��Ŀ����
11����1��ij��ˮ��Ӳ�Ƚϴ���Ҫ����Ϊ���к��н϶��D��A��Cl- ��SO42- B��HCO3- C��Na+D��Ca2+��Mg2+
��2����������Ӳ�ȵ�Ӳˮ�ɲ���ҩ���������ӽ�������������������ҩ�����������â�ʯ�Һ͢ڴ������Ӧ�ü�����Ǣ�ʯ�ң�Ŀ���Ǣ�Ӧ�ȼ�ʯ�ң���Ӵ�����������ĸ����ӿ�ͨ������ʹ֮�������������з�������Ҫ��ѧ��ӦΪ��д����Ӧ�Ļ�ѧ��Ӧ����ʽ�������ӷ�Ӧ��ֻд�����ӷ�Ӧ����ʽ����CaO+H2O=Ca2++2OH-��Mg2++2OH-=Mg��OH��2����CO32-+Ca2+=CaCO3����
��3����������Ҫ�Ļ���ԭ�ϣ�����Ҫ�����������Тٶ���������Ʊ� �ڶ��������������������� ����������ת��Ϊ���ᣬ������Ҫ�����������Ǣ٣�ԭ���Ƿ�ֹ�ڽӴ����д����ж���
���� ��1����ˮ�к��н϶�ĸ����ӡ�þ���ӣ�����ˮ��Ӳ�Ƚϴ�
��2��ҩ�������������Լ�Ϊ����������ƣ���������ˮ��Ӧ�����������ƣ��������Ƶ������������������þ���ӷ�Ӧ����������þ������̼����������ӷ�Ӧ����̼��Ƴ������ݴ˽��н��
��3���ӷ���¯�г�����������Ҫ���������Ϊ�˷�ֹ�ڽӴ����еĴ����ж���
��� �⣺��1�����ھ�ˮ�к��н϶�Ca2+��Mg2+�����¾�ˮ��Ӳ�Ƚϴ�
�ʴ�Ϊ��D��
��2����������Ӳ�ȵ�Ӳˮ�ɲ���ҩ���������ӽ�������������������ҩ������������ʯ�Һʹ���ȼ���ʯ�ң�������Ӧ��CaO+H2O=Ca2++2OH-���������Ƶ������������������þ���ӷ�Ӧ����������þ������Mg2++2OH-=Mg��OH��2�����ټ���̼���ƽ�������ת����̼��Ƴ�������Ӧ�����ӷ���ʽΪ��CO32-+Ca2+=CaCO3����
�ʴ�Ϊ��ʯ�ң����ʯ�ң�Ҫ��ȥ��þ���ӣ�Ӧ�ȼ�ʯ�ң���Ӵ�����������ĸ����ӿ�ͨ������ʹ֮����������CaO+H2O=Ca2++2OH-��Mg2++2OH-=Mg��OH��2����CO32-+Ca2+=CaCO3����
��3���Ʊ���������У��ӷ���¯�г���������٣��ڴ�����ǰ��Ҫ���������ֹ�ڽӴ����еĴ����ж���
�ʴ�Ϊ���٣���ֹ�Ӵ��ҵĴ����ж���
���� ���⿼����ˮ�ľ����봦�������ӷ���ʽ��д�����Ṥҵ��֪ʶ����Ŀ�ѶȲ�����ȷˮ����������Ϊ���ؼ���ע���������Ṥҵԭ��������������ѧ���ķ������������Ӧ��������

 ��У����ϵ�д�
��У����ϵ�д�| A�� | ʯ�ࣺCaSO4•2H2O���������������ģ�� | |
| B�� | �ؾ�ʯ��BaCO3��������ɫ���ϡ�ҽ���ϡ����͡� | |
| C�� | �̷���FeSO4•7H2O����������ˮ������ֹȱ����ƶѪ��ҩ�� | |
| D�� | ������CuSO4•5H2O������������ũҩ |
| A�� | pH=1����Һ�У�K+ CH3COO- SO42- Na+ | |
| B�� | ����0.1mol/LCa2+����Һ�У�Na+SO42- K+ Cl- | |
| C�� | ����0.1mol/LMg2+����Һ�У�Na+SO42- K+ HCO3- | |
| D�� | �����̪�Ժ�ɫ����Һ��K+ HCO3-Ba2+Cl- |
| ������ | K+��NH4+��Fe3+��Ba2+ |
| ������ | Cl-��CO32-��HCO3-��SO42- |
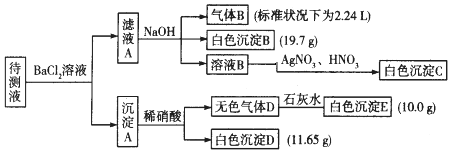
��1��ԭ��Һ�п϶����ڵ������ǣ���Na+��CO32-��HCO3-��SO42-��K+��NH4+���϶������ڵ�������Fe3+��Ba2+��
��2��д�����ɰ�ɫ����B�����ӷ���ʽ��HCO3-+Ba2++OH-=BaCO3��+H2O��д������A����ϡ��������ӷ���ʽ��BaCO3+2H+=Ba2++H2O+CO2����
��3������ҺA�ͳ���A�ֿ��IJ����ǹ��ˣ�ϴ�ӣ�ϴ�ӵIJ������ز�����������©���м�ˮ���պý�û��������ˮ��Ȼ���º��ظ�����2-3�Σ�
| A�� | Al2O3--��Al��OH��3 | B�� | Cu--��CuCl2 | C�� | Fe--��FeCl2 | D�� | Na--��NaOH |
| A�� | ��ʯī���缫���AlC13��Һ��2Cl-+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$Cl2��+H2��+2OH- | |
| B�� | ���Ը��������Һ��H2O2��Һ��ϣ�2MnO4-+3H2O2+6H+�T2Mn2++4 O2��+6H2O | |
| C�� | ��NaHSO4��Һ�еμ�Ba��OH��2��Һ�����ٲ���������Ba2++OH-+H++SO42-�TBaSO4��+H2O | |
| D�� | ��Fe��NO3��2��NaBr�����Һ�еμ�ϡ���6Br -+2NO3-+8H+�T3Br2+2NO��+4H2O |