��Ŀ����
A��B��C��D��E���ֶ�����Ԫ�أ�A��Dͬ���ڣ�A�ĵ��ʼȿ������ᷴӦ���ֿ���NaOH��Һ��Ӧ��B�ĵ����ڷŵ�����������������Ӧ��CԪ�ص����Ӳ������ӣ�DԪ��ԭ�ӵ��������������������������3/4��EԪ��ԭ�ӵ��������������������������3����
��1��A��ԭ�ӽṹʾ��ͼΪ ��
��2��0.1 mol��L A����������Һ��0.1 mol��L NaOH��Һ�������ϣ���Ӧ������
����ʽΪ ��
��3����A�ĵ��ʺ�NiO(OH)Ϊ�缫��NaOH��ҺΪ���Һ���һ�����͵�أ��ŵ�ʱNiO(OH)ת��ΪNi(OH)2���õ���ܷ�Ӧ�Ļ�ѧ����ʽ�� ��
��4�����������Ԫ��A��B��ɣ��������õ��Ե�ԡ�
�����������ˮ������Ӧ���ɺ�B�Ļ������ң��ҷ����к���10�����ӡ�д����
��Ӧ�Ļ�ѧ����ʽ�� ��
��ҵ��A�ĵ��ʺͻ��������ڸ���1700K��Ӧ�Ʊ��ס���֪�÷�Ӧ������Ϊ����
����Ӧ���÷�Ӧ�Ļ�ѧ����ʽ�� ��
��5��D��E����Ԫ����Ƚϣ��ǽ����Խ�ǿ����(��Ԫ������) ��������֤�ý��۵���(��д���) ��
a.�Ƚ�������Ԫ�صij������ʵķе�
b.�Ƚ�������Ԫ�صĵ������������ϵ�����
c.�Ƚ�������Ԫ�ص���̬�⻯����ȶ���
��6��C��D��E����γɱ��������ַ��ӣ����Ǿ�����18�����ӣ�����붡��Ӧ����D���ʵĻ�ѧ����ʽΪ ��
��1��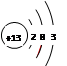 ��1�֣�
��1�֣�
��2��Al3+ + 3OH-�� Al(OH)3����2�֣�
��3��Al+3NiO(OH)+H2O+NaOH==NaAlO2+3Ni(OH)2��2�֣�
��4��AlN + 3H2O �� Al(OH)3��+ NH3����2�֣� 2Al + 2NH3 2AlN + 3H2��2�֣�
2AlN + 3H2��2�֣�
��5������1�֣�bc ��2�֣� ��6��H2S+H2O2=S��+2H2O��2�֣�
�������������A��B��C��D��EԪ�طֱ�ΪAl��N��H��S��O����2������������������������������������3��Ni���ϼ���+3�۱�Ϊ+2�ۣ������ϼ������Խ���������ƫ�����Σ��ʷ���ʽΪ Al+3NiO(OH)+H2O+NaOH==NaAlO2+3Ni(OH)2����4����ΪAlN��ˮ��ΪAl(OH)3��NH3����5���ǽ�����ǿ������ͨ�����������ϵ��������⻯����ȶ��ԡ�����������Ӧˮ��������ǿ���жϣ���6��H��S��O����Ԫ���γɵ�18���ӷ���ΪH2S��H2O2�������ܷ���������ԭ��Ӧ��
���㣺�������ʽṹ�й����⡣

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�X��Y��Z��W��Ԫ�����ڱ���ԭ������������������ֶ�����Ԫ�أ��������Ϣ���±���
| Ԫ�� | �����Ϣ |
| X | X������������Ӧ��ˮ���ﻯѧʽΪH2XO3 |
| Y | Y�ǵؿ��к�����ߵ�Ԫ�� |
| Z | Z�Ļ�̬ԭ�����������Ų�ʽΪ3s23p1 |
| W | W��һ�ֺ��ص�������Ϊ28��������Ϊ14 |
(1)Wλ��Ԫ�����ڱ���________���ڵ�________�壻W��ԭ�Ӱ뾶��X��________(���С��)��
(2)Z�ĵ�һ�����ܱ�W��________(���С��)��XY2�ɹ�̬��Ϊ��̬����˷���������������________����Ԫ�ء�X��Y��ԭ�ӿɹ�ͬ�γɶ��ַ��ӣ�д������һ�����γ�ͬ�ַ��Ӽ��������������________��
(3)���£���Z���������ᷴӦ�����ɫ��Һ�еμ�NaOH��Һֱ���������ܹ۲쵽��������________��W�ĵ���������ᷴӦ����������ɫ���壬�÷�Ӧ�Ļ�ѧ����ʽ��_________________________________________________��
���в���ǰ36��Ԫ�ص����ʻ�ԭ�ӽṹ���±���
| Ԫ�ر�� | Ԫ�����ʻ�ԭ�ӽṹ |
| R | ��̬ԭ�ӵ��������3��δ�ɶԵ��ӣ��������2������ |
| S | ��������ˮ���ҷ�Ӧ��������Һ�������� |
| T | ��̬ԭ��3d�������1������ |
| X | 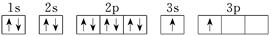 |
��1��RԪ�صĵ�һ������Ҫ������ͬ�������ڵ�Ԫ�أ�ԭ����____________________________________��
Rԭ�ӵĽṹʾ��ͼ�� ��
��2��SԪ�ص�������ϼ�Ϊ________��ԭ����________________________________________��
��3��TԪ��ԭ�ӵļ۵��ӹ���Ϊ________��
��4��X�ĵ����Ų�ͼΥ����____________����X���ʡ�������μ����������εȿ����������ȼ��ʱ�����������ɫ�Ĺ⣬����ԭ�ӽṹ��֪ʶ���ͷ����ԭ��_________________________________��
a��b��c��d��e��f��gΪ�����ɶ�������Ԫ�ع��ɵ����ӣ����Ƕ���10�����ӣ���ṹ�ص����£�
| ���Ӵ��� | a | b | c | d | e | f | g |
| ԭ�Ӻ��� | ���� | ���� | ˫�� | ��� | ���� | ��� | ��� |
| ���������λ��ɣ� | 0 | 1+ | 1- | 0 | 2+ | 1+ | 0 |
����b�����Ӱ뾶����e�����Ӱ뾶��d���ɼ��Լ����ɵ���ԭ�ӷ��ӣ�c��f��Ӧ���γ�����g���ӡ���д����
��1��a���ӵĺ�������Ų�ʽ______________________________��
��2��b��e��ӦԪ�ص�����������Ӧˮ����ļ���ǿ��Ϊ��________��________ (�û�ѧʽ��ʾ)��
��3��d����ˮ�ĵ��뷽��ʽ__________________________________________��
��4��c������__________��f������__________���û�ѧʽ��ʾ����
��5��g���ӵĹ��ۼ�����_____�����Ǹ��࣬˵�����ۼ����� �ԡ�
X��Y��Z��W���ֳ���Ԫ�أ�����X��Y��ZΪ������Ԫ�ء��й���Ϣ���±���
| | ԭ�ӻ���������Ϣ | ���ʼ��仯���������Ϣ |
| X | ZX4�������ɴ�Z�ᴿZ���м���� | X������������Ӧ��ˮ����Ϊ��������ǿ�� |
| Y | Yԭ�ӵ��������������ڵ��Ӳ��� | Y���������ǵ��͵��������������������һ�ּ���ǰ;�ĸ��²��� |
| Z | Zԭ�ӵ������������Ǵ�����������1/2 | Z�����ǽ������ϵ����ǣ��䵥������ȡ���ģ���ɵ�·����Ҫԭ�� |
| W | Wԭ�ӵ�����������С��4 | W�ij������ϼ���+3��+2��WX3ϡ��Һ�ʻ�ɫ |
��1��W�����ڱ���λ��Ϊ ��W��OH��2�ڿ����в��ȶ������ױ��������ɰ�ɫѸ�ٱ�ɻ���ɫ������ɺ��ɫ����Ӧ�Ļ�ѧ����ʽΪ ��
��2��X�ļ������ӵĽṹʾ��ͼΪ ��X������������Ӧˮ�����ˮ��Һ��Y�������ﷴӦ�����ӷ���ʽΪ ��
��3��Z����������ͨѶ���������� ����ҵ���Ʊ�Z�ĵ��ʵĻ�ѧ��Ӧ����ʽΪ ������Z��ͬһ����Ԫ�أ��Ž��з���Ԥ������һԪ�صĴ��ڣ�����������뵼�徧��ܣ������о��������л���������ԵĿ��������ԣ����NaOH ��Һ��Ӧ������H2O2����ʱ����NaOH ��Һ��Ӧ���������Σ��䷽��ʽΪ ��
��4����50 mL l mol��L-1��YX3��Һ����μ���0��5 mol��L-1��NaOH��Һ���õ�1��56 g�����������NaOH��Һ��������� ���������һ�������
X��Y��Z��W���ֳ���Ԫ�أ�����X��Y��ZΪ������Ԫ�ء��й���Ϣ���±���
| | ԭ�ӻ���������Ϣ | ���ʼ��仯���������Ϣ |
| X | ZX4�������ɴ�Z�ᴿZ���м���� | X������������Ӧ��ˮ����Ϊ��������ǿ�� |
| Y | Yԭ�ӵ��������������ڵ��Ӳ��� | Y���������ǵ��͵��������������������һ�ּ���ǰ;�ĸ��²��� |
| Z | Zԭ�ӵ������������Ǵ�����������1/2 | Z�����ǽ������ϵ����ǣ��䵥������ȡ���ģ���ɵ�·����Ҫԭ�� |
| W | Wԭ�ӵ�����������С��4 | W�ij������ϼ���+3��+2��WX3ϡ��Һ�ʻ�ɫ |
��2��X�ļ������ӵĽṹʾ��ͼΪ ��X������������Ӧˮ�����ˮ��Һ��Y�������ﷴӦ�����ӷ���ʽΪ ��
��3��Z����������ͨѶ���������� ����ҵ���Ʊ�Z�ĵ��ʵĻ�ѧ��Ӧ����ʽΪ ������Z��ͬһ����Ԫ�أ��Ž��з���Ԥ������һԪ�صĴ��ڣ�����������뵼�徧��ܣ������о��������л���������ԵĿ��������ԣ����NaOH ��Һ��Ӧ������H2O2����ʱ����NaOH ��Һ��Ӧ���������Σ��䷽��ʽΪ ��
��4����50 mL l mol·L-1��YX3��Һ����μ���0.5 mol·L-1��NaOH��Һ���õ�1.56 g�����������NaOH��Һ��������Ϊ mL��
X��Y��Z��T��W����Ԫ�ص����ʻ�ԭ�ӽṹ���±���
| Ԫ�� | Ԫ�����ʻ�ԭ�ӽṹ |
| X | ԭ�ӵ������������Ǵ�����������2�� |
| Y | �����µ���Ϊ˫ԭ�ӷ��ӣ����⻯��ˮ��Һ�ʼ��� |
| Z | ��̬ԭ�����������Ų�ʽΪ��n+1��sn(n+1)pn2 |
| T | ��Zͬ���ڣ�Ԫ�������+7�� |
| W | ԭ������ΪY��TԪ��֮�ͣ�������к��и�Ԫ�� |
(1)Ԫ��X��һ��ͬλ�ؿɲⶨ�������������ͬλ�صķ�����______��WԪ�ػ�̬ԭ�ӵ����Ų�ʽ
Ϊ______________��
(2)Ԫ��Z��Ԫ��T��ȣ��ǽ����Խ�ǿ����_______(��Ԫ�ط��ű�ʾ�������б�������֤����һ��ʵ����___________________��
A.������Z�ĵ��ʺ�T�ĵ���״̬��ͬ
B. T���⻯���Z���⻯���ȶ�
C.һ��������Z��T�ĵ��ʶ���������������Һ��Ӧ
D. T�ĵ縺�Ա�Z��
(3)�����ܼ�XZ2�ķ����У����еĦҼ���м�������Ϊ______�������γɵľ�������Ϊ______�� Y�ij����⻯����Һ������Ҫԭ����________________________��
(4)Ԫ��X���Ԫ�ؿ����γ�һ�����ӻ�����û�����������Ӻ�CO��Ϊ�ȵ����壬�û�����ĵ���ʽΪ ������Ԫ��X���ӻ������� ��
 HCNO + NH3������ƽ�������������գ�
HCNO + NH3������ƽ�������������գ� 2N2��g����3H2O��g��
2N2��g����3H2O��g��