��Ŀ����
18����ˮAlCl3�������л��ϳɵĴ�����ʳƷ���ɼ��ȣ���ҵ������������Ҫ�ɷ���A12O3��Fe2O3����ʯ�ͽ�����Ҫ�ɷ���C������ͼ��ʾ���̽���һϵ�з�Ӧ���Ʊ���ˮAlCl3��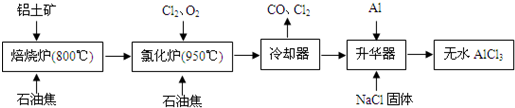
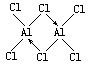
��1���Ȼ����ڼ�������������������̬�Ȼ����Ļ�ѧʽΪAl2Cl6��ÿ��Ԫ�ص�ԭ���������ﵽ8�����ȶ��ṹ����AlCl3�Ƿ��Ӿ��壬��ṹʽΪ
 ��
����2���Ȼ�¯��Al2O3��Cl2��C��Ӧ�Ļ�ѧ����ʽ��A12O3+3C12+3C$\frac{\underline{\;����\;}}{\;}$ 2A1C13+3CO��
��3����ȴ���ų���β���к��д���CO������Cl2������Na2SO3��Һ��ȥCl2���˷�Ӧ�����ӷ���ʽΪ��SO32-+C12+H2O�TSO42-+2C1-+2H+��
��4������������Ҫ����AlCl3��FeCl3�����������Al���������ǣ���ȥFeCl3��
��5��AlCl3��Ʒ��FeԪ�غ���ֱ��Ӱ����Ʒ�ʣ�Ϊ�ⶨ��Ʒ��FeԪ�صĺ������ֳ�ȡ16.25g��ˮAlCl3��Ʒ�����ڹ�����NaOH��Һ�����˳�����������ᆳϴ�ӡ����ա���ȴ�����ز�����������Ϊ0.32g�����Ʒ��FeԪ�صĺ���Ϊ��96%��
���� ���̷�����֪��������Ҫ�ɷ���A12O3��Fe2O3����ʯ�ͽ��ڱ���¯�и��¼��ȷ�Ӧ���õ���������Ȼ�¯�и��·�Ӧ�����Ȼ�����һ����̼����ȴ��õ��Ȼ������壬�������м�������ԭ�Ȼ����������Ȼ�������������ʹ�Ȼ�������������ȴ�õ������Ȼ�����
��1�����ݹ���������������Ʊ���ˮAlCl3��˵��AlCl3�е�ܵͣ�ӦΪ���Ӿ��壻
��2�����ݹ������̿�֪�������к���AlCl3�ȣ�����Al2O3��C12��C��Ӧ������A1C13����������β����֪��������CO��
��3��Cl2��ǿ�����ԣ���SO32-����ΪSO42-����������ԭΪ2C1-��
��4������������Ҫ����AlCl3��FeCl3��FeCl3�۵㡢�е�ϵͣ������Ʊ���ˮAlCl3��Ӧ��ȥFeCl3��
��5���Ʊ���ˮAlCl3��������FeCl3��������������Ϊ0.32gΪFe2O3��������ԭ���غ����FeCl3���������������AlCl3���������ٸ��ݲ�Ʒ�Ĵ��ȶ�����㣮
��� �⣺��1�����ݹ���������������Ʊ���ˮAlCl3��˵��AlCl3�е�ܵͣ�ӦΪ���Ӿ��壬�ṹʽΪ�� ���ʴ�Ϊ�����ӣ�
���ʴ�Ϊ�����ӣ� ��
��
��2�����ݹ������̿�֪�Ȼ�¯�IJ������ȴ���������Ʊ���ˮAlCl3��˵���Ȼ�¯�IJ����к���A1C13��������β������CO������Al2O3��C12��C��Ӧ������A1C13��CO����Ӧ����ʽΪ
A12O3+3C12+3C$\frac{\underline{\;����\;}}{\;}$2A1C13+3CO���ʴ�Ϊ��A12O3+3C12+3C$\frac{\underline{\;����\;}}{\;}$2A1C13+3CO��
��3��Cl2��ǿ�����ԣ���SO32-����ΪSO42-����������ԭΪ2C1-����Ӧ���ӷ���ʽΪSO32-+C12+H2O�TSO42-+2C1-+2H+���ʴ�Ϊ��SO32-+C12+H2O�TSO42-+2C1-+2H+��
��4������������Ҫ����AlCl3��FeCl3��FeCl3�۵㡢�е�ϵͣ������Ʊ���ˮAlCl3��Ӧ��FeCl3ȥ�����Լ�������AlĿ���dz�ȥFeCl3��
�ʴ�Ϊ����ȥFeCl3��
��5��������ԭ���غ㣬�������FeCl3����Ϊmg����
Fe2O3����������2FeCl3
160 325
0.32g mg
���� $\frac{0.32g}{160}$=$\frac{mg}{325}$����ã�m=0.65g��
����AlCl3��Ʒ�Ĵ���Ϊ$\frac{16.25g-0.65g}{16.25g}$��100%=96%��
�ʴ�Ϊ��96%��
���� ���⿼���˿������Լ��洢��֪ʶ�����зֽ⡢Ǩ��ת�������飬ʹ����õ������Ӧ�����������ֳ���ѧ����ʵ�ģ��ܹ�ͨ��������������Ϳ�ѧʵ���л�ѧ�����Լ�������ݺ�ͼ�εȵĹ۲죬��ȡ�йصĸ���֪ʶ��ӡ�����÷������Ƚϡ����ɵȷ���������ȡ����Ϣ���г����ӹ���Ӧ�õ�������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | I2 | B�� | Br2 | C�� | Cl2 | D�� | F2 |
| ��ѧ����ʽ | K ��t1�� | K ��t2�� |
| F2+H2?2HF | 1.8��1036 | 1.9��1032 |
| Cl2+H2?2HCl | 9.7��1012 | 4.2��1011 |
| Br2+H2?2HBr | 5.6��107 | 9.3��106 |
| I2+H2?2HI | 43 | 34 |
��2��HX�ĵ���ʽ��
 ��
����3�����ۼ��ļ����湲�õ��Ӷ�ƫ�Ƴ̶ȵ��������ǿ��HX���ۼ��ļ�����ǿ������˳����HF��HCl��HBr��HI��
��4��������K�ı仯�������ƶϳ�������±��ԭ�Ӻ˵���������ӣ�ad��
a������ͬ�����£�ƽ��ʱX2��ת��������
b��X2��H2��Ӧ�ľ��ҳ̶�����
c��HX�Ļ�ԭ������
d��HX���ȶ���������
| A�� | ���³�ѹ�£�11.2 L Cl2�����е�ԭ����ĿΪNA | |
| B�� | 9�� H2O�����е���ԭ����ĿΪNA | |
| C�� | ��ͬ��ͬѹʱ��1mol�κ�������������ͬ�Ҿ�ԼΪ22.4L | |
| D�� | 0.1 molCH4���Ӻ��е�ԭ����ĿΪ NA |
| A�� | NaOH+HCl�TNaCl+H2O | B�� | MnO2+4HCl��Ũ���TMnCl2+2H2O+Cl2�� | ||
| C�� | Fe+2HCl�TFeCl2+H2�� | D�� | CaCO3+2HCl�TCaCl2+H2O+CO2�� |
| A�� | ���ԣ�HClO4��H2SO4��H3PO4 | B�� | ԭ�Ӱ뾶��Cl��S��O | ||
| C�� | �ȶ��ԣ�HBr��HCl��HF | D�� | �����ԣ�Al��Mg��Na |