��Ŀ����
8���ں��¡����ݵ������£��з�Ӧ2A��g��+2B��g��?C��g��+3D��g�����ִ�����;���ֱ���ƽ�⣮;����A��B����ʼŨ�Ⱦ�Ϊ2mol•L-1��C��D����ʼŨ��Ϊ0��;����C��D����ʼŨ��Ϊ2mol•L-1��6mol•L-1��A��B����ʼŨ��Ϊ0��������������ȷ���ǣ�������| A�� | ��;�����մﵽƽ��ʱ����ϵ�ڻ������İٷ������ͬ | |
| B�� | ��;�����մﵽƽ��ʱ����ϵ�ڻ������İٷ���ɲ�ͬ | |
| C�� | ;����ƽ������ʱ��һ������;����ƽ������ʱ�� | |
| D�� | ��ƽ��ʱ��;�������û��������ܶȵ���;�������û�����ܶȵ�һ�� |
���� �ɷ�Ӧ2A��g��+2B��g��?C��g��+3D��g����֪����Ӧǰ������Ļ�ѧ��������ȣ�ѹǿ��ƽ���ƶ�û��Ӱ�죬�����������������ȫת��ΪA��Bʱ��������ʵ�����ֵ��ȣ���ﵽ��ͬƽ��״̬���ݴ˽����жϣ�
��� �⣺��Ӧ2A��g��+2B��g��?C��g��+3D��g����֪����Ӧǰ������Ļ�ѧ��������ȣ�ѹǿ��ƽ���ƶ�û��Ӱ�죬�����������������ȫת��ΪA��Bʱ��������ʵ�����ֵ��ȣ���ﵽ��ͬƽ��״̬��
A��ƽ��״̬��ͬ����ϵ�ڻ������İٷ������ͬ����A��ȷ��
B������;�����մﵽ��ͬƽ��״̬����ϵ�ڻ�����İٷ������ͬ����B����
C����;���ﵽƽ��ʱŨ�ȴ�ѹǿ��Ӧ����Ӧ�ϴ�;����ƽ������ʱ�����;����ƽ������ʱ�䣬��C����
D����Ϊ��Чƽ��״̬����ϵ�ڻ������İٷ������ͬ���������ƽ����Է���������ͬ���������������ȣ������������Ǣ�����������2������ﵽƽ��ʱ��;����������ܶ�Ϊ;�����ܶȵ�$\frac{1}{2}$����D��ȷ��
��ѡAD��
���� ���⿼���Чƽ�����⣬��Ŀ�Ѷ��еȣ�ע���Чƽ����2��������ٻ�ѧ��Ӧǰ������Ļ�ѧ������֮�Ͳ��ȵķ�Ӧ�����������ʵ�����ȫ��ȣ��ڷ�Ӧǰ������Ļ�ѧ��������ȣ��������ʵ���֮����ȣ�

 53���ò�ϵ�д�
53���ò�ϵ�д�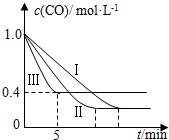 ���ü�����ˮ��Ӧ�Ʊ���������ԭ�����۲����ʸߣ�����ʵ���ƹ��ֵ��
���ü�����ˮ��Ӧ�Ʊ���������ԭ�����۲����ʸߣ�����ʵ���ƹ��ֵ����֪����CH4��g��+H2O��g��?CO��g��+3H2��g����H=+206.2kJ•mol-1
��CO��g��+H2O��g��?CO2��g��+H2��g����H=-42.3kJ•mol-1
��1�������ˮ�������ɶ�����̼���������Ȼ�ѧ����ʽΪCH4��g��+2H2O��g��=4H2��g��+CO2��g����H=+163.9 kJ/mol��
��2��Ϊ��̽����Ӧ�����Է�ӦCO��g��+H2O��g��?CO2��g��+H2��g����H=-42.3kJ•mol-1��Ӱ�죬ij�С�����������ʵ�飬ʵ��������ͼ��ʾ
| ��� | �¶� | ѹǿ | cʼ��CO�� | cʼ��H2O�� |
| �� | 530�� | 3MPa | 1.0mol•L-1 | 3.0mol•L-1 |
| �� | X | Y | 1.0mol•L-1 | 3.0mol•L-1 |
| �� | 630�� | 5MPa | 1.0mol•L-1 | 3.0mol•L-1 |
��ʵ���ӿ�ʼ��ƽ�⣬��ƽ����Ӧ�ٶ���v ��CO��=0.12mol•L-1•min-1��
��ʵ����ƽ��ʱCO��ת���ʴ��� ʵ����ƽ��ʱCO��ת���ʣ�����ڡ�����С�ڡ����ڡ�����
����530��ʱ��ƽ�ⳣ��K=1������1L������Ͷ��0.2mol CO��g����0.2mol H2O��g����0.1mol CO2��g����0.1mol H2��g������ʱ��ѧ��Ӧ��������������������� �ƶ���
| A�� | Na2CO3 | B�� | NaOH | C�� | Mg��OH��2 | D�� | NH3•H2O |
| A�� | �ױ������Ȼ�̼ | B�� | �������� | C�� | ��������һ�ȱ��� | D�� | �ƾ����� |
| A�� | ����ij��ɫ��ĩ������ʻ�ɫ��֤��ԭ��ĩ�к���Na+����K+ | |
| B�� | ʵ����Ҫ����500mL0.2mol•L-1KCl��Һ������������ƽ����7.45gKCl���� | |
| C�� | ����ʢ��NH4Cl������Թܣ��Թܵײ�������ʧ���Թܿ��о������ᣬ֤��NH4Cl���������� | |
| D�� | ������ʵ��ʱ����������ƿ��Ӧ�����ʯ���Է����У�����ڷ���ǰ�������Ǽӷ�ʯ��Ӧ����ֹͣ���ȣ���ȴ�ӷ�ʯ |
| A�� | ���������ﲻһ�����Ƿǽ��������� | |
| B�� | HCl��H2S��NH3���ǵ���� | |
| C�� | ǿ��ǿ������ӻ����� | |
| D�� | FeBr3��FeCl2��CuS������ֱ���û��Ϸ�Ӧ�Ʊ� |
 2NH3��g����H=-92kJ•mol-1��
2NH3��g����H=-92kJ•mol-1��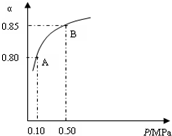 ��������͵����������dz��õĹ�ҵԭ�ϣ���Ҳ�Ǵ�������Ҫ��Ⱦ��ۺ���������Ⱦ�ǻ�����ѧ��ǰ����Ҫ�о�����֮һ��
��������͵����������dz��õĹ�ҵԭ�ϣ���Ҳ�Ǵ�������Ҫ��Ⱦ��ۺ���������Ⱦ�ǻ�����ѧ��ǰ����Ҫ�о�����֮һ��