��Ŀ����
A��B��C��D��E�� F�����ڱ��ж����ڵ�����Ԫ�أ��й����ʻ�ṹ��Ϣ���±���
| Ԫ�� | �й����ʻ�ṹ��Ϣ |
| A | ����������л���A��һ�ֵ������� |
| B | B������A���ӵ�������ͬ���������������е������Ӱ뾶��С�� |
| C | C��Bͬ���ڣ���������������ԭ�Ӱ뾶���ģ�ϡ��������⣩ |
| D | D������ڻ�ҩ��һ�ֳɷ֣�Ҳ������ɱ������ |
| E | E��Dͬ���ڣ����ڸ�������ԭ�Ӱ뾶��СKs5u |
| F | F���⻯�������������ˮ���ﷴӦ����һ�����ӻ����� |
��1��A��C��ԭ�Ӹ�����Ϊ1��1�γɵĻ�����ĵ���ʽΪ ����0��6 mol�û�����Ͷ�뵽100 mL 3 mol/L BE3��Һ�е����ӷ���ʽΪ �� ��
��2��F���⻯������ �����Ի�Ǽ��ԣ����γɵ� �����Ի�Ǽ��ԣ����ӣ�д��ʵ�����Ʊ����⻯��Ļ�ѧ����ʽ ��
��3����ͼ������ʵ��֤��D��E�ķǽ����Ե�ǿ����
�� ��Һa��b�ֱ�Ϊ �� ��д��ѧʽ����
����Һa�����a��Ӧ�����ӷ���ʽΪ ��
�۷ǽ�����D E������ڻ�С�ڣ������ԭ�ӽṹ�ĽǶȽ���ԭ�� ��
��1�� 2 Na2O2+2H2O��4Na��+4OH��+ O2 ��, Al3��+4OH���� AlO2-+2 H2O
��2�� ���� ���� 2NH4Cl+ Ca(OH)2 CaCl2+2NH3��+2 H2O
��3�� �� HCl H2S �� 2 MnO4-+16 H��+10 Cl-��2Mn2��+5 Cl2��+8 H2O
�� �� D��ԭ�Ӱ뾶����E��ԭ�Ӱ뾶,���Եõ��ӵ�������E����
��������������������֪��AΪO��BΪAl��CΪNa��DΪS��EΪCl��FΪN��A��C��ԭ�Ӹ�����Ϊ1��1�γɵĻ�����ΪNa2O2,��3���з���װ��ͼ��֪����Ӧ��Ϊ������غ�Ũ���ᣬ2 MnO4-+16 H��+10 Cl-��2Mn2��+5 Cl2��+8 H2O���ҷǽ�����Cl>S��
���㣺Ԫ�������ɡ�Ԫ�ػ�����֪ʶ����ѧ����

��ϰ��ϵ�д�
 ��������ܸ�ϰϵ�д�
��������ܸ�ϰϵ�д�
�����Ŀ
���в���ǰ36��Ԫ�ص����ʻ�ԭ�ӽṹ���±���
| Ԫ�ر�� | Ԫ�����ʻ�ԭ�ӽṹ |
| R | ��̬ԭ�ӵ��������3��δ�ɶԵ��ӣ��������2������ |
| S | ��������ˮ���ҷ�Ӧ��������Һ�������� |
| T | ��̬ԭ��3d�������1������ |
| X | 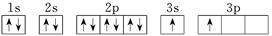 |
��1��RԪ�صĵ�һ������Ҫ������ͬ�������ڵ�Ԫ�أ�ԭ����____________________________________��
Rԭ�ӵĽṹʾ��ͼ�� ��
��2��SԪ�ص�������ϼ�Ϊ________��ԭ����________________________________________��
��3��TԪ��ԭ�ӵļ۵��ӹ���Ϊ________��
��4��X�ĵ����Ų�ͼΥ����____________����X���ʡ�������μ����������εȿ����������ȼ��ʱ�����������ɫ�Ĺ⣬����ԭ�ӽṹ��֪ʶ���ͷ����ԭ��_________________________________��
X��Y��Z��W���ֳ���Ԫ�أ�����X��Y��ZΪ������Ԫ�ء��й���Ϣ���±���
| | ԭ�ӻ���������Ϣ | ���ʼ��仯���������Ϣ |
| X | ZX4�������ɴ�Z�ᴿZ���м���� | X������������Ӧ��ˮ����Ϊ��������ǿ�� |
| Y | Yԭ�ӵ��������������ڵ��Ӳ��� | Y���������ǵ��͵��������������������һ�ּ���ǰ;�ĸ��²��� |
| Z | Zԭ�ӵ������������Ǵ�����������1/2 | Z�����ǽ������ϵ����ǣ��䵥������ȡ���ģ���ɵ�·����Ҫԭ�� |
| W | Wԭ�ӵ�����������С��4 | W�ij������ϼ���+3��+2��WX3ϡ��Һ�ʻ�ɫ |
��1��W�����ڱ���λ��Ϊ ��W��OH��2�ڿ����в��ȶ������ױ��������ɰ�ɫѸ�ٱ�ɻ���ɫ������ɺ��ɫ����Ӧ�Ļ�ѧ����ʽΪ ��
��2��X�ļ������ӵĽṹʾ��ͼΪ ��X������������Ӧˮ�����ˮ��Һ��Y�������ﷴӦ�����ӷ���ʽΪ ��
��3��Z����������ͨѶ���������� ����ҵ���Ʊ�Z�ĵ��ʵĻ�ѧ��Ӧ����ʽΪ ������Z��ͬһ����Ԫ�أ��Ž��з���Ԥ������һԪ�صĴ��ڣ�����������뵼�徧��ܣ������о��������л���������ԵĿ��������ԣ����NaOH ��Һ��Ӧ������H2O2����ʱ����NaOH ��Һ��Ӧ���������Σ��䷽��ʽΪ ��
��4����50 mL l mol��L-1��YX3��Һ����μ���0��5 mol��L-1��NaOH��Һ���õ�1��56 g�����������NaOH��Һ��������� ���������һ�������
A��B��C��D��E���ֶ�����Ԫ�أ�ԭ���������������й���Ϣ���¡�
| Ԫ�� | �й���Ϣ |
| A | ����������Ӧ��ˮ���������⻯�ﷴӦ�������ӻ����� |
| B | �ؿ��к�������Ԫ�� |
| C | �����뱣����ú���� |
| D | ���ʼȿ������ᷴӦ���ֿ���NaOH��Һ��Ӧ |
| E | ԭ�������������ȴ�����������1�� |
��ش��������⣺
��1��A���⻯���ˮ��Һ��ʹ��̪��Һ��죬ԭ���ǣ��õ��뷽��ʽ��ʾ����ʵ������ȡ���⻯��Ļ�ѧ����ʽ�� ��
��2��A��B�����������Ϊ7��16����ԭ�ӷ��ӣ����л�������������ʵ��ŷ��йص��ǣ�����ţ���
������ ������ЧӦ �۳������ƻ� �ܹ⻯ѧ��Ⱦ
��3��C������������ˮ������E���ʷ�Ӧ��������������Һ�������ӷ���ʽ�� ��
��4��D��Ԫ�����ڱ��е�λ���ǡ���Ԫ��A��D���ij������������õ��Ե�ԣ���������ˮ������Ӧ�Ļ�ѧ����ʽ�� ��


 HCNO + NH3������ƽ�������������գ�
HCNO + NH3������ƽ�������������գ� 2N2��g����3H2O��g��
2N2��g����3H2O��g��