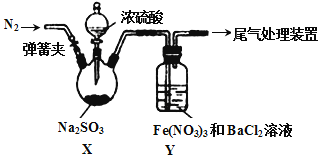
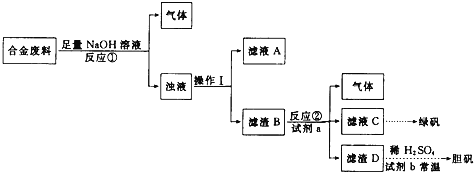
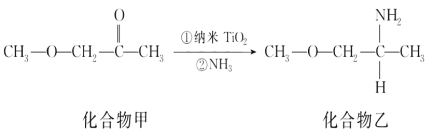
��Ŀ����
����Ŀ��C��N��Ti��Mn��Cu������Ҫ�IJ���Ԫ�أ��䵥�ʼ�����������������ж��й㷺��Ӧ�á�
��1��Mnλ��Ԫ�����ڱ���_________����Mn2���ļ۲�����Ų�ͼΪ_____��
��2��Nԭ�Ӻ�����______�ֿռ��˶�״̬��ͬ�ĵ��ӡ�NO2+ �����幹����_________��������Ϊ�ȵ�����ķ�����________��д��һ�֣���
��3��Cu2����NH3���γ�[Cu(NH3)4]2�������ӣ�0.5 mol [Cu(NH3)4]2���к��������ĸ���Ϊ_____����֪NF3��NH3������ͬ�Ŀռ乹�ͣ���NF3������Cu2���γ������ӣ���ԭ����___________________________________________________________��
��4������TiO2��һ��Ӧ�ù㷺�Ĵ����������һ��ʵ����ͼ��ʾ�����������̼ԭ�ӵ��ӻ���ʽΪ___________����������ԭ�ӵĵ�һ�������ɴ�С��˳��Ϊ___________���������ҵķе����Ը��ڻ�����ף���Ҫԭ����______��
���𰸡�d  5 ֱ���� CO2����N2O��CS2�������𰸺������ɣ� 8NA ����8��6.02��1023��4.816��1024�� F �ĵ縺�Ա� N ��N��F �ɼ����Ӷ�ƫ�� F������ NF3 �е�ԭ�Ӻ˶���µ��ӶԵ�����������ǿ�����ڸ����µ��Ӷ��γ���λ���� sp3 ��sp2�ӻ� N>O>C >H �������ҷ��Ӽ�������
5 ֱ���� CO2����N2O��CS2�������𰸺������ɣ� 8NA ����8��6.02��1023��4.816��1024�� F �ĵ縺�Ա� N ��N��F �ɼ����Ӷ�ƫ�� F������ NF3 �е�ԭ�Ӻ˶���µ��ӶԵ�����������ǿ�����ڸ����µ��Ӷ��γ���λ���� sp3 ��sp2�ӻ� N>O>C >H �������ҷ��Ӽ�������
��������
��1��MnΪ��25��Ԫ�أ�λ�ڵ�������VIIB�壻 Mn2���ļ۲�����Ų�ʽΪ3d5��
��2��Nԭ�Ӻ����Ų�ʽΪ1s22s22p3�����ݼ۲���ӻ�������ȷ���ռ乹�ͣ��ȵ�����ķ�����CO2��N2O��CS2�ȣ�
��3��[Cu(NH3)4]2�������������У��������к���3��������Cu2����NH3���γ�4����������ϼ�16��������NF3��NH3������ͬ�Ŀռ乹�ͣ�Fԭ�ӱ�H�ĵ縺��ǿ����N��F��Ĺ��õ��ӶԵ�������ǿ�����µ�ԭ�Ӻ˶���µ��ӶԵ�����������ǿ�����ڸ����µ��Ӷ��γ���λ����
��4�������к���1���Ҽ���˫���к���1���Ҽ���1���м���ͬһ����Ԫ�أ�Ԫ�ص�һ����������ԭ��������������������ƣ�����IIA�塢��VA��Ԫ�ص�һ�����ܴ���������Ԫ�أ�����Ĵ��ڵ��������۷е����ߡ�
��1��MnΪ��25��Ԫ�أ�λ�ڵ�������VIIB�壬����d����Mn�ļ۵����Ų�ʽΪ3d54s2����Mn2���ļ۲�����Ų�ʽΪ3d5���Ų�ͼΪ![]() ��
��
��2��Nԭ�Ӻ����Ų�ʽΪ1s22s22p3����5�ֿռ��˶�״̬��NO2+ ������Nԭ�ӵŵ��Ӷ���=![]() ��a-bx��=
��a-bx��=![]() ��5-1-2��2��=0����2����������ռ乹��Ϊֱ���ͣ��ȵ�����ķ�����CO2��N2O��CS2�ȣ�
��5-1-2��2��=0����2����������ռ乹��Ϊֱ���ͣ��ȵ�����ķ�����CO2��N2O��CS2�ȣ�
��3��[Cu(NH3)4]2�������������У��������к���3��������Cu2����NH3���γ�4����������ϼ�16����������0.5 molʱ����8mol��������8NA��NF3��NH3������ͬ�Ŀռ乹�ͣ�Fԭ�ӱ�H�ĵ縺��ǿ����N��F��Ĺ��õ��ӶԵ�������ǿ�����µ�ԭ�Ӻ˶���µ��ӶԵ�����������ǿ�����ڸ����µ��Ӷ��γ���λ����
��4����������У�CH2��CH3�ṹ��CΪsp3�ӻ���![]() �ṹ��CΪsp2�ӻ������к���C��H��O��N����Ԫ�أ���һ�������ɴ�С��˳��ΪN>O>C >H����������Ӽ䲻���γ���������������е�Nԭ�ӿ��Ժ���һ�����γ���������·��Ӽ�����������е����ߡ�
�ṹ��CΪsp2�ӻ������к���C��H��O��N����Ԫ�أ���һ�������ɴ�С��˳��ΪN>O>C >H����������Ӽ䲻���γ���������������е�Nԭ�ӿ��Ժ���һ�����γ���������·��Ӽ�����������е����ߡ�

����Ŀ��п�̸ɵ��������ʹ�õĻ�ѧ��أ������������ͼ1��ʾ��

![]()

��1��п�̸ɵ�صĵ�·��ÿͨ��![]() �������ϸ����������� ______ g������ʱ
�������ϸ����������� ______ g������ʱ![]() �����������ŵ�����������壬����һ����������Ǻ�
�����������ŵ�����������壬����һ����������Ǻ�![]() �����������ĵ缫��Ӧʽ�� ______
�����������ĵ缫��Ӧʽ�� ______
��2��ij�о�С��Ե���ں�ɫ��״�����������ʵ�飺
��֪��![]() ��������������������ʵ�鱨�棺
��������������������ʵ�鱨�棺
ʵ�鲽�� | ʵ������ | ʵ����ۺͽ��� |
ȡ����������ɫ��Һ�������У���μ���NaOH��Һ��ֱ���������ټ��� | ���ɰ�ɫ������ ______ | ��ɫ��Һ�д��� ���� |
��3�����ò����з������![]() ���о�����
�������![]() ��
��![]() �����е����ã�ʵ��װ����ͼ2��ʾ���������ʵ���Ũ�ȡ������
�����е����ã�ʵ��װ����ͼ2��ʾ���������ʵ���Ũ�ȡ������![]() ��Һ������ƿ�У��ֱ����2��ʵ��
��Һ������ƿ�У��ֱ����2��ʵ��![]() ������������ͬ�����²ⶨ
������������ͬ�����²ⶨ![]() ��
��
��� | ��ƿ�е����� | ʵ���¼ | ʵ���������� |
ʵ��һ | ���� | �ռ���56mL���� |
|
ʵ��� | ���� | ��ɫ��ĩ�����ܽ⣬�ռ���112mL���� |
|
ʵ��1��2�вμӷ�Ӧ��![]() �����ʵ���֮��Ϊ ______ ��ʵ����з�Ӧ�����ӷ���ʽΪ ______��
�����ʵ���֮��Ϊ ______ ��ʵ����з�Ӧ�����ӷ���ʽΪ ______��
����Ŀ��ú�����г����о���ͬ�¶���ƽ�ⳣ����Ͷ�ϱȼ����ʵ����⡣��֪��CO(g)+H2O(g)![]() CO2(g)+H2(g)��ƽ�ⳣ�����¶ȵı仯���±�������������ȷ����
CO2(g)+H2(g)��ƽ�ⳣ�����¶ȵı仯���±�������������ȷ����
�¶�/�� | 400 | 500 | 830 | 1000 |
ƽ�ⳣ��K | 10 | 9 | 1 | 0.6 |
A. �÷�Ӧ������Ӧ�����ȷ�Ӧ
B. �÷�Ӧ�ﵽƽ����������ݻ����������¶ȣ�����Ӧ���ʲ��䣬�����ڻ�������ѹǿ����
C. 830��ʱ���ں��ݷ�Ӧ���а����ʵ�����n(CO):n(H2O)��n(H2):n(CO2)=2:4:6:1Ͷ�뷴Ӧ������������Ӧ����ʼʱ��v��<v��
D. 830��ʱ����2L���ܱ������м���4molCO(g)��6molH2O(g)�ﵽƽ��ʱ��CO��ת������60%