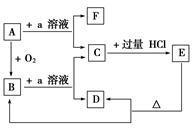
��Ŀ����
������Ҫ�İ뵼����ϣ��������ִ����ӹ�ҵ�Ļ������ش��������⣺
��1����̬Siԭ���У�����ռ�ݵ�����ܲ���� �����ܲ���е�ԭ�ӹ����Ϊ ��������Ϊ ��
��2������Ҫ�Թ����Ρ� �Ȼ��������ʽ�����ڵؿ��С�
��3�����ʹ��������ʯ�ṹ���Ƶľ��壬����ԭ����ԭ��֮���� ���ϣ��侧���й���8��ԭ�ӣ�����������λ�ù��� ��ԭ�ӡ�
��4�����ʹ��ͨ������(SiH4)�ֽⷴӦ���Ʊ�����ҵ�ϲ���Mg2Si��NH4CI��Һ�������з�Ӧ�Ƶ�SiH4���÷�Ӧ�Ļ�ѧ����ʽΪ ��
��5��̼����йػ�ѧ������������ʾ����Ҫ�����ͽ��������й���ʵ��
| ��ѧ�� | C-C | C-H | C-O | Si-Si | Si-H | Si-O |
| ���ܣ�KJ/mol�� | 356 | 413 | 336 | 226 | 318 | 452 |
��SiH4���ȶ���С��CH4���������������ԭ���� ��
��6���ڹ������У�SiO44�������壨����ͼa��ͨ�����ö��������ӿ��γɵ�״����״����״���Ǽ���״�Ĵ���ṹ��ʽ��ͼbΪһ�����������ṹ�Ķ�����������Siԭ�ӵ��ӻ���ʽΪ ��Si��O��ԭ����֮��Ϊ ��ѧʽΪ ��
��1��M��9��4 ��2���������裻 ��3�����ۼ���3
��4��Mg2Si��4NH4Cl=SiH4��4NH3��2MgCl2
��5���ٹ����е�Si��Si����Si��H���ļ���С������������C��C����C��H���ļ��ܣ��ȶ��Բ���ѣ����³������������γɣ����Թ���������������϶�Զ���������ࡣ
�����ڼ���Խ������Խ�ȶ���C��H���ļ��ܴ���C��O���ļ��ܣ���C��H����C��O���ȶ�����Si��H���ļ���ȴԶС��Si��O���ļ��ܣ�����Si��H�����ȶ������������γ��ȶ��Ը�ǿ��Si��O�������������������
��6��sp3��1:3��[SiO3]n2n��(��SiO32��)
����

����ͭ���Ʊ�Cu��Zn��Alϵ��������Ҫԭ�ϡ�����������ȡ����ͭ��ʵ�鷽���ɹ�ѡ�ã���ͭ��ϡ���ᷴӦ��ȡ��3Cu + 8HNO3��ϡ��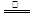 3Cu��NO3��2 + 2NO��+ 4H2O
3Cu��NO3��2 + 2NO��+ 4H2O
��ͭ��Ũ���ᷴӦ��ȡ��Cu + 4HNO3��Ũ��= Cu��NO3��2 + 2NO2��+ 2H2O
�����Ƚ�ͭм�ڿ����м�����������ͭ������ͭ��ϡ���ᷴӦ��ȡ��2Cu + O2 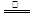 2CuO��CuO + 2HNO3 = Cu(NO3)2 + H2O �����й�˵����ȷ���ǣ� ��
2CuO��CuO + 2HNO3 = Cu(NO3)2 + H2O �����й�˵����ȷ���ǣ� ��
| A����ȡ����������ͭ����������������� |
| B����ȡ����������ͭ���ڲ������ж�����Ȣ��� |
| C�����ַ����У��ڢ۷�����û��� |
| D�����ַ����ķ�Ӧ�����������������н��� |
����ѧ��ѡ��2����ѧ�뼼����(15��)
��ʯ��Ҫ������ơ�Ca3(PO4)2��H2O������ʯ��Ca3(OH)(PO4)3������ʽ���ڡ�ͼ(a)ΪĿǰ��������ʯ���õĴ������������ʪ��������ָ��ʯ�ù�������ֽ��Ʊ����ᡣͼ(b)���ȷ�������������������ʯ�Ƶ��������̡�

�������ʵ�����������£�
| | �۵�/�� | �е�/�� | ��ע |
| ���� | 44 | 280.5 | |
| PH3 | -133.8 | -87.8 | ������ˮ�����л�ԭ�� |
| SiF4 | -90 | -86 | ��ˮ�� |
��1����������ʯ����Ҫ����;�����������ϣ�Լռ��ʯʹ������ �G��
��2������ʯΪԭ�ϣ�ʪ�����������Ca3F(PO4)3��Ӧ�Ļ�ѧ����ʽΪ�� ������1���ۺϺ�������������Լ30%����ʯ�������Ƶ�85�G����Ʒ���� �֡�
��3����ͼ(b)��ʾ���ȷ���������ĵ�һ���ǽ��������衢������̿����ʯ��ϣ����·�Ӧ���ɰ��ס�¯������Ҫ�ɷ��ǣ� (�ѧʽ)������1����Ҫ�������ǣ� ������2����Ҫ�������ǣ�
��4��β������Ҫ���� ������������PH3��H2S��HF�ȣ���β����ͨ�봿����Һ���ɳ�ȥ
��ͨ�����������Һ���ɳ�ȥ (���ѧʽ)
��5�������ʪ�����ᣬ�ȷ����Ṥ�ո��ӣ��ܺĸߣ����ŵ��ǣ� ��
��������������Ⱦ��Ϊ���أ����������������ü�����⡣
(1)���������һ�������ȼҵ��Ʒ������������������ķ������������£�
(��)����SO2�ķ���ͨ���ⱥ��ʳ��ˮ�����õ�����Һ�У���NaHSO3��Һ��
(��)����ⱥ��ʳ��ˮ�������巴Ӧ���Ƶ����ᡣ
(��)���������NaHSO3��Һ�У���Ӧ���õ���SO2������գ����ɵ�NaClѭ�����á�
��д������(��)��Ӧ�Ļ�ѧ����ʽ�� ��
��д������(��)�е�ⱥ��ʳ��ˮ�Ļ�ѧ����ʽ�� ��
��д������(��)��Ӧ�����ӷ���ʽ�� ��
(2)����ѧ���������Fe2����Fe3�������ӵĴ����ã������½�SO2������SO42-��ʵ��SO2�Ļ������á�ij�о���ѧϰС��ݴ���������·�������ʵ���������²ⶨת������SO2������SO42-��ת���ʡ�
�ٸ�С�������ͼװ����ʵ���Ҳⶨģ��������SO2�����������X��Һ������ ��(��д���)
| A����ĵ�����Һ | B�����Ը��������Һ |
| C������������Һ | D���Ȼ�����Һ |
 ��ת���ʣ���֪�������٣�����ⶨ�������� �� ��
��ת���ʣ���֪�������٣�����ⶨ�������� �� ��