��Ŀ����
1��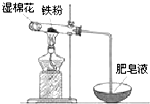 �����ճ���������;��㡢�������Ľ������ϣ�
�����ճ���������;��㡢�������Ľ������ϣ���1�������£�������������ʢװŨ�����ԭ����Ũ����ʹ�������γ�һ�������ȶ�������Ĥ��
��2��ijʵ��С��������ͼװ����֤����ˮ�����ķ�Ӧ��
��ʪ�����������ṩˮ�������Թ��з�Ӧ�Ļ�ѧ����ʽ��3Fe+4H2O��g��$\frac{\underline{\;����\;}}{\;}$Fe3O4+4H2��
��ʵ�������ȡ��������Ӧ��Ĺ������Թ��У�����������ᣬ������ȫ�ܽ⣬������Һ�д��ڵ���������b������ţ���
a��һ����Fe2+��H+��Fe3+
b��һ����Fe2+��H+��������Fe3+
c��һ����Fe2+��Fe3+�������� H+
d��һ����Fe3+��H+��������Fe2+
��3������ȡһ��������������������Ũ�����У����ȣ���ַ�Ӧ���ռ����壮���ⶨ�����к���SO2��CO2��H2��
������Ũ���ᷴӦ�Ļ�ѧ����ʽ��2Fe+6H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$Fe2��SO4��3+3SO2��+6H2O��
�������л���CO2��ԭ���ǣ��û�ѧ����ʽ��ʾ��C+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CO2+2SO2+2H2O��
�۽��ռ���������ͨ��������ˮ�У���Ӧ�����ӷ���ʽΪSO2+Br2+2H2O=4H++2Br-+SO42-��
���� ��1��������Fe��Ũ���ᷢ���ۻ���
��2����ʪ������ˮ�������Է�Ӧ��˵�ṩ��ˮ�����ڸ�������ˮ��Ӧ����������������������
�ڲ�����������������+2�۵�����+3�۵��������п����й���������
��3�������ǽ������л�ԭ�ԣ�Ũ������������ԣ�����������+3������Ũ���ᱻ��ԭ��+4�۵���
��̼���ʺ�Ũ�����ڼ��ȵ����������ɶ�����̼�Ͷ�������
�����ɵ�SO2���л�ԭ�ԣ�ͨ��������ˮ�У�����SO2+Br2+2H2O=2HBr+H2SO4��
��� �⣺��1�������£�����Ũ�����з����ۻ�����ʹ�������γ�һ�������ȶ�������Ĥ��������Ʒ��������ʢ��Ũ���ᣬ
�ʴ�Ϊ��Ũ����ʹ�������γ�һ�������ȶ�������Ĥ��
��2������Ϊ������ˮ�����ķ�Ӧ���Թ��������ۣ�����ʪ���������ṩˮ���������ڸ�������ˮ��Ӧ������������������������Ӧ����ʽΪ3Fe+4H2O��g��$\frac{\underline{\;����\;}}{\;}$Fe3O4+4H2��
�ʴ�Ϊ���ṩˮ������3Fe+4H2O��g��$\frac{\underline{\;����\;}}{\;}$Fe3O4+4H2��
������ˮ������Ӧ������Ϊ����������������������������ᣬ������ȫ�ܽ⣬�ط�����Ӧ��Fe3O4+8HCl=FeCl2+2FeCl3+4H2O������������Һ�д��ڵ���������һ����Fe2+��H+��������������ɫ�����п��ܺ��й������������ܽ�����������ȫ��ת���ɶ��������ӣ�Fe+2Fe3+�T3Fe2+������������Һ�д��ڵ���������һ����Fe2+��H+��������Fe3+��
�ʴ�Ϊ��b��
��3��������Ũ���ᷴӦ����������+3������Ũ���ᱻ��ԭ��+4�۵��������������Ͷ��������ˮ����ӦΪ2Fe+6H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$Fe2��SO4��3+3SO2��+6H2O��
�ʴ�Ϊ��2Fe+6H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$Fe2��SO4��3+3SO2��+6H2O��
���ڷ�Ӧ�Ĺ����У�̼���ʿ��Ժ�Ũ���ᷴӦ���ɶ�����̼��������������壬������ӦΪC+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CO2+2SO2+2H2O��
�ʴ�Ϊ��C+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CO2+2SO2+2H2O��
��SO2���л�ԭ�ԣ�ͨ��������ˮ�У�����SO2+Br2+2H2O=2HBr+H2SO4�����ӷ���ʽ����ʽΪSO2+Br2+2H2O=4H++2Br-+SO42-��
�ʴ�Ϊ��SO2+Br2+2H2O=4H++2Br-+SO42-��
���� ���⿼������ʵ�鷽������ƣ�Ϊ��Ƶ���㣬�������ʵ����ʡ������ķ�Ӧ��Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬ע��������ԭ��Ӧ��Ӧ�ã���Ŀ�ѶȲ���

| A�� | CH4û��ͬ���칹�� | B�� | CH3Clû��ͬ���칹�� | ||
| C�� | CH2Cl2û��ͬ���칹�� | D�� | CCl4û��ͬ���칹�� |
| A�� | �п�ѧ�ҳƹ��ǡ�21���͵���Դ��������Ҫ��������Ϊ�뵼����ϵĹ���̫���ܷ�������о�����Ҫ������ | |
| B�� | ��K2FeO4����Cl2��������ˮ������ɱ���������ã����о�ˮ���� | |
| C�� | ������㡱���õ�ֲ������ʹKMnO4��Һ��ɫ�������ڸ߷��ӻ����� | |
| D�� | �����Ȼ��Ƶ���ѩ����ʹ�û�ӿ������ĸ�ʴ |
| A�� | ����ת������һ��ʵ�֣��ֹ�$��_{����}^{Cl_{2}}$SiCl4$��_{����}^{H_{2}}$Si | |
| B�� | Ԫ��C�ĵ���ֻ���ڽ��ʯ��ʯī����ͬ�������� | |
| C�� | Ӳ�ȣ�����裼���ʯ | |
| D�� | �������ý�̿��ԭSiO2��ȡ�ֹ� |
| A�� | H2SO4��NaOH-��ʴƷ | B�� | CH4��C2H4-��ȼҺ�� | ||
| C�� | K��Na-��ʪ��ȼ��Ʒ | D�� | KMnO4��HNO3-������ |
| A�� | 37Cl��35Cl | B�� | O2��O3 | ||
| C�� | CH3CH2OH��CH3OCH3 | D�� | CH4��C2H6 |
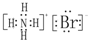 ��
�� ��1����֪�缫���ϣ�����ͭ������ʯī��п�������������Һ��CuCl2��Һ��Fe2��SO4��3��Һ�����ᣮ��Ҫ��ش��������⣺
��1����֪�缫���ϣ�����ͭ������ʯī��п�������������Һ��CuCl2��Һ��Fe2��SO4��3��Һ�����ᣮ��Ҫ��ش��������⣺