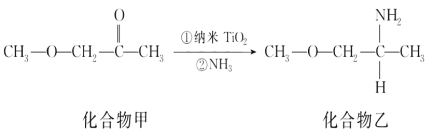
��Ŀ����
����Ŀ��ij�о���ѧϰС���һ��δ֪�ɷֵĺϽ���Ͻ��������´�����
��ش��������⣺
![]() ��������ʵ�����̣�ȷ���úϽ��п϶����еijɷ��� ______
��������ʵ�����̣�ȷ���úϽ��п϶����еijɷ��� ______ ![]() �ѧʽ
�ѧʽ![]() ��
��
![]() �������õ��IJ��������� ______ ��
�������õ��IJ��������� ______ ��
![]() ������D��ȡ������һ����ɫ��ѧ���գ��Լ�b��һ�ֳ��õ�Һ̬���������仯ѧʽΪ ______ ��д���ù�����
������D��ȡ������һ����ɫ��ѧ���գ��Լ�b��һ�ֳ��õ�Һ̬���������仯ѧʽΪ ______ ��д���ù�����![]() �����Ļ�ѧ����ʽ ______ ��
�����Ļ�ѧ����ʽ ______ ��
![]() �ס�����λͬѧ����֤�˷�Ӧ�����ɵ���������������ͬѧ��Ϊ�Ͻ���Ӧ����������ͬѧ�������ϵ�֪��Ҳ����NaOH��Һ��Ӧ����
�ס�����λͬѧ����֤�˷�Ӧ�����ɵ���������������ͬѧ��Ϊ�Ͻ���Ӧ����������ͬѧ�������ϵ�֪��Ҳ����NaOH��Һ��Ӧ����![]() ��
��![]() �������Ϊ�Ͻ��п��ܺ��裬Ҳ����ͬʱ�������衣����Ʒ���֤���Ͻ����Ƿ�������
�������Ϊ�Ͻ��п��ܺ��裬Ҳ����ͬʱ�������衣����Ʒ���֤���Ͻ����Ƿ�������![]() д��ѡ���Լ����۲쵽��������
д��ѡ���Լ����۲쵽��������![]() �� ______ ��
�� ______ ��
![]() ��ҺC����Ԫ�صĺ�������������
��ҺC����Ԫ�صĺ�������������![]() ��Һ�ζ����ⶨ�������ӷ���ʽ��ʾ�õζ����ķ�Ӧԭ���� ______ ��
��Һ�ζ����ⶨ�������ӷ���ʽ��ʾ�õζ����ķ�Ӧԭ���� ______ ��
���𰸡�Fe��Cu ©�����ձ��������� ![]()
![]() ȡ������ҺA���Թ��У���εμ�ϡ����
ȡ������ҺA���Թ��У���εμ�ϡ����![]() �����������
�����������![]() ���������������ɰ�ɫ������������������ȫ�ܽ⣬��Ͻ��к������������裻�����������ܽ⣬��Ͻ���ͬʱ�������裻������ʼ�ղ��ܣ���Ͻ��к��й裬������
���������������ɰ�ɫ������������������ȫ�ܽ⣬��Ͻ��к������������裻�����������ܽ⣬��Ͻ���ͬʱ�������裻������ʼ�ղ��ܣ���Ͻ��к��й裬������ ![]()
��������
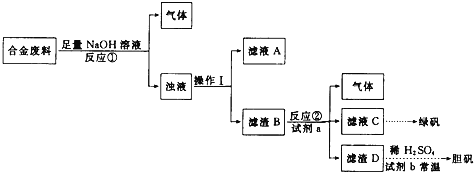
�Ͻ���ϼ�����������������Һ�õ����壬�������õ��ľ����֪һ����������ͭ��ͬʱ�������������Ʒ�Ӧ���������Ӧ����ΪAl��Si�ȣ���Һ�к�������ͭ�����˺�����B����ϡ�������ܽ�������ҺCΪ����������Һ������DΪͭ����ϡ������Һ�м���������⣬����ͭ��Ӧ��������ͭ��Һ��ͨ������Ũ������ȴ�ᾧ������ϴ�ӵõ�����ͭ���壬�ݴ˽���⡣
��1�����������Ϣ����������е��ܽ��������õ������õ�����˵������B�к��н���ͭ��֤���Ͻ��к���ͭ���̷��к�������������֤���Ͻ��к�������
�ʴ�Ϊ��Fe��Cu��
��2��������Ϊ���ˣ������õ��IJ���������©�����ձ�����������
�ʴ�Ϊ��©�����ձ�����������
��3��˫��ˮ��һ����ɫ������������ʵ�ֽ���ͭ������֮��ķ�Ӧ���Լ�b��һ�ֳ��õ�Һ̬������������bΪ![]() ��ͭ
��ͭ![]() �����Ļ�ѧ����ʽΪ
�����Ļ�ѧ����ʽΪ![]() ��
��
�ʴ�Ϊ��![]() ��
��![]() ��
��
��4���Ͻ������������裬������ҺA�к���ƫ�����ƺ����ƣ�����������������ǿ����Һ��������������ʵ����֤�����ʵ��֤���Ͻ����Ƿ��������
����Ϊȡ������ҺA���Թ��У���εμ�ϡ����(�����������)���������������ɰ�ɫ������������������ȫ�ܽ⣬��Ͻ��к������������裻�����������ܽ⣬��Ͻ���ͬʱ�������裻������ʼ�ղ��ܣ���Ͻ��к��й裬��������
�ʴ�Ϊ��ȡ������ҺA���Թ��У���εμ�ϡ����(�����������)���������������ɰ�ɫ������������������ȫ�ܽ⣬��Ͻ��к������������裻�����������ܽ⣬��Ͻ���ͬʱ�������裻������ʼ�ղ��ܣ���Ͻ��к��й裬��������
��5�����������ܱ�����������������������ӣ������������ԭ�������ӣ���Ӧ�����ӷ���ʽΪ![]() ��
��
�ʴ�Ϊ��![]() ��
��

����Ŀ������±��ش�������������Ϊ�����µ���������
�� | ���볣��(Ka) | �� | ���볣��(Ka) | �� | ���볣��(Ka) | �� | ���볣��(Ka) |
CH3COOH | 1.8��10-5 | H2CO3 | K1=4.4��10-7 K2=4.7��10-11 | H2C2O 4 | K1=5.4��10-2 K2=5.4��10-5 | H2S | K1=1.3��10-7 K2=7.1��10-15 |
HClO | 3��10-8 |
��ش��������⣺
��1��ͬŨ�ȵ�CH3COO-��HCO3-��CO32-��HC2O4-��ClO-��S2-�н��H+��������������__________��
��2��������0.1 molL-1��CH3COOH��Һ�ڼ�ˮϡ�����У����б���ʽ������һ����С����_______�����������
A.c(H+) B. c(H+)/c(CH3COOH) C. c(H+)/c(OH-) D. c(OH-)
��3��0.1 molL-1��H2C2O4��Һ��0.1 molL-1��KOH����Һ�������Ϻ�������Һ�����ԣ�����Һ�и�����Ũ���ɴ�С��˳��Ϊ______________��
��4��pH��ͬ��NaClO��CH3COOK��Һ������Һ�����ʵ���Ũ�ȵĴ�С��ϵ�ǣ�CH3COONa______NaClO������Һ�У�[c(Na+)-c(ClO-)]______[c(K+)-c(CH3COO-)]���������������=������
��5����0.1 molL-1CH3COOH ��Һ�еμ� NaOH ��Һ�� c(CH3COOH): c(CH3COO-)=5 : 9����ʱ��ҺpH=_______________��
����Ŀ��ijͬѧ�������ȷ�Ӧ��������������������������Ӧ�ų��������ȣ�������ҫ�۵Ĺ�â������ֽ©�����²����մ���������������ɳ���������ġ���ѧ�ֲᡷ֪��Al��Al2O3��Fe��Fe2O3�۵㡢�е��������£�
���� | Al | Al2O3 | Fe | Fe2O3 |
�۵�/�� | 660 | 2054 | 1535 | 1462 |
�е�/�� | 2467 | 2980 | 2750 | -- |
I.��ͬѧ�Ʋ⣬���ȷ�Ӧ���õ���������Ӧ�������Ͻ������ǣ��÷�Ӧ�ų�������ʹ���ۻ����������۵�����ͣ���ʱҺ̬���������ۺ��γ������Ͻ��������һ����ʵ�鷽����֤���������õĿ�״�������к��н���������ʵ�������Լ���_______����Ӧ�����ӷ���ʽΪ____________________________��
��. ʵ���о����֣����ᷢ��������ԭ��Ӧʱ�������Ũ��Խϡ����Ӧ��ԭ�����е�Ԫ�صĻ��ϼ�Խ�͡�ijͬѧȡһ������������������һ������ϡ�������ַ�Ӧ����Ӧ������������ų����ڷ�Ӧ���������Һ�У���μ���4mol��L��1������������Һ����������������Һ�����(mL)������ij��������ʵ���(mol)�Ĺ�ϵ��ͼ��ʾ���Իش��������⣺
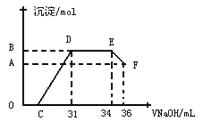
��1��ͼ��OC��û�г������ɣ��˽η�����Ӧ������Ϊ��___________________��
��2����DE�Σ����������ʵ���û�б仯����˽η�����Ӧ�����ӷ�Ϊ_________��
��3����c=13mLʱ��ԭ��Һ��Fe3+��Al3+�����ʵ���֮��Ϊ________________________��
����Ŀ��������Ϊһ���ִ���ͨ�������ڽ���ǧ��������β���к���CO��NOx���ж����壬����Ⱦ����Ҳ��Ϊ������ἱ���������⡣
��1����������������ʱ������N2��O2��Ӧ���������仯ʾ��ͼ��ͼ��

��÷�Ӧ���Ȼ�ѧ����ʽΪ_____________________________��
��2����������װβ������װ�ã���ʹCO��NOx�ж��������Ӧת��Ϊ�����塣
2xCO��2NOx ="=" 2xCO2��N2����ת�Ƶ������ʵ���Ϊ0.4x molʱ���÷�Ӧ���ɱ�״���µ�N2���_____________________L��
��3��һ����̼��һ����;�൱�㷺�Ļ�������ԭ�ϡ����Ի�ԭ��������������������ϳɺܶ��л�����״��ȡ���ѹǿΪ0.1 MPa�����£���a mol CO��3a mol H2�Ļ�������ڴ��������·�Ӧ���ɼ״��� CO(g)��2H2(g)![]() CH3OH(g)����H�� -b kJmol-1
CH3OH(g)����H�� -b kJmol-1
���÷�Ӧ��ƽ�ⳣ������ʽΪ_____________________________��
���������ݻ����䣬���д�ʩ�����Ӽ״����ʵ���______________
A�������¶� | B����CH3OH(g)����ϵ�з��� |
C������He��ʹ��ϵ��ѹǿ���� | D���ٳ���1 mol CO��3 mol H2 |
�����ⶨ��ͬ�¶��¸÷�Ӧ��ƽ�ⳣ�����£�
�¶ȣ����� | 250 | 300 | 350 |
K | 2.041 | 0.270 | 0.012 |
��ijʱ�̡�250����ø÷�Ӧ�ķ�Ӧ�����������Ũ��Ϊc(CO) ��0.4 mol/L��c(H2)��0.4 mol/L��c(CH3OH)��0.8 mol��L-1, ���ʱv��___________v��(�������)��
��4���״�����Ҫ�Ļ�������ԭ�ϣ�����һ�����͵�ȼ�ϣ��������������һ��ȼ�ϵ�أ�һ���缫ͨ���������һ���缫����״�����صĵ�����Dz�����Y2O3��ZrO2���壬���ڸ������ܴ���O2-���ӡ��õ�ص�������ӦʽΪ_____________________________����ع���ʱ�������������O2-��_______���ƶ���