��Ŀ����
14��N2O5��һ����������������һ���¶��¿ɷ������з�Ӧ��2N2O5��g��?4NO2��g��+O2��g����H��0��һ���¶��£���10L�����ܱ�������ͨ��N2O5���IJ���ʵ�����ݼ��±�������˵������ȷ���ǣ�������| t/s | 0 | 500 | 1000 | 1500 |
| c��N2O5��/mol | 5.00 | 3.5 | 2.50 | 2.50 |
| A�� | 500 s��N2O5�ֽ�����Ϊ3��10-4mol•��L•s��-1 | |
| B�� | ��Ӧ��ƽ����������м���5molN2O5���壬ƽ�������ƶ�����Ӧ����ЧӦ��� | |
| C�� | ��ͬ�¶��£���ʼʱ�������г���5molNO2��1.25molO2���ﵽƽ��ʱ��NO2��ת����С��50% | |
| D�� | �ﵽƽ�����ͬ�������������г���2.5molN2O5��1.25molO2����ʱƽ�⽫�����ƶ� |
���� A������ͼ�����ݷ�������500s��N2O5��g�����ĵ�Ũ�ȼ���ֽ��ʣ�
B���������м���5molN2O5���壬�൱������ѹǿƽ�����淴Ӧ�����ƶ���
C����ʼʱ�������г���5molNO2��1.25molO2����ȫת��������൱�ڳ���2.5molN2O5��g������Ϊԭ����һ�룬�ݴ˷�����
D������Ũ�Ȼ���ƽ�ⳣ��֮��Ĺ�ϵ�жϣ�
��� �⣺A������ͼ�����ݷ�������500s��N2O5��g�����ĵ�Ũ��=5.00mol/L-3.5mol/L=1.5mol/L���ֽ�����=$\frac{1.5mol/L}{500s}$=3��10-3 mol/��L•s������A��ȷ��
B���������м���5molN2O5���壬�൱������ѹǿƽ�����淴Ӧ�����ƶ������Ƿ�Ӧ����ЧӦ���B����
C����ʼʱ�������г���5molNO2��1.25molO2����ȫת��������൱�ڳ���2.5molN2O5��g������Ϊԭ����һ�룬����ԭ������൱�ڼ�Сѹǿ��ƽ�������ƶ�������NO2��ת����С��50%����C��ȷ��
D���ɱ������ݿ�֪��1000sʱ��Ӧ����ƽ�⣬ƽ��ʱc��N2O5��=0.25mol/L��c��NO2��=0.5mol/L��c��O2��=0.125mol/L��ƽ�ⳣ��K=$\frac{[O{\;}_{2}]•[NO{\;}_{2}]{\;}^{4}}{[N{\;}_{2}O{\;}_{5}]{\;}^{2}}$=$\frac{0.125��0.5{\;}^{4}}{0.25{\;}^{2}}$=0.125����ﵽƽ�����ͬ�������������г���2.5molN2O5��1.25molO2����Ũ�Ȼ�Qc=$\frac{[O{\;}_{2}]•[NO{\;}_{2}]{\;}^{4}}{[N{\;}_{2}O{\;}_{5}]{\;}^{2}}$=$\frac{0.25��0.5{\;}^{4}}{0.5{\;}^{2}}$=0.0625��K������ƽ�⽫�����ƶ�����D��ȷ��
��ѡB��
���� ���⿼�黯ѧƽ�ⷴӦ���ʵļ��㡢��ѧƽ���йؼ��㡢��ѧƽ��Ӱ�����ء�ƽ�ⳣ���ȣ��Ѷ��еȣ�

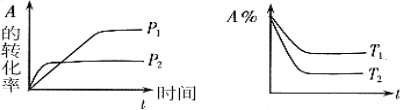
| A�� | ѹǿP1��P2 | B�� | a+b��c+d | C�� | T1��T2 | D�� | ��H��0 |
����NaOH��Na2CO3���Һ�����ȣ�������Сʱ��
�ų�ϴ�ӷ�Һ����ˮ��ϴ��¯������ϡ���ᣬ���ݣ�
����ϴҺ�м���Na2SO3��Һ��
������ϴ��꣬��NaNO2��Һ�ۻ���¯��
��1����NaOH�ܽ��������Ļ�ѧ����ʽ��SiO2+2NaOH�TNa2SiO3+H2O��
��2����֪��20��ʱ�ܽ��/g
| CaCO3 | CaSO4 | Mg��OH��2 | MgCO3 |
| 1.4��10-3 | 2.55��10-2 | 9��10-4 | 1.1��10-2 |
��3���ڲ�����У�
�ٱ�������ˮ���������⣬����CaCO3��Mg��OH��2��SiO2��
����ϴ�����У��ܽ���������ٹ�¯��ʴ�������ӷ���ʽ������ԭ��2Fe3++Fe�T3Fe2+��
��4��������У�����Na2SO3��Ŀ����SO32-+2Fe3++H2O=SO42-+2Fe2++2H+����Fe3+��ԭ��Fe2+����ֹFe3+��ʴ��¯����������ӷ���ʽ�����ֱ��
��5��������У��ۻ���Ĺ�¯����Ḳ��һ�����ܵ�Fe3O4����Ĥ��
����ɲ���ƽ�䷴Ӧ�����ӷ���ʽ��
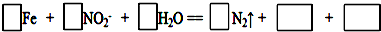
�������16.8g�����ۻ�����ת�Ƶĵ�����Ϊ4.82��1023��������λ��Ч���֣���
| A�� | Cl2 | B�� | HNO3 | C�� | Na2O2 | D�� | H2O2 |
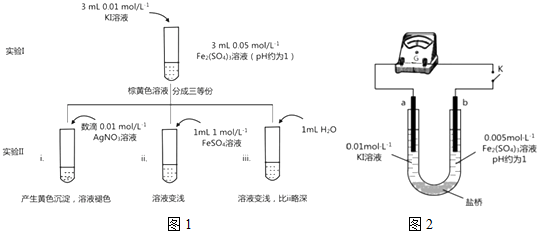
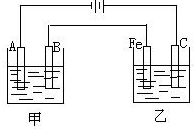 ��ͼ���ס���Ϊ��������������أ���ش��������⣺
��ͼ���ס���Ϊ��������������أ���ش��������⣺