��Ŀ����
8��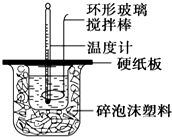 ijʵ��С�������50mL 1.0mol/L�����50mL 1.1mol/L ����������Һ����ͼװ���н����кͷ�Ӧ���ڴ��ձ��ײ�������ĭ���ϣ���ֽ������ʹ�����С�ձ���������ձ�������ƽ��Ȼ�����ڴ�С�ձ�֮����������ĭ���ϣ���ֽ���������ձ�������ĭ���ϰ壨��Ӳֽ�壩���ǰ壬�ڰ��м俪����С�ף�����ʹ�¶ȼƺͻ��β��������ͨ����ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��Իش��������⣺
ijʵ��С�������50mL 1.0mol/L�����50mL 1.1mol/L ����������Һ����ͼװ���н����кͷ�Ӧ���ڴ��ձ��ײ�������ĭ���ϣ���ֽ������ʹ�����С�ձ���������ձ�������ƽ��Ȼ�����ڴ�С�ձ�֮����������ĭ���ϣ���ֽ���������ձ�������ĭ���ϰ壨��Ӳֽ�壩���ǰ壬�ڰ��м俪����С�ף�����ʹ�¶ȼƺͻ��β��������ͨ����ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��Իش��������⣺��1����ʵ�������Թ�����NaOH��ԭ���DZ�֤������ȫ���кͣ��ڴ�С�ձ��������ĭ���ϵ������DZ��¸��ȣ���ֹ����ɢʧ��
��2����ʵ��С����������ʵ�飬ÿ��ȡ��Һ��50mL������¼��ԭʼ���ݣ����±�����
| ʵ����� | ��ʼ�¶�t1/�� | ��ֹ�¶ȣ�t2��/�� | �²� ��t2-t1��/�� | ||
| ���� | NaOH��Һ | ƽ��ֵ | |||
| 1 | 25.1 | 24.9 | 25.0 | 31.6 | 6.6 |
| 2 | 25.1 | 25.1 | 25.1 | 31.8 | 6.7 |
| 3 | 25.1 | 25.1 | 25.1 | 31.9 | 6.8 |
��3�����õ�Ũ�ȵĴ�����NaOH��Һ��Ӧ�����õ��к��Ȼ�ȣ�2������ġ�Hƫ���ƫ����ƫС�����䡱������ԭ���Ǵ��������ᣬ��Ӧʱ��Ҫ�����������ڴ���ĵ��룬���ԣ���õ��кͷ�Ӧ�ķ�Ӧ����ֵ��ƫС���÷�ӦΪ���ȷ�Ӧ����H��ƫ��
��4�����к��Ȳⶨʵ���д�����ˮϴ���¶ȼ��ϵ�����������¶ȼƲⶨNaOH��Һ�¶ȵIJ��裬���˲������裬���õ��к��ȡ�H��ƫ���ƫ����ƫС�����䡱����
���� ��1����ʵ�������Թ�����NaOH��ԭ���DZ�֤������ȫ���кͣ��к��Ȳⶨʵ��ɰܵĹؼ��DZ��¹�����
��2����������η�Ӧ���¶Ȳ���ݹ�ʽQ=cm��T���������0.05mol��ˮ�ų��������Ӷ�����к��ȣ������к��ȵĸ���д�����кͷ�Ӧ���Ȼ�ѧ����ʽ��
��3���ô���������ᣬ�������Ҫ������������õ�����ƫС��
��4��û����ˮϴ���¶ȼ��ϵ�������Һ�����²ⶨNaOH��Һ�¶�ƫ�ߣ��¶Ȳ�ƫС����õ�����ƫС��
��� �⣺��1���ù������������Ʊ�֤���ᷴӦ��ȫ�����������Ϊ����ȷ���㣻�к��Ȳⶨʵ��ɰܵĹؼ��DZ��¹������ڴ�С�ձ��������ĭ���ϵ������Ǽ���ʵ������е�������ʧ��
�ʴ�Ϊ����֤������ȫ���кͣ����¸��ȣ���ֹ����ɢʧ��
��2��3��ʵ���ǰ���¶Ȳ�ֱ�Ϊ��6.6�棬6.7�棬6.8�棬����Ч�����ε�ƽ��ֵΪ6.7�棬50mL 1.0mol/L�����50mL 1.1mol/L ����������Һ��������Ϊm=100mL��1g/mL=100g��c=4.18J/��g•�棩�����빫ʽQ=cm��T������0.05mol��ˮ�ų�����Q=4.18J/��g•�棩��100g��6.7��=28.006KJ��������0.5mol��ˮ�ų�����28.006KJ����������1mol��ˮ�ų�����Ϊ28.006KJ��2=56.01kJ������ʵ���õ��к��ȡ�H=-56.0kJ/mol���Ȼ�ѧ����ʽΪH+��aq��+OH-��aq��=H2O��l����H=-56.0 kJ/mol��
�ʴ�Ϊ��H+��aq��+OH-��aq��=H2O��l����H=-56.0 kJ/mol��
��3���ô���������ᣬ�������Ҫ������������ɷų�������ƫС����õ��кͷ�Ӧ�ķ�Ӧ����ֵ��ƫС���÷�ӦΪ���ȷ�Ӧ����H��ƫ��
�ʴ�Ϊ��ƫ���������ᣬ��Ӧʱ��Ҫ�����������ڴ���ĵ��룬���ԣ���õ��кͷ�Ӧ�ķ�Ӧ����ֵ��ƫС���÷�ӦΪ���ȷ�Ӧ����H��ƫ��
��4��û����ˮϴ���¶ȼ��ϵ�������Һ�����²ⶨNaOH��Һ�¶�ƫ�ߣ��¶Ȳ�ƫС����õ�����ƫС����Ϊ�к��ȡ�HΪ��ֵ�������к��ȡ�Hƫ�ʴ�Ϊ��ƫ��
���� ���⿼���к��ȵIJⶨ�����ڿ��鿼���Ķ��ֲ��������ͼ����������ѶȲ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | 22.4L�κ����壬�����ʵ�����Ϊ1mol | |
| B�� | �DZ�״���£�l mol�κ��������һ������22.4L | |
| C�� | 0.6mol H2��0.2mo1 O2��0.2mol CO2��ɵĻ�������ڱ�������Լ22.4L | |
| D�� | ����Ħ�����ָ�����ڱ�״����1mol�������� |
| A�� | 1869�������ѧ���Ž��з���Ԫ�������� | |
| B�� | 1803��Ӣ����ѧ�ҵ����ٽ���ԭ��ѧ˵ | |
| C�� | 1771�귨����ѧ����������������ѧ˵ | |
| D�� | 1661��Ӣ����ѧ�Ҳ��������˻�ѧԪ�صĸ��� |
| A�� | ��CaCl2��Һ��ͨ��CO2��Ca2++CO2+H2O�TCaCO3��+2H+ | |
| B�� | Ư����Һ��ͨ�����SO2��ClO��+H2O+SO2�THSO3-+HClO | |
| C�� | ��FeSO4��Һ�м���H2O2��Һ��Fe2++2H2O2+4H+�TFe3++4H2O | |
| D�� | �ڳ���ʯ��ˮ�м�������С�մ���Һ��Ca2++OH-+HCO3-�TCaCO3��+H2O |
| A�� | 2.4g����þ���þ����ʱʧȥ�ĵ�����Ϊ0.1NA | |
| B�� | ��״���£�11.2LCCl4�����ķ�����Ϊ0.5NA | |
| C�� | 0.5mol/LAlCl3��Һ��Cl-����ĿΪ2NA | |
| D�� | 17g�����к��еĵ�����Ϊ10NA |
| A�� | 6mol | B�� | 5mol | C�� | 4mol | D�� | 3mol |
| A�� | ����ˮ��ͨ��Cl2����ɱ��������������HClO������ | |
| B�� | ��������������ˮ���������Ƴ���ʯ��ˮ��Һ | |
| C�� | ��ˮ��Һ�ȡ������ijɷ���ͬ��Cl2��Cl-�Ļ�ѧ����Ҳ��ͬ | |
| D�� | ���Ƶ���ˮ����ɫ������Ȼ��Ư�ס�ɱ���Ĺ��� |
 ��
��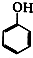 +NaOH��
+NaOH�� +H2O��
+H2O�� ��
�� $\stackrel{����KMnO_{4}��Һ}{��}$CO2��
$\stackrel{����KMnO_{4}��Һ}{��}$CO2��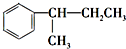 $\stackrel{����KMnO_{4}��Һ}{��}$
$\stackrel{����KMnO_{4}��Һ}{��}$ ��
��