��Ŀ����
����Ŀ�������½�������ʵ�飬����ʵ��������������õ��Ľ�����ȷ����
ѡ�� | ʵ����������� | ���� |
A | �����£�������Һ��ͨ����CO2����Һ����� | ̼������Աȱ��ӵ�ǿ |
B | �����Ҵ���Ũ����Ļ����Һ��������������ͨ����������KMnO4��Һ����Һ�Ϻ�ɫ��ȥ | ����ϩ���� |
C | ��5 mL 0.1 mol��L1 KI��Һ�м���1 mL 0.1 mol��L1 FeCl3��Һ����ַ�Ӧ����ȡ��Һ����ˮ���еμ�KSCN��Һ����Һ��Ѫ��ɫ | I����Fe3+�ķ�Ӧ��һ���� |
D | ��NaHCO3��Һ�еμ���ɫʯ����Һ����Һ���� | Kw��Ka1(H2CO3)��Ka2(H2CO3) |
A.AB.BC.CD.D
���𰸡�AC
��������
A�������£�������Һ��ͨ����CO2��̼�Ὣ�����û���������Һ����ǣ�����ǿ�������ᣬ���Եó����ۣ�̼������Աȱ��ӵ�ǿ����A��ȷ��
B�������Ҵ���Ũ����Ļ����Һ��������������ͨ����������KMnO4��Һ����Һ�Ϻ�ɫ��ȥ�������ǻӷ������Ҵ������Ը������������Ҳ�������Ҵ�������ȥ��Ӧ�õ�����ϩʹ���Ը�����ص���ɫ��ɫ����B����
C����5 mL 0.1 mol��L1KI��Һ�м���1 mL 0.1 mol��L1FeCl3��Һ�������Ӻ������ӷ���������ԭ��Ӧ���ɵ���������ӣ���Ӧ����ȡ��Һ����ˮ���еμ�KSCN��Һ����Һ��Ѫ��ɫ��˵����Һ�л����������ӣ�û�з�Ӧ�꣬˵��I����Fe3+�ķ�Ӧ��һ���ȣ���C��ȷ��
D����NaHCO3��Һ�еμ���ɫʯ����Һ����Һ������˵����Һ�ʼ��ԣ�ͬʱ˵��̼�������ˮ��̶ȴ��ڵ���̶ȣ�HCO3-+H2OH2CO3+OH-��Kh=![]() ��H2CO3HCO3-+H+��Ka1=
��H2CO3HCO3-+H+��Ka1=![]() ��HCO3-H++CO32-��Ka2=
��HCO3-H++CO32-��Ka2=![]() ��Ka1(H2CO3)��Ka2(H2CO3)��
��Ka1(H2CO3)��Ka2(H2CO3)��![]() ��
��![]() ��
��![]() ��Kh��Ka����
��Kh��Ka����![]() ��
��![]() ����ʽ����ͬ����c(H+)�����Եõ�
����ʽ����ͬ����c(H+)�����Եõ�![]() ����Kw��Ka1(H2CO3)��Ka2(H2CO3)����D����
����Kw��Ka1(H2CO3)��Ka2(H2CO3)����D����
��ѡAC��

����Ŀ��ijʵ��С�����ʵ�飬��������KMnO4��Һ��H2C2O4��Һ��Ӧ���ⶨ��Һ��ɫ��ʧ����ʱ��ķ������о�Ũ�ȶԷ�Ӧ���ʵ�Ӱ�졣��ѡ���ʵ��ҩƷ�У�0.01mol��L��1����KMnO4��Һ��0.1mol��L��1����KMnO4��Һ��0.1mol��L��1H2C2O4��Һ��0.2mol��L��1H2C2O4��Һ��
��1��H2C2O4��Һ������KMnO4��Һ��Ӧ�����ӷ���ʽΪ_______________��
��2�����������ʵ����Ʊ���
ʵ�� | ����KMnO4��Һ | H2C2O4��Һ | ��ɫʱ��/s | ||
c/(mol��L��1) | V/mL | c/(mol��L��1) | V/mL | ||
�� | 0.01 | 4 | 0.1 | 2 | t1 |
�� | a | 4 | b | 2 | t2 |
�ٱ���a=________��b=________��
�ڼ���ʵ��KMnO4��ƽ����Ӧ�����ǣ�____________���ú�t1��ʽ�ӱ�ʾ����
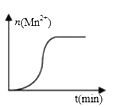
��3�����ij��ʵ�飨���£�ʱ����Һ��Mn2+���ʵ�����ʱ���ϵ��ͼ�������n(Mn2+)�ڷ�Ӧ��ʼʱ�仯����һ��ʱ�����������ԭ��___________________��