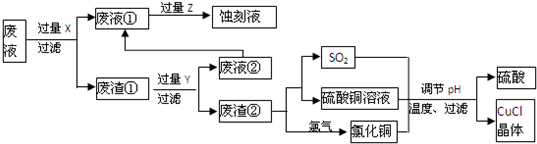
��Ŀ����
16����һƿ�������Һ�����п��ܺ���NH4+��K+��Na+��Mg2+��Ba2+��Al3+��Fe3+��Cl-����-��NO3-��CO32-��SO42- �еļ��֣�ȡ����Һ��������ʵ�飺��1��ȡpH��ֽ���飬��Һ��ǿ���ԣ�
��2��ȡ��������Һ����������CCl4������������ˮ������CCl4����Ϻ�ɫ��
��3����ȡ��������Һ������NaOH��Һ��ʹ��Һ��������Ϊ���ԣ��ڵμӹ����к͵μ���Ϻ���Һ������������
��4��ȡ����������������Һ��Na2CO3��Һ���а�ɫ�������ɣ���
��5����������ʵ����ʵȷ���ٸ���Һ�϶����ڵ�������Ba2+��I-���ڿ϶������ڵ�������Mg2+��Fe3+��Al3+��NO3-��CO32-��SO42-���ۻ�����ȷ����������K+��Na+��NH4+��Cl-����Ҫȷ�����е������ӿ����õķ���ԭ����K+��Na+����ɫ��Ӧ��NH4+��NaOH��Һ�����ȣ�������������ʪ��ĺ�ɫʯ����ֽ���飬������
���� ��1��ȡpH��ֽ���飬��Һ��ǿ���ԣ��������ӷ�Ӧ�����Ӳ��ܹ����棻
��2��ȡ��������Һ����������CCl4������������ˮ������CCl4����Ϻ�ɫ��˵����Һ��һ�����е����ӣ��ܹ�������ӷ�Ӧ�����Ӳ����棻
��3����ȡ��������Һ������NaOH��Һ��ʹ��Һ��������Ϊ���ԣ��ڵμӹ����к͵μ���Ϻ���Һ�������������ݴ��жϷ��������������ӷ�Ӧ���ɳ��������Ӳ����ڣ�
��4��ȡ����������������Һ��Na2CO3��Һ���а�ɫ�������ɣ���֪һ������Ba2+���ų�������ӷ�Ӧ�����ӣ�
��� �⣺��1������ʵ�飨1��������Һ��ǿ���ԣ�˵����Һ�п϶�����H+����H+��CO32-��Ӧ������Ӧ�����ܹ��棬˵����Һ�п϶�������CO32-��
��2������ʵ�飨2������CCl4����Ϻ�ɫ��˵����I2����������I-�����������������ģ��Ӷ�˵����Һ�к���I-����I-��Fe3+��NO3-��H+�ܷ���������ԭ��Ӧ�������ܹ��棬˵����Һ�п϶�������Fe3+��NO3-��
��3������ʵ�飨3��������Һ��������Ϊ���ԣ��ڵμӹ����к͵μ���Ϻ���Һ��������������Fe3+��Mg2+��Al3+����Ӧ����������˵����Һ�п϶�������Fe3+��Mg2+��Al3+��
��4������ʵ�飨4������ȡ����������������Һ��Na2CO3��Һ���а�ɫ�������ɣ�˵����Һ�п϶�����Ba2+����Ba2+����SO42-����������˵����Һ�в���SO42-��
��5��������ʵ����ʵȷ��������Һ�п϶����ڵ������ǣ�Ba2+��I-���϶������ڵ������ǣ�Mg2+��Fe3+��Al3+��NO3-��CO32-��SO42-��
������ȷ���Ƿ���ڵ������ǣ�K+��Na+��NH4+��Cl-������ K+��Na+����ɫ��Ӧ���飬����ɫ��ɫ��������ɫ���ܲ�������ɫ��NH4+��NaOH��Һ�����ȣ�������������ʪ��ĺ�ɫʯ����ֽ���飬������˵����笠����Ӵ��ڣ�
�ʴ�Ϊ��Ba2+��I-��Mg2+��Fe3+��Al3+��NO3-��CO32-��SO42-��K+��Na+��NH4+��Cl-�� K+��Na+����ɫ��Ӧ��NH4+��NaOH��Һ�����ȣ�������������ʪ��ĺ�ɫʯ����ֽ���飬������
���� ���⿼�����ʵļ��鼰����Ϊ��Ƶ���㣬���ճ�������֮��ķ�Ӧ�����Ӽ����Ϊ�ƶϵĹؼ������ط������ƶ��������ۺϿ��飬��Ŀ�Ѷ��еȣ�

 ѧ���쳵�����ּ��������ҵ�½����������ϵ�д�
ѧ���쳵�����ּ��������ҵ�½����������ϵ�д� �����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д�
�����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д� Сѧ�����ҵ���ϴ�ѧ������ϵ�д�
Сѧ�����ҵ���ϴ�ѧ������ϵ�д� ���Ž�����ٰθ��νӹ㶫���������ϵ�д�
���Ž�����ٰθ��νӹ㶫���������ϵ�д� �����������ҵ�������������ϵ�д�
�����������ҵ�������������ϵ�д�| A�� | ȡ����Ӧ | B�� | ������Ӧ | C�� | ��ԭ��Ӧ | D�� | �ӳɷ�Ӧ |
| ѡ�� | �� | �� |
| A | SO2��Ư���� | SO2��ʹ��ˮ��ɫ |
| B | SiO2�е����� | SiO2�������Ʊ����ά |
| C | Fe3+�������� | ����30%��FeCl3��Һ��ʴͭ������ӡˢ��·�� |
| D | C12�о綾 | ��������й©ʱ�����ռ���Һ��ʪ�������棬��Ѹ���뿪�ֳ� |
| A�� | A | B�� | B | C�� | C | D�� | D |
 �±���ijѧ��Ϊ̽��AgCl����ת��ΪAg2S�����ķ�Ӧ����ʵ��ļ�¼��
�±���ijѧ��Ϊ̽��AgCl����ת��ΪAg2S�����ķ�Ӧ����ʵ��ļ�¼��| �� �� | �� �� |
| ��ȡ5mL 0.1mol/L AgNO3��һ�����0.1mol/L NaCl��Һ����ϣ��� | ����������ɫ���� |
| ������������Һ�м���2.5mL 0.1mol/L Na2S��Һ�� | ����Ѹ�ٱ�Ϊ��ɫ |
| ��������ɫ��Һ�������ڿ����У����Ͻ��裮 | �ϳ�ʱ�������Ϊ���ɫ |
| �����˳����е����ɫ��������������HNO3��Һ�� | ��������ɫ���壬���������ܽ� |
| �������˵õ���ҺX�Ͱ�ɫ����Y����X�еμ�Ba��NO3��2��Һ�� | ������ɫ���� |
��2����֪��25��ʱKsp��AgCl��=1.8��10-10��Ksp��Ag2S��=6��10-30���˳���ת����Ӧ��ƽ�ⳣ��K=5.4��109��
��3������V�в����İ�ɫ�����Ļ�ѧʽΪBaSO4������������ɫ����������AgCl�⣬������S��
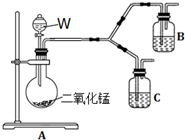
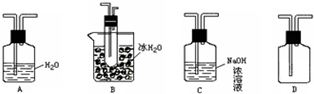
��4��Ϊ�˽�һ��ȷ�ϲ���������ɫ����������ԭ�����������ͼ��ʾ�ĶԱ�ʵ��װ�ã�
��װ��A�в���������Բ����ƿ�����ܺͷ�Һ©�����Լ�WΪ����������Һ��
��װ��C�е��Լ�ΪNaCl��Һ��Ag2S����Һ�Ļ���B���Լ�ΪAg2S����Һ��
��ʵ�������C�г�����Ϊ���ɫ��B��û�����Ա仯��
���C�з�Ӧ�Ļ�ѧ����ʽ��
��Ag2S+��NaCl+��O2+��H2O?��AgCl+��S+��NaOH
C��NaCl�������ǣ�������Ag2S������Sʱ��Ag+������NaCl������������������ӽ������AgCl������ʹc��Ag+����С��������������ԭ��Ӧ��ƽ�����ƣ�
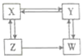 X��Y��Z��WΪ����ȩ���ᡢ���е�һ�֣�����֮�������ͼ��ʾ��ת����ϵ�������ж���ȷ���ǣ�������
X��Y��Z��WΪ����ȩ���ᡢ���е�һ�֣�����֮�������ͼ��ʾ��ת����ϵ�������ж���ȷ���ǣ�������| A�� | X�����ᣬY���� | B�� | Y��ȩ��W�Ǵ� | C�� | Z��ȩ��W������ | D�� | X�Ǵ���Z���� |
 CH3COOC2H5+H2O��
CH3COOC2H5+H2O��
 Ԫ�ؼ��仯�����ѧϰ��Ӧ������ѧ��ѧ����Ҫ����֮һ��
Ԫ�ؼ��仯�����ѧϰ��Ӧ������ѧ��ѧ����Ҫ����֮һ��