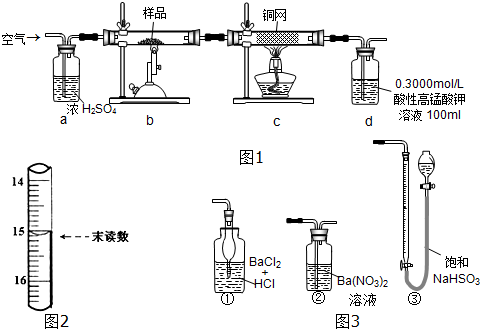
��Ŀ����
10������˵����ȷ���ǣ�������| A�� | ������0.4 mol/L HB��Һ��0.2 mol/L NaOH��Һ�������Ϻ���Һ��pH=3��������Һ������Ũ�ȵĴ�С˳��Ϊ��c ��Na+����c ��B-����c ��H+����c ��OH-�� | |
| B�� | ����ʱ��pH��Ϊ2��CH3COOH��Һ��HCl��Һ��pH��Ϊ12�İ�ˮ��NaOH��Һ��������Һ����ˮ�����c��H+����� | |
| C�� | ������0.1 mol/L��������Һ ��NH4Al��SO4��2����NH4Cl����NH3•H2O����CH3COONH4��c ��NH$_4^+$���ɴ�С��˳���ǣ��ڣ��٣��ܣ��� | |
| D�� | 0.1 mol/L NaHB��Һ����pHΪ4��c��HB-����c��H2B����c��B$_{\;}^{2-}$�� |
���� A��0.4mol/LHB��Һ��0.2mol/LNaOH��Һ�������Ϻ�����Ӧ����ʵ������0.1mol/LHB��Һ��0.1mol/L NaB��Һ������Һ�����ԣ�Ȼ�����õ���غ㼰�����غ������������Ũ�ȵĹ�ϵ��
B����������ˮ���룬���������ӻ��������������Ũ����ͬ��������ˮ����̶���ͬ��
C����������ˮ������笠����ӵ�ˮ�⣻��笠�����ˮ�⣻��������룬�ҵ���ij̶Ⱥ������ܴ��������ˮ��ٽ�笠�����ˮ�⣻
D��0.1mol•L-1��NaHB��ҺpHΪ4��˵��HB-Ϊ���������HB-����̶ȴ�����ˮ��̶ȣ��ݴ��жϣ�
��� �⣺A����Ϻ�Ϊ0.1mol/LHB��Һ��0.1mol/L NaB��Һ����Һ��PH=3����Һ�����ԣ�������ĵ�������ε�ˮ�⣬��c��B-����c��HB������������0.1mol/L����c��B-����c��Na+����c��HB������Һ������˵����Һ��c��H+����c��OH-��������Һ�е������Ӻ�����������Ũ�ȶ���С��С�����Ũ�ȣ���������Ũ�ȴ�С˳����c��B-����c��Na+����c��HB����c��H+����c��OH-������A����
B����������ˮ���룬���������ӻ��������������Ũ����ͬ��������ˮ����̶���ͬ��pH=2��CH3COOH��Һ��HCl��Һ��������Ũ�ȵ���pH=12�İ�ˮ��NaOH��Һ������������Ũ�ȣ���������ˮ�̶���ȣ�������ˮ�����c��H+����ȣ���B��ȷ��
C��ͬŨ�ȵ�������Һ����NH4Al��SO4��2��NH4Cl��NH3•H2O����CH3COONH4�������������ˮ������笠����ӵ�ˮ�⣻����笠�����ˮ�⣻��������룬�ҵ���ij̶Ⱥ������ܴ��������ˮ��ٽ�笠�����ˮ�⣬��c��NH4+���ɴ�С��˳���ǣ��٣��ڣ��ܣ��ۣ���C����
D��0.1mol•L-1��NaHB��ҺpHΪ4��˵��HB-Ϊ���������HB-����̶ȴ�����ˮ��̶ȣ���������B2-��ˮ������H2B������c��B2-����c��H2B����������ˮ��̶Ȳ���c��HB-���������c��HB-����c��B2-����c��H2B������D����
��ѡB��
���� ��������ˮ�⡢����Ũ�ȴ�С�Ƚϵ�֪ʶ���ѶȽϴ�ע���������õ���غ㡢�����غ�������غ������⣮

 ǧ�������������ĩ�����Ծ�����ϵ�д�
ǧ�������������ĩ�����Ծ�����ϵ�д�
| A�� | ��Zn����ϡHCl��Ӧ��H2 | B�� | �ô���ʯ��ϡHCl��Ӧ��CO2 | ||
| C�� | ��Cu��ϡHNO3��Ӧ��NO | D�� | ��MnO2��ŨHCl��Ӧ��Cl2 |
| A�� | ���õ���ˮ����û�з��ӣ�ֻ������ | |
| B�� | ������ˮ�ڹ��յ������£����Բ������壬������������ | |
| C�� | ������ˮ�еμ���������Һ��û���κ����� | |
| D�� | ������ˮ����ʹ����IJ�����ɫ |
| A�� | �����������м���������30%NaOH��Һ���ڼ��ȵ������¹۲��������ʧ | |
| B�� | �Ҵ���Ũ�����ϣ�������140����ȡ��ϩ | |
| C�� | ��2mL�ļױ��м���3��KMnO4������Һ���������۲���Һ��ɫ��ȥ | |
| D�� | 1 mol•L-1CuSO4��Һ2 mL��0.5mol•L-1NaOH��Һ2mL��Ϻ����40%����ȩ��Һ0.5mL��������й۲�ש��ɫ���������� |
| A�� | �����̪��Һ�Ժ�ɫ����Һ�У�K+��Na+��Cu2+��SO42- | |
| B�� | ��$\frac{c��{H}^{+}��}{c��O{H}^{-}��}$=10-12����Һ�У�K+��Ba2+��Cl-��ClO- | |
| C�� | ˮ���������c��H+��=10-13mol•L-1 ����Һ�У�Na+��Cl-��NO3-��CH3COO- | |
| D�� | pH=0����Һ�У�Na+��NO3-��S2-��Cl- |