��Ŀ����
6��1000mlij��ɫ����Һ�г�����0.2mol•L-1��Na+�⣬�����ܺ������������е�һ�ֻ���֣�| ������ | K+��NH4+��Fe3+��Ba2+ |
| ������ | Cl-��CO32-��HCO3-��SO42- |
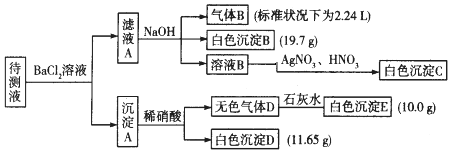
��1��ԭ��Һ�п϶����ڵ������ǣ���Na+��CO32-��HCO3-��SO42-��K+��NH4+���϶������ڵ�������Fe3+��Ba2+��
��2��д�����ɰ�ɫ����B�����ӷ���ʽ��HCO3-+Ba2++OH-=BaCO3��+H2O��д������A����ϡ��������ӷ���ʽ��BaCO3+2H+=Ba2++H2O+CO2����
��3������ҺA�ͳ���A�ֿ��IJ����ǹ��ˣ�ϴ�ӣ�ϴ�ӵIJ������ز�����������©���м�ˮ���պý�û��������ˮ��Ȼ���º��ظ�����2-3�Σ�
���� ����Һ���Ȼ�����Һ��Ӧ�õ�����A������Һ�п��ܺ���CO32-��SO32-��SO42-��������м���ϡ�����������壬���в��ֳ������ܽ⣬����Һ�д���CO32-�����ܴ���SO32-��SO42-�е����ֻ�һ�֣��������ӹ���֪����Һ�в�����Ba2+��
��ҺA����Ba2+�����������NaOH��Һ�õ�����B����ɫ����B������Һ��һ������NH4+��HCO3-��һ��������Fe3+������BΪNH3����ɫ����BΪBaCO3����ҺB��ͨ����������dz����ɫ��Һ����Һ��һ��û��Br-����ҺB�м�����������������Һ�õ���ɫ����C��CΪAgCl��˵����ҺB�к���Cl-�����ڼ����Ȼ�����Һ������ȷ��ԭ��Һ���Ƿ���Cl-����ϵ���غ㼰���ӷ�Ӧ�����
��� �⣺����Һ���Ȼ�����Һ��Ӧ�õ�����A������Һ�п��ܺ���CO32-��SO32-��SO42-��������м���ϡ�����������壬���в��ֳ������ܽ⣬����Һ�д���CO32-�����ܴ���SO32-��SO42-�е����ֻ�һ�֣��������ӹ���֪����Һ�в�����Ba2+��
��ҺA����Ba2+�����������NaOH��Һ�õ�����B����ɫ����B������Һ��һ������NH4+��HCO3-��һ��������Fe3+������BΪNH3����ɫ����BΪBaCO3����ҺB��ͨ����������dz����ɫ��Һ����Һ��һ��û��Br-����ҺB�м�����������������Һ�õ���ɫ����C��CΪAgCl��˵����ҺB�к���Cl-�����ڼ����Ȼ�����Һ������ȷ��ԭ��Һ���Ƿ���Cl-��
n��NH4+��=n��NH3��=$\frac{2.24L}{22.4L/mol}$=0.1mol����c��NH4+��=0.1mol/L����Һ��c��Na+��=0.2mol/L��
B��̼�ᱵ�������ʵ���Ϊ$\frac{19.7g}{197g/mol}$=0.1mol����n��HCO3-��=0.1mol����c��HCO3-��=0.1mol/L��
E��̼��ƣ������ʵ���Ϊ$\frac{10g}{100g/mol}$=0.1mol����n��CO32-��=0.1mol����c��CO32-��=0.1mol/L��
D�����ᱵ�������ʵ���=$\frac{11.65g}{233g/mol}$=0.05mol����n��SO42-��=0.05mol����c��SO42-��=0.05mol/L��
c��HCO3-��=0.1mol/L��c��CO32-��=0.1mol/L��c��SO42-��=0.05mol/L����λ���Ϊ�����=1��0.1mol/L+1��0.2mol/L=0.3mol/L����λ����ڸ����=1��0.1mol/L+2��0.1mol/L+2��0.05mol/L=0.4mol/L����λ���Ϊ����ɣ���λ����ڸ���ɣ���һ������K+������Һ��û��Cl-��K+����Ũ����С�����ݵ���غ��֪��c��K+����СŨ��Ϊ0.4mol/L-0.3mol/L=0.1mol/L��
��1��������������֪��ԭ��Һ�п϶����ڵ������ǣ���Na+��CO32-��HCO3-��SO42-��K+��NH4+����Һ��һ��û�е��������ǣ�Fe3+��Ba2+��
�ʴ�Ϊ��CO32-��HCO3-��SO42-��K+��NH4+��Fe3+��Ba2+��
��2��������������֪����ɫ����BΪ̼�ᱵ������HCO3-��Ba2+��OH-��Ӧ���ɣ���Ӧ���ӷ���ʽΪHCO3-+Ba2++OH-=BaCO3��+H2O������A����ϡ��������ӷ���ʽΪBaCO3+2H+=Ba2++H2O+CO2����
�ʴ�Ϊ��HCO3-+Ba2++OH-=BaCO3��+H2O��BaCO3+2H+=Ba2++H2O+CO2����
��3������ҺA�ͳ���A�ֿ��IJ����ǹ��ˣ�ϴ�ӣ�ϴ�ӵIJ������ز�����������©���м�ˮ���պý�û��������ˮ��Ȼ���º��ظ�����2-3�Σ�
�ʴ�Ϊ���ز�����������©���м�ˮ���պý�û��������ˮ��Ȼ���º��ظ�����2-3�Σ�
���� ���⿼��������ƶϣ�Ϊ��Ƶ���㣬�������ʵ����ʡ������ķ�Ӧ�����ʵ�������ؼ���Ϊ���Ĺؼ������ط������ƶ������Ŀ��飬ע��Ԫ�ػ�����֪ʶ��ʵ��Ľ�ϣ���Ŀ�ѶȲ���

 ���ſ����ϵ�д�
���ſ����ϵ�д� ���Ŀ����ϵ�д�
���Ŀ����ϵ�д� ������ӱ������ͯ������ϵ�д�
������ӱ������ͯ������ϵ�д�| A�� | SO2 | B�� | Na2O | C�� | CO | D�� | Na2O2 |
| A�� | �����ƹ�̫���ܡ����ܡ������ܼ�ˮ�ܵȵ�ʹ�ã����ٻ�ʯȼ�ϵ�ʹ�� | |
| B�� | ��ǿ����������ˮ�ѵ����״���������ˮ�帻Ӫ���� | |
| C�� | ���Ͼɵ������ֹ���е��ؽ���������Ⱦ���� | |
| D�� | ����ʵʩ����ȼ�ϡ������������������������������͵�����������Ⱦ |
| A�� | 20��ʱ�������ܶ�Ϊ2.7 g/cm3����1��Alԭ�ӵ����Ϊ��$\frac{10}{{N}_{A}}$��cm3 | |
| B�� | 25�桢101KPa�£�16 g O2��O3�Ļ�������к�������Ϊ8NA | |
| C�� | 25�桢101KPa�£�11.2L Cl2���еķ�����С��0.5NA | |
| D�� | 0�桢101KPa�£��������CH4��CCl4�к�Cԭ������� |
| A�� | ��1����2����3����4 | B�� | ��1����2����4����3 | C�� | ��2����1����3����4 | D�� | ��2����1����4����3 |
| A�� | HI��H2��I2��Ũ����� | B�� | HI��H2��I2��Ũ�Ȳ��ٷ����仯 | ||
| C�� | HI��H2��I2���ܱ������й��� | D�� | �������������ɫ���ٷ����仯 |
| A�� | MnO4-��Mn2+ | B�� | H2O2��H2O | C�� | H2��H2O | D�� | Zn��Zn2+ |