҺ̬�·ų�������������д���Ȼ�ѧ����ʽ��
���
�⣺��1��1gH2S�����ʵ���Ϊn=
=
=
mol��ȼ������Һ̬ˮ�Ͷ�����������ʱ����17.24KJ����1molH2Sȼ�շ���586.16KJ�����Ȼ�ѧ����ʽΪ��H2S��g��+3/2O2��g��=SO2��g��+H2O��l����H=-586.16 kJ?mol-1��
�ʴ�Ϊ��H2S��g��+3/2O2��g��=SO2��g��+H2O��l����H=-586.16 kJ?mol-1��
��2����Ӧ����ʽΪ��N2H4+2H2O2�TN2+4H2O��0.4molҺ̬�·ų�256.652KJ����������1molҺ̬�·ų�������Ϊ
=641.63kJ��
���Է�Ӧ���Ȼ�ѧ����ʽΪ��N2H4��g��+2H2O2��l���TN2��g��+4H2O��g����H=-641.63kJ/mol��
�ʴ�Ϊ��N2H4��g��+2H2O2��l��=N2��g��+4H2O��g����H=-641.63kJ/mol��
| m |
| M |
| 1g |
| 34g/mol |
| 1 |
| 34 |
�ʴ�Ϊ��H2S��g��+3/2O2��g��=SO2��g��+H2O��l����H=-586.16 kJ?mol-1��
��2����Ӧ����ʽΪ��N2H4+2H2O2�TN2+4H2O��0.4molҺ̬�·ų�256.652KJ����������1molҺ̬�·ų�������Ϊ
| 256.652KJ |
| 0.4 |
���Է�Ӧ���Ȼ�ѧ����ʽΪ��N2H4��g��+2H2O2��l���TN2��g��+4H2O��g����H=-641.63kJ/mol��
�ʴ�Ϊ��N2H4��g��+2H2O2��l��=N2��g��+4H2O��g����H=-641.63kJ/mol��
���������⿼���Ȼ�ѧ����ʽ����д����Ŀ�ѶȲ���ע���Ȼ�ѧ����ʽ�뻯ѧ����ʽ������

��ϰ��ϵ�д�
�����Ŀ
����˵���д�����ǣ�������
| A�����ݶԽ��߹�������������ʾ��������� |
| B��[Cu��H2O��4]2+��Cu�ṩ�չ����H2O��O�ṩ�¶Ե����γ���λ�� |
| C��Ԫ�ص縺��Խ���ԭ�ӣ��������ӵ�����Խǿ |
| D�����Է��ӻ�Ϊ�������ǵ�����û������ |
���ֶ�����Ԫ�������ڱ��е�λ����ͼ������ֻ��MΪ����Ԫ�أ�����˵������ȷ���ǣ�������

| A��ԭ�Ӱ뾶Z��M |
| B��Zλ��Ԫ�����ڱ��е�2���ڡ��ڢ�A�� |
| C��X�������̬�⻯������ȶ��Ա�Z��ǿ |
| D��Y������������Ӧˮ��������Ա�X��ǿ |
������������ȷ���ǣ�������
| A��������������ᷴӦ������Ӧ |
| B������Na2SO4��Һ����ʹ�����ʱ��� |
| C����֬�����������������ijЩά���ص����� |
| D�����ǡ���֬����������һ�������¾�����ˮ�� |
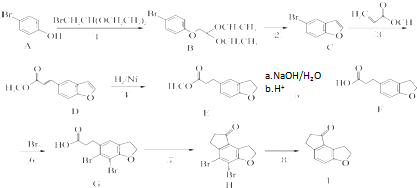
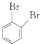 Ϊԭ���Ʊ�
Ϊԭ���Ʊ�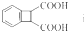 �ĺϳ�·������ͼ�����Լ���ѡ�������ϳ�·�߳��õı�ʾ����Ϊ��A
�ĺϳ�·������ͼ�����Լ���ѡ�������ϳ�·�߳��õı�ʾ����Ϊ��A
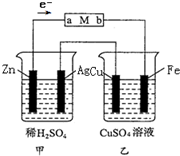 ����װ����ij�С�����ʵ���װ��ͼ���ش��������⣮
����װ����ij�С�����ʵ���װ��ͼ���ش��������⣮