��Ŀ����
ij����С�����Fe(OH)3������Ʊ�ʵ�鲢���������ʡ�
��1�����мס��ҡ�������ͬѧ����Fe(OH)3������Ʊ���
�� ��ͬѧ�IJ����ǣ�ȡһС�ձ�������25 mL����ˮ���������ڣ����ˮ����μ���1��2 mL FeCl3������Һ��������������Һ�ʺ��ɫ��ֹͣ���ȡ�
�� ��ֱ�Ӽ��ȱ���FeCl3��Һ��
�� �����ˮ�еμӱ���FeCl3��Һ��Ϊ��ʹ��Ӧ���г�֣����10���ӡ�
����Ϊ��λͬѧ���Ʊ������������� ���������ý����ʲô������֤����Fe(OH)3�������ɣ� ��
��2��Fe(OH)3�����ȶ����ڵ���Ҫԭ����_________________��
A������ֱ��С��1 nm B������������� C�������������˶� D������������ֽ
��3��Fe(OH)3����������FeCl3��Һ��ʵ�������____________��
A��Fe(OH)3�������ӵ�ֱ����1��100 nm֮�� B��Fe(OH)3������ж����ЧӦ
C��Fe(OH)3�����Ǿ�һ�ķ�ɢϵ D��Fe(OH)3����ķ�ɢ��������ֽ
��4���ᴿ����Fe(OH)3���峣�õķ�����________���� ��Fe(OH)3�����е�������ֱ�������������˹��̵�ʵ������_______________________��
��Fe(OH)3�����е�������ֱ�������������˹��̵�ʵ������_______________________��
 �ŵ������ϵ�д�
�ŵ������ϵ�д�| A�� | ��0.1mol•L-1�İ�ˮ�м�����������粒��壬����Һ��$\frac{c��O{H}^{-}��}{c��N{H}_{3}•{H}_{2}O��}$���� | |
| B�� | ���ʵ���Ũ����ȵĢ٣�NH4��2SO4��Һ����NH4HCO3��Һ����NH4Cl ��Һ���ܣ�NH4��2Fe��SO4��2��Һ�У�c��${NH}_{4}^{+}$���Ĵ�С��ϵ���ܣ��٣��ڣ��� | |
| C�� | ��0.2 mol•L-1��ijһԪ��HA��Һ��0.1 mol•L-1 NaOH��Һ�������Ϻ���ҺpH����7����Ӧ��Ļ��Һ��c��HA����c��Na+����c��A-�� | |
| D�� | ���ʵ���Ũ��֮��Ϊ1��1��NaClO��NaHCO3�����Һ�У�c��HClO��+c��ClO-���Tc��${HCO}_{3}^{-}$��+c��${CO}_{3}^{2-}$�� |
| A�� | 94g | B�� | 14g | C�� | 47g | D�� | 7g |
| A�� | NaOH | B�� | KMnO4 | C�� | KSCN | D�� | ���� |
| A�� | 3�� | B�� | 4�� | C�� | 5�� | D�� | 6�� |
| A�� | ��Ũ������������̹��������� | |
| B�� | ��״���£���1 g��ƬͶ��20 mL 18 mol•L-1������ | |
| C�� | ��100 mL 4 mol•L-1�����м���5.6 g �� | |
| D�� | ��5��107 Pa��500�������ý���������£��õ����������ϳɰ� |


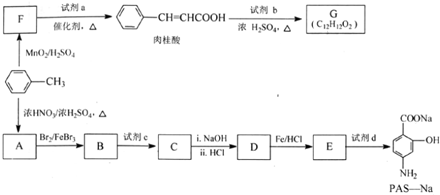
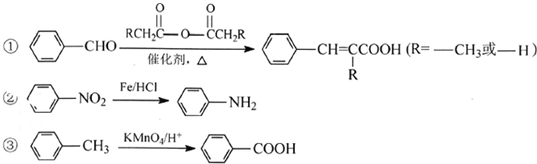
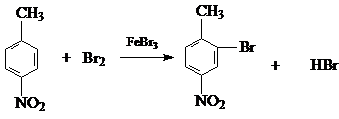 ��
��  ��
�� 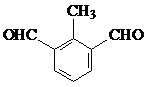 ��
�� ��
��