��Ŀ����
A��B��C��D��E����Ԫ�أ�����ԭ�ӵĺ˵�������ε����Ҿ�С��18��AԪ��ԭ�Ӻ���ֻ��1�����ӣ�C�ǵؿ��к�������Ԫ�أ�B��C���γ����ֻ�����BC��BC2��B���������������۾���ֵ��ȣ�BC�ж���BC2���������DԪ�ص�����������������Ӳ���������֮һ��E��������Arԭ����ͬ�ĵ��Ӳ�ṹ��
��1��д��A��C����Ԫ�صķ��ţ�A �� C ��
��2��д��Dԭ�Ӻ�E�����ӵĽṹʾ��ͼ��Dԭ�� ��E������ ��
��3����A��B��C��D����Ԫ����ɵij���������Ļ�ѧʽΪ ��
����뷽��ʽΪ ��
��4��д��A��C��D����Ԫ����ɵij����������ϡ��Һ�������º�E�ĵ��ʷ�Ӧ�Ļ�ѧ����ʽ ��
��1��H�� O��
��2�� 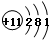

��3��NaHCO3 �� NaHCO3 = Na+ + HCO3����
��4��Cl2 + 2NaOH =" NaCl" + NaClO +H2O��
�������������AԪ��ԭ�Ӻ���ֻ��1�����ӣ�����ΪHԪ�أ�C�ǵؿ��к�������Ԫ�أ�ΪOԪ�أ�B��C���γ����ֻ�����BC��BC2��B���������������۾���ֵ��ȣ�BC�ж���BC2�������������BΪ̼Ԫ�أ�DԪ�ص�����������������Ӳ���������֮һ��Ϊ��Ԫ�أ�E��������Arԭ����ͬ�ĵ��Ӳ�ṹ������Ϊ��Ԫ�ء���3����A��B��C��D����Ԫ����ɵij���������Ļ�ѧʽΪNaHCO3��HCO3��Ϊ��Ԫ�������ʽ�������ܲ���4�������ͼ���绯��Ӧ��
���㣺����Ԫ�ؼ��仯������ƶϡ�

���в���ǰ36��Ԫ�ص����ʻ�ԭ�ӽṹ���±���
| Ԫ�ر�� | Ԫ�����ʻ�ԭ�ӽṹ |
| R | ��̬ԭ�ӵ��������3��δ�ɶԵ��ӣ��������2������ |
| S | ��������ˮ���ҷ�Ӧ��������Һ�������� |
| T | ��̬ԭ��3d�������1������ |
| X | 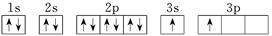 |
��1��RԪ�صĵ�һ������Ҫ������ͬ�������ڵ�Ԫ�أ�ԭ����____________________________________��
Rԭ�ӵĽṹʾ��ͼ�� ��
��2��SԪ�ص�������ϼ�Ϊ________��ԭ����________________________________________��
��3��TԪ��ԭ�ӵļ۵��ӹ���Ϊ________��
��4��X�ĵ����Ų�ͼΥ����____________����X���ʡ�������μ����������εȿ����������ȼ��ʱ�����������ɫ�Ĺ⣬����ԭ�ӽṹ��֪ʶ���ͷ����ԭ��_________________________________��
a��b��c��d��e��f��gΪ�����ɶ�������Ԫ�ع��ɵ����ӣ����Ƕ���10�����ӣ���ṹ�ص����£�
| ���Ӵ��� | a | b | c | d | e | f | g |
| ԭ�Ӻ��� | ���� | ���� | ˫�� | ��� | ���� | ��� | ��� |
| ���������λ��ɣ� | 0 | 1+ | 1- | 0 | 2+ | 1+ | 0 |
����b�����Ӱ뾶����e�����Ӱ뾶��d���ɼ��Լ����ɵ���ԭ�ӷ��ӣ�c��f��Ӧ���γ�����g���ӡ���д����
��1��a���ӵĺ�������Ų�ʽ______________________________��
��2��b��e��ӦԪ�ص�����������Ӧˮ����ļ���ǿ��Ϊ��________��________ (�û�ѧʽ��ʾ)��
��3��d����ˮ�ĵ��뷽��ʽ__________________________________________��
��4��c������__________��f������__________���û�ѧʽ��ʾ����
��5��g���ӵĹ��ۼ�����_____�����Ǹ��࣬˵�����ۼ����� �ԡ�
X��Y��Z��W���ֳ���Ԫ�أ�����X��Y��ZΪ������Ԫ�ء��й���Ϣ���±���
| | ԭ�ӻ���������Ϣ | ���ʼ��仯���������Ϣ |
| X | ZX4�������ɴ�Z�ᴿZ���м���� | X������������Ӧ��ˮ����Ϊ��������ǿ�� |
| Y | Yԭ�ӵ��������������ڵ��Ӳ��� | Y���������ǵ��͵��������������������һ�ּ���ǰ;�ĸ��²��� |
| Z | Zԭ�ӵ������������Ǵ�����������1/2 | Z�����ǽ������ϵ����ǣ��䵥������ȡ���ģ���ɵ�·����Ҫԭ�� |
| W | Wԭ�ӵ�����������С��4 | W�ij������ϼ���+3��+2��WX3ϡ��Һ�ʻ�ɫ |
��1��W�����ڱ���λ��Ϊ ��W��OH��2�ڿ����в��ȶ������ױ��������ɰ�ɫѸ�ٱ�ɻ���ɫ������ɺ��ɫ����Ӧ�Ļ�ѧ����ʽΪ ��
��2��X�ļ������ӵĽṹʾ��ͼΪ ��X������������Ӧˮ�����ˮ��Һ��Y�������ﷴӦ�����ӷ���ʽΪ ��
��3��Z����������ͨѶ���������� ����ҵ���Ʊ�Z�ĵ��ʵĻ�ѧ��Ӧ����ʽΪ ������Z��ͬһ����Ԫ�أ��Ž��з���Ԥ������һԪ�صĴ��ڣ�����������뵼�徧��ܣ������о��������л���������ԵĿ��������ԣ����NaOH ��Һ��Ӧ������H2O2����ʱ����NaOH ��Һ��Ӧ���������Σ��䷽��ʽΪ ��
��4����50 mL l mol��L-1��YX3��Һ����μ���0��5 mol��L-1��NaOH��Һ���õ�1��56 g�����������NaOH��Һ��������� ���������һ�������
���в��ֶ�����Ԫ�ص����ʻ�ԭ�ӽṹ���±���
| Ԫ�ر�� | Ԫ�����ʻ�ԭ�ӽṹ |
| T | ��������ˮ���ҷ�Ӧ������ǿ������Һ�к������ֵ�������ͬ������������ |
| X | �������������ڲ��������3�� |
| Y | ��������Ԫ�صļ������а뾶��С |
| Z | T��X��Z��ɵ�36���ӵĻ�����A�Ǽ�������������Ҫ�ɷ� |
��2��������A����Һ�ʼ��ԣ������ӷ���ʽ������ԭ�� ���û��������������õ����� ��
��3����Y��Z�γɵĻ������ˮ��Һ��μ��뵽T������������Ӧˮ�������Һ��ֱ���������ߵα�����д���˹����з�����Ӧ�����ӷ���ʽ
�� ��
X��Y��Z��W���ֳ���Ԫ�أ�����X��Y��ZΪ������Ԫ�ء��й���Ϣ���±���
| | ԭ�ӻ���������Ϣ | ���ʼ��仯���������Ϣ |
| X | ZX4�������ɴ�Z�ᴿZ���м���� | X������������Ӧ��ˮ����Ϊ��������ǿ�� |
| Y | Yԭ�ӵ��������������ڵ��Ӳ��� | Y���������ǵ��͵��������������������һ�ּ���ǰ;�ĸ��²��� |
| Z | Zԭ�ӵ������������Ǵ�����������1/2 | Z�����ǽ������ϵ����ǣ��䵥������ȡ���ģ���ɵ�·����Ҫԭ�� |
| W | Wԭ�ӵ�����������С��4 | W�ij������ϼ���+3��+2��WX3ϡ��Һ�ʻ�ɫ |
��2��X�ļ������ӵĽṹʾ��ͼΪ ��X������������Ӧˮ�����ˮ��Һ��Y�������ﷴӦ�����ӷ���ʽΪ ��
��3��Z����������ͨѶ���������� ����ҵ���Ʊ�Z�ĵ��ʵĻ�ѧ��Ӧ����ʽΪ ������Z��ͬһ����Ԫ�أ��Ž��з���Ԥ������һԪ�صĴ��ڣ�����������뵼�徧��ܣ������о��������л���������ԵĿ��������ԣ����NaOH ��Һ��Ӧ������H2O2����ʱ����NaOH ��Һ��Ӧ���������Σ��䷽��ʽΪ ��
��4����50 mL l mol·L-1��YX3��Һ����μ���0.5 mol·L-1��NaOH��Һ���õ�1.56 g�����������NaOH��Һ��������Ϊ mL��
X��Y��Z��W�Ƕ����ڵ�����Ԫ�أ��й����ǵ���Ϣ���±���ʾ��
| Ԫ�� | ���ֽṹ֪ʶ | �������� |
| X | Xԭ�Ӻ������ռ��9��ԭ�ӹ�� | X��һ�����������γ��������Ҫ���� |
| Y | Yԭ�ӵĴ�������������������������һ�� | Y���γɶ�����̬�⻯�� |
| Z | Zԭ�ӵ���������������4 | ZԪ�ص���������ϼ���������ϼ۴����͵���6 |
| W | Wԭ�ӵ���������������2n-3��nΪԭ�Ӻ�����Ӳ����� | ��ѧ��Ӧ��Wԭ����ʧȥ���������γ�Wn+ |
��д���пհף�����ʾ����������ĸX��Y��Z��W����
��1��X�ж���ͬ�������壬д��X2����ʽ�� ��
��2��X��Y��Z����Ԫ�ص縺���ɴ�С˳��Ϊ_____��
��3��WԪ��ԭ�ӻ�̬ʱԭ�ӵĵ����Ų�ʽ_________________��
��4��Zλ��Ԫ�����ڱ��� ���ڵ� ��
��5����W��һ���������dz��õľ�ˮ��������ʱ����Һ��pH ����������������������������ǣ� �������ӷ���ʽ��ʾ����
��6��.25�桢101 kPaʱ��32 g Y����ͼ���̬�⻯����ȫȼ�������ȶ���������ʱ�ų�1780��6 kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ ��