��Ŀ����
14��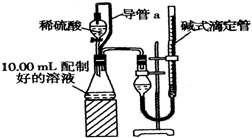 ʵ���Ҳⶨ̼���ƺ�̼�����ƻ������̼���Ƶ���������W��Na2CO3������ȡ�˻����5.0g������ˮ�У����250mL��Һ
ʵ���Ҳⶨ̼���ƺ�̼�����ƻ������̼���Ƶ���������W��Na2CO3������ȡ�˻����5.0g������ˮ�У����250mL��Һ����һ�������������û�ѧ��Ӧ��HCO3-��CO32-��ȫת��Ϊ��������������������������ɴ˼��������Цأ�Na2CO3��
��1����ȡ100mL���ƺõ���Һ���ձ��У��μ�����������������Һ��HCO3-��CO32-��ȫת��Ϊ������Ӧѡ�õ��Լ���D���ţ�
A��CaCl2��Һ B��MgSO4��Һ C��NaCl��Һ D��Ba��OH��2��Һ
��2�����ˣ���ȡ����������˲�������Ҫ�IJ����������ձ��Ͳ������⣬������ͨ©����©����
��3��������ϴ�ӣ�����ָ����������������Ϊmg���ɴ˿��Լ��㣨Na2CO3��������˲��У�����δ�����־ͳ��������æأ�Na2CO3��ƫС���ƫ����ƫС������Ӱ�조��
������������������ȡ10.00mL���ƺõ���Һ������ϡ������Һ��Ӧ���ⶨ����������ͨ��״����Լ20�桢1.01��105Pa����������ɴ˼���������W��Na2CO3��
��1��װ���е���a��������ƽ��ѹǿ��ʹҺ��˳�����£���������ϡ�����������������������ȥ����a��ʹ����������ƫ��ƫ����ƫС������Ӱ�족��
��2����Ӧ������Ϊ��ȷ�ز�������������������ڶ���ʱӦע�⣺
�ٴ���ȴ�����²ſ�ʼ�����ڶ���ǰ����Һ����ƽ���۾�������Һ����ʹ���ƽ
��3��ʵ��ǰ���ʽ�ζ�����Һ������ֱ�ΪV1 mL��V2mL�������CO2�����Ϊ��V1-V2��ml
���������ζ�������ȡ25.00mL���ƺõ���Һ������ƿ�У��μ�2�η�̪�Լ���ҡ�ȣ���0.2000mol/L������ζ����յ㣨��֪�յ�ʱ��ӦH++CO32-=HCO3-ǡ����ȫ����ʱ��ҺPH��8�����ظ��˲���2�Σ�������������ƽ��ֵΪ20.00mL
��1����ȡ25.00mL���ƺõ���Һ��Ӧѡ���ʽ�ζ������������
��2���жϵζ��յ���������ɺ�ɫͻ��Ϊ��ɫ����30s���ָ�
��3���˷���æأ�Na2CO3��=84.8%
���� ����һ����1���μ�����������������Һ��HCO3-��CO32-��ȫת��Ϊ�������ؼ��Ǹó������ܹ���̼��������ӷ�Ӧ���ɳ�����
��2�����ݹ��˲����ķ����ж���Ҫʹ�õ��������ƣ�
��3������δ�����־ͳ����������ֵƫ��������������ȵ�������̼�����Ʋ����ij�������̼���Ʋ����ij������ݴ��ж϶Բⶨ�����Ӱ�죻
����������1������̼�������ᷴӦ����������̼���壬ƿ��ѹǿ��������װ���е���a��������ƽ��ѹǿ��ʹҺ��˳�����£���������ϡ��������������
��2�������DZ�����������ѹ��ͬ������ע������Ϊ���ٴ���ȴ�����²ſ�ʼ�������ڶ���ǰ����Һ����ƽ��
��3����ʽ�ζ�����ǰ��������Ƕ�����̼��������ݴ˼����������̼�������
����������1��̼���ơ�̼�����ƵĻ��Һ�Լ��ԣ�Ӧ���ü�ʽ�ζ�����ȡ��
��2����̪�ı�ɫ��Χ��8.2��10.0����Ӧ����ʱ��Һ�ɺ�ɫͻ��Ϊ��ɫ���ݴ��жϵζ��յ㣻
��3�����ݷ�ӦH++CO32-=HCO3-�����50 mL��Ʒ��Һ�к��е�̼���Ƶ����ʵ�����Ȼ������250mL��Ʒ��Һ�к��е�̼���Ƶ����ʵ������ٸ���m=nM�����̼���Ƶ����������������������ı���ʽ�������Ʒ��̼���Ƶ�����������
��� �⣺����һ����1��A��CaCl2��Һֻ����̼�����Ӧת��Ϊ̼�����������A����
B��MgSO4��Һ��HCO3-��CO32-������Ӧ����B����
C��NaCl��Һ��HCO3-��CO32-������Ӧ����C����
D��Ba��OH��2��Һ��HCO3-��CO32-��������Ӧ����̼�ᱵ��������D��ȷ��
�ʴ�Ϊ��D��
��2�����˲�������Ҫ�IJ��������У��ձ�������������ͨ©�������Գ��ձ��⣬���в���������ͨ©����©����������˲�������Ҫ�IJ����������ձ��Ͳ����������ͨ©����©������
�ʴ�Ϊ����ͨ©����©������
��3������˲��У�����δ�����־ͳ����������ֵƫ��������������ȵ�������̼�����Ʋ����ij�������̼���Ʋ����ij����������������ֵƫ����̼�����Ƶ�����ƫ����̼���Ƶĺ���ƫС���ʴ�Ϊ��ƫС��
����������1������̼�������ᷴӦ����������̼���壬ƿ��ѹǿ��������װ���е���a��������ƽ��ѹǿ��ʹҺ��˳�����£���������ϡ������������������ȥ����a��ʹ����������ƫ��
�ʴ�Ϊ��ƽ��ѹǿ��ʹҺ��˳�����£���������ϡ������������������ƫ��
��2�������DZ�����������ѹ��ͬ������ע������Ϊ���ٴ���ȴ�����²ſ�ʼ�������ڶ���ǰ����Һ����ƽ����Ҫ��֤���۾�������Һ����ʹ���ƽ��
�ʴ�Ϊ������ȴ�����²ſ�ʼ����������ǰ����Һ����ƽ��
��3����ʽ�ζ�����ǰ��������Ƕ�����̼������������CO2�����Ϊ����V1-V2��mL��
�ʴ�Ϊ����V1-V2����
����������1����Һ�Լ��ԣ����Ӧ���ü�ʽ�ζ�����ȡ��
�ʴ�Ϊ����ʽ�ζ��ܣ�
��2����̪�ı�ɫ��Χ��8.2��10.0�������жϵζ��յ���������ɺ�ɫͻ��Ϊ��ɫ����30s���ָ���
�ʴ�Ϊ���ɺ�ɫͻ��Ϊ��ɫ����30s���ָ���
��3����Ӧ�ﵽ�յ�ʱ�����������ӷ���ʽΪ��H++CO32-=HCO3-��
�������������������֪20.00ml��֪������̼���Ƶ����ʵ���Ϊ��0.2000 mol/L��0.02000L=0.004mol��
��ԭ�������̼���Ƶ����ʵ���Ϊ��0.004mol��$\frac{250mL}{25mL}$=0.04mol������Ϊ��0.04mol��106g/mol=4.24g��
����̼������������Ϊ��$\frac{4.24g}{5.0g}$��100%=84.8%��
�ʴ�Ϊ��84.8��
���� ���⿼��̼���ƺ�̼�����ƻ������̼���ƺ����ⶨ��ʵ�鷽����������ۣ���Ŀ�Ѷ��еȣ���ȷ��ѧʵ���������������̼���ơ�̼�����Ƶ�����Ϊ������ؼ��������ֿ�����ѧ���ķ�����������������ѧʵ�顢��ѧ����������

 Сѧ���AB��ϵ�д�
Сѧ���AB��ϵ�д�| A�� | 2һ��һ2һ��ϩ | B�� | 3��3һ����һ2һ���� | ||
| C�� | 4һ������ | D�� | 2��3��4һ�������� |
| A�� | ��ҵ�Ͻ����Ṥҵβ��SO2����ͨ�백ˮ�У�SO2+OH-�THSO3- | |
| B�� | ��NaAlO2��Һ��ͨ�����CO2��AlO2-+CO2+2H2O�TAl��OH��3��+HCO3- | |
| C�� | �ö��Ե缫���KOH��Һʱ�����缫��Ӧ��2H2O-4e-�TO2��+4H+ | |
| D�� | NaHSO4��Һ��Ba��OH��2��Һ��ַ�Ӧ����Һ�����ԣ�Ba2++2OH-+2H++SO42-�TBaSO4��+2H2O |
| A�� | �ö��Ե缫��ⱥ��ʳ��ˮʱ�������ĵ缫��ӦʽΪ��2Cl-�TCl2��+2e- | |
| B�� | ����ȼ�ϵ�صĸ�����Ӧʽ��O2+2H2O+4e-�T4OH- | |
| C�� | ��ͭ����ʱ�����Դ�����������Ǵ�ͭ���缫��ӦʽΪ��Cu�TCu2++2e- | |
| D�� | ���������绯ѧ��ʴ��������Ӧʽ��Fe�TFe2++2e- |
| A�� | C3H8O | B�� | C2H6O | C�� | C2H4O2 | D�� | C4H10 |
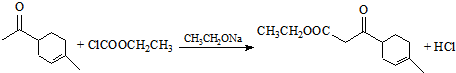
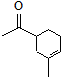 ��
��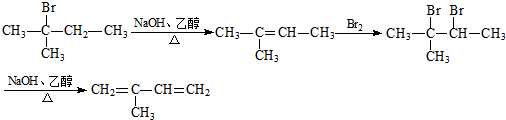 ��
��