��Ŀ����
�弰�仯����㷺Ӧ�����л��ϳɡ���ѧ����������
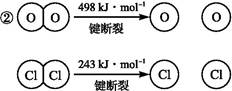
��1����ˮ�����������Ԫ�صı仯���£�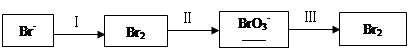
�ٹ��̢�ˮ�Լ��ԣ�����pH��3.5����ͨ��������
��.ͨ��������Ӧ�����ӷ���ʽ��______��
��.����ˮpH�����Cl2�������ʣ���ƽ��ԭ��������ԭ����______��
�ڹ��̢����ȿ�������ϳ�������Ũ̼������Һ���ա���ɲ���ƽ���з���ʽ��
Br2�� Na2CO3��
Na2CO3�� NaBrO3��
NaBrO3�� CO2��
CO2�� ______
______
�۹��̢��������ữ�ɵ�Br2��Na2SO4�Ļ����Һ��
��ͬ�����£����������ữ������������������٣�ԭ����______��
��2��NaBrO3��һ�ַ����Լ����������ữ��NaI��Һ����μ���NaBrO3��Һ��������2.6 mol NaBrO3ʱ����÷�Ӧ����Һ����͵�Ĵ�����ʽ�����ʵ����ֱ�Ϊ��
| ���� | I2 | Br2 | IO3- |
| ���ʵ���/mol | 0.5 | 1.3 | |
��1���٢�.Cl2��2Br-��Br2��2Cl-
��.Cl2��H2O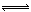 H+��Cl-��HClO�� ����c(H+)��ƽ�������ƶ�������Cl2��ˮ��Ӧ
H+��Cl-��HClO�� ����c(H+)��ƽ�������ƶ�������Cl2��ˮ��Ӧ
��3 3 1 3 5 NaBr
�������л�ԭ�ԣ����������Ӧ
��2��3
���������������1���٢�.ͨ��������Cl2��Br?��Ӧ��Cl2��2Br-��Br2��2Cl-
��.Cl2��ˮ�ķ�ӦΪ���淴Ӧ��Cl2��H2O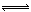 H+��Cl-��HClO��������Һ��pH������c(H+)��ƽ�������ƶ�������Cl2��ˮ��Ӧ��
H+��Cl-��HClO��������Һ��pH������c(H+)��ƽ�������ƶ�������Cl2��ˮ��Ӧ��
�ڸ��ݻ��ϼ۵ı仯��Ԫ���غ����֪������ΪNaBr���л��ϼ�����������ƽ��ѧ����ʽ��
��HCl��ClԪ��Ϊ-1�ۣ�Ϊ��ͼ�̬������������л�ԭ�ԣ��ܱ������������������������١�
��2������������ԭ��Ӧ�е�ʧ��������ȣ��ɵ�5��2.6mol=2��0.5mol+6��n(IO3?)����n(IO3?)=2mol������Ԫ���غ㣬ԭ��Һ��n(NaI)=0.5mol��2+2mol=3mol��
���㣺���⿼�����ӷ���ʽ����д����ѧƽ���ƶ�����ѧ����ʽ����ƽ�����ݵ����غ�ļ��㡣

 ��ѧȫ��������ѵ��ϵ�д�
��ѧȫ��������ѵ��ϵ�д�ij��ҵ��ˮ�����±��е�ijЩ���ӣ��Ҹ������ӵ����ʵ���Ũ����ȣ���Ϊ0.1 mol/L(����ֵ����ˮ�ĵ��뼰���ӵ�ˮ��)��
| ������ | K����Ag����Mg2����Cu2����Al3����NH4+ |
| ������ | Cl����CO32����NO3����SO42����I�� |
��ͬѧ��̽����ˮ����ɣ�����������ʵ�飺
��.ȡ����ɫ��Һ5 mL���μ�һ�ΰ�ˮ�г������ɣ��������������ӡ�
��.�ò�˿պȡ��Һ���ڻ��������գ�����ɫ�ܲ����۲죬����ɫ���档
��.��ȡ��Һ����������ᣬ����ɫ�������ɣ�����ɫ������������ɺ���ɫ��
��.��������õ���Һ�м���BaCl2��Һ���а�ɫ�������ɡ�
���ƶϣ�
(1)�ɢ��жϣ���Һ��һ�������е���������___________________________________��
(2)���м�������������ɫ��������ӷ���ʽ��________________________��
(3)��ͬѧ����ȷ��ԭ��Һ��������������________����������________�����ݴ��Ʋ�ԭ��ҺӦ�ó�________�ԣ�ԭ����____________________________(�������ӷ���ʽ˵��)��
(4)��ȡ100 mLԭ��Һ������������NaOH��Һ���˹������漰�����ӷ���ʽΪ____________________________����ַ�Ӧ����ˣ�ϴ�ӣ����ճ��������أ��õ��Ĺ�������Ϊ________g��
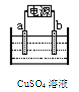
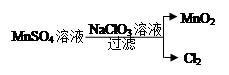
 2Cl2+2H2O,��ʵ���ȵ�ѭ�����á�
2Cl2+2H2O,��ʵ���ȵ�ѭ�����á�