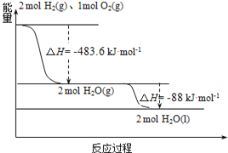
��Ŀ����
����Ŀ��M�ǽ����������̵�ĸ߷����л��������ṹ��ʽΪ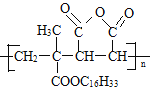 ����ʯ���ѽ����ϳ�M��·��ͼ��ͼ��
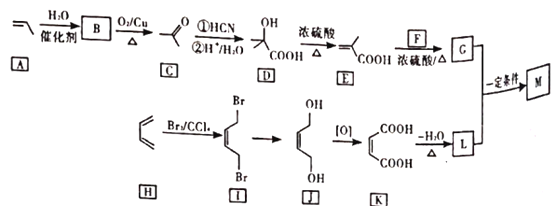
����ʯ���ѽ����ϳ�M��·��ͼ��ͼ��
�ش��������⣺
��1��B�Ļ�ѧ����Ϊ______��
��2��F�ķ���ʽΪ_____��
��3��G�й����ŵ�������_____��G��L��һ������������M�ķ�Ӧ����Ϊ____��
��4��I��J�ķ�Ӧ����ʽΪ_____��
��5��д��2�ַ���������C��ͬ���칹��Ľṹ��ʽ____�����˴Ź�������Ϊ����壬�������Ϊ3:2:1����֪˫�����ǻ�����ʱ���ʲ��ȶ���
��6������ɱ���ϩΪ��ʼԭ���Ʊ�![]() �ĺϳ�·�ߣ����Լ���ѡ��____��
�ĺϳ�·�ߣ����Լ���ѡ��____��
���𰸡�2-����������� C16H34O ̼̼˫�������� �Ӿ۷�Ӧ ![]() +2NaOH
+2NaOH![]()
![]() +2NaBr CH3CH2CHO��
+2NaBr CH3CH2CHO��![]()
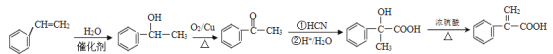
��������
��ת����ϵ��֪������![]() ���ṹ��ʽΪCH3-CH=CH2����H2O��һ�������·����ӳɷ�Ӧ������B��
���ṹ��ʽΪCH3-CH=CH2����H2O��һ�������·����ӳɷ�Ӧ������B��![]() ��
�� ![]() ��������������
��������������![]() ���������Ȼ�����
���������Ȼ�����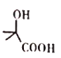 ���÷�����Ũ���������·�����ȥ��Ӧ����
���÷�����Ũ���������·�����ȥ��Ӧ����![]() ��������ϳɷ�������֪��M
��������ϳɷ�������֪��M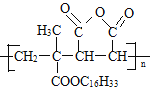 ����G(
����G(![]() )��L(
)��L(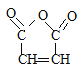 )�����Ӿ۷�Ӧ��Ӧ�����ĸ߾��
)�����Ӿ۷�Ӧ��Ӧ�����ĸ߾��![]() ����E(
����E(![]() )��F����������Ӧ�γɵģ�����F�Ǵ��������ʽΪC16H34O��HΪ1,4-����ϩ����������Ȼ�̼��Һ�����ӳɷ�Ӧ����
)��F����������Ӧ�γɵģ�����F�Ǵ��������ʽΪC16H34O��HΪ1,4-����ϩ����������Ȼ�̼��Һ�����ӳɷ�Ӧ����![]() ��֮���������NaOH��ˮ��Һ�ڼ��������·���ˮ�ⷴӦ(��ȡ����Ӧ)����
��֮���������NaOH��ˮ��Һ�ڼ��������·���ˮ�ⷴӦ(��ȡ����Ӧ)����![]() ��
��![]() ���м��������õ�
���м��������õ�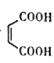 �������ˮ�õ���L��
�������ˮ�õ���L�� ���ݴ˷�������
���ݴ˷�������
(1)����ͼʾ��֪A��CH3-CH=CH2����������H2O��һ�������·����ӳɷ�Ӧ����![]() ������Ϊ2-�������������
������Ϊ2-�������������
(2)M ����G(
����G(![]() )��L(
)��L( )�����Ӿ۷�Ӧ��Ӧ�����ĸ߾��
)�����Ӿ۷�Ӧ��Ӧ�����ĸ߾��![]() ����E(
����E(![]() )��F����������Ӧ�γɵģ�����F�Ǵ���F�ķ���ʽΪC16H34O��
)��F����������Ӧ�γɵģ�����F�Ǵ���F�ķ���ʽΪC16H34O��
(3)G�Ľṹ��ʽ��![]() �����й����ŵ�������̼̼˫����������G��L��һ������������M�ķ�Ӧ�����ǼӾ۷�Ӧ���ʴ�Ϊ��̼̼˫�����������Ӿ۷�Ӧ��
�����й����ŵ�������̼̼˫����������G��L��һ������������M�ķ�Ӧ�����ǼӾ۷�Ӧ���ʴ�Ϊ��̼̼˫�����������Ӿ۷�Ӧ��
(4)I��![]() ����������NaOH��ˮ��Һ�ڼ��������·���ˮ�ⷴӦ(��ȡ����Ӧ)����
����������NaOH��ˮ��Һ�ڼ��������·���ˮ�ⷴӦ(��ȡ����Ӧ)����![]() ������I��J�ķ�Ӧ����ʽΪ
������I��J�ķ�Ӧ����ʽΪ![]() +2NaOH
+2NaOH![]()
![]() +2NaBr��
+2NaBr��
(5)��C�ṹ��ʽ��֪C�ķ���ʽ��C3H6O������ͬ���칹���У��˴Ź�������Ϊ����壬�������Ϊ3�U2�U1�Ľṹ��ʽ��CH3CH2CHO ��![]() ��
��
(6) ![]() �к��й�����Ϊ̼̼˫�����Ȼ����Աȱ���ϩ��
�к��й�����Ϊ̼̼˫�����Ȼ����Աȱ���ϩ��![]() �Ľṹ��ʽ����������һ��̼ԭ�ӣ�ģ��������A��B��C��D��ʵ��̼�ɹǼܵĹ����������Ȼ��������ȥ���ǻ����ɣ��ɱ���ϩΪ��ʼԭ���Ʊ�
�Ľṹ��ʽ����������һ��̼ԭ�ӣ�ģ��������A��B��C��D��ʵ��̼�ɹǼܵĹ����������Ȼ��������ȥ���ǻ����ɣ��ɱ���ϩΪ��ʼԭ���Ʊ�![]() �ĺϳ�·��Ϊ
�ĺϳ�·��Ϊ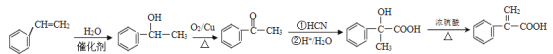 ��
��