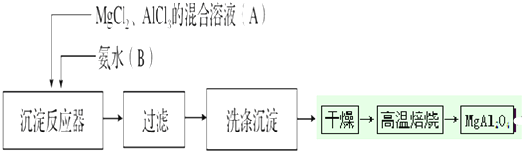
��Ŀ����
12��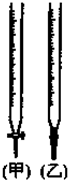 ���к͵ζ����ⶨ�ռ�Ĵ��ȣ����ռ��к��������Ӧ�����ʣ��Ը���ʵ��ش�
���к͵ζ����ⶨ�ռ�Ĵ��ȣ����ռ��к��������Ӧ�����ʣ��Ը���ʵ��ش���1��ȡ10.00mL����Һ��������ȡ������ס����ҡ���
��2����0.20mol•L-1������ζ������ռ���Һ���ζ�ʱ������ת��ʽ�ζ��ܵIJ������������ֲ�ͣ��ҡ����ƿ������ע����ƿ����Һ��ɫ�ı仯��ֱ���ζ����յ㣮
��3���Ա���������Һ�ζ�δ֪Ũ�ȵ�����������ҺΪ�����ж����в�����������ƫ����ƫС������Ӱ�족�����жϡ���
�ٶ������ζ�ǰƽ�ӣ��ζ�����ƫС
��δ�ñ�Һ��ϴ�ζ���ƫ��
���ô���Һ��ϴ��ƿ��Ӱ�죮
���� ��1�����ݼ�����Һʢ���ڼ�ʽ�ζ����У�
��2�����ݵζ�ʱ������ע����ƿ����Һ��ɫ�ı仯��
��3������C�����⣩=$\frac{C��������V������}{V�����⣩}$����ϴ�������Ա���Һ�����Ӱ�������
��� �⣺��1������Һ�Ǽ�����Һ��Ӧʢ���ڼ�ʽ�ζ����У��ʴ�Ϊ���
��2���ζ�ʱ������ע����ƿ����Һ��ɫ�ı仯���Ա�ȷ�ж��յ�ĵ���ʴ�Ϊ����ƿ����Һ��ɫ�ı仯��
��3���ٶ������ζ�ǰƽ�ӣ��ζ����ӣ����±�Һ�����ƫС������C�����⣩=$\frac{C��������V������}{V�����⣩}$������֪C�����⣩ƫС���ʴ�Ϊ��ƫС��
��δ�ñ�Һ��ϴ�ζ��ܣ���ζ����к�����������ˮ������Һ��Ũ��ƫС���ζ�ʱ���ĵı�Һ�����ƫ����C�����⣩=$\frac{C��������V������}{V�����⣩}$������֪C�����⣩ƫ�ʴ�Ϊ��ƫ��
���ô���Һ��ϴ��ƿ�������Һ�����ʵ������䣬���ĵı�Һ��������䣬����C�����⣩=$\frac{C��������V������}{V�����⣩}$������֪C�����⣩���䣬�ʴ�Ϊ����Ӱ�죮
���� ������Ҫ�������к͵ζ����������������ѶȲ���ע������к͵ζ�ʵ���������������

���þƾ�ϴ���Թ��ڱ��ϲ����ĵ���þƾ�ϴ���Լ�ƿ�в����ı��Ӣ���ϡ��ˮϴ���Թܱ������������ȼ�Һϴ���Թ��ڱ��ϵ���֬��
| A�� | �٢ۢ� | B�� | �ڢۢ� | C�� | �٢ڢ� | D�� | �٢ڢۢ� |
a��Na2S2O3��BaS2O3��BaS��������ˮ��
b��SO2��Na2S��Na2CO3��Ӧ������Na2S2O3��
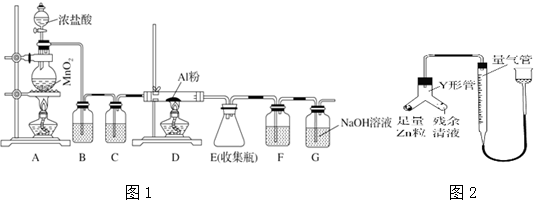
ij��ѧС��ݴ˽������Ʊ���������ƣ�Na2S2O3����̽����ʵ���Ʊ�װ����ͼ��ʾ��ʡ�Լг�װ�ã���

�ش����⣺
��1������a�������Ƿ�Һ©����
��2��װ��A�з�����Ӧ�Ļ�ѧ����ʽ��Na2SO3+H2SO4=Na2SO4+SO2��+H2O��
��3��װ��B������֮һ�ǹ۲�SO2���������ʣ�
��B�����ʢװ��Һ����c��
a������ˮ b������Na2SO3��Һ c������NaHSO3��Һ d������NaHCO3��Һ
����ʹSO2����������ƿC����ȷ�IJ����ǿ��Ʒ�Һ©���Ļ�����ʹŨ���Ỻ������A�У�
��4����װ��C������Na2S2O3��
����ɷ�Ӧ����ʽ��4SO2+2Na2S+1Na2CO3=3Na2S2O3+CO2
�ڷ�Ӧ��ʼ��ʹA�з�����Ӧһ�������ʹC�з�Ӧ��������ԭ����A�з�Ӧһ�����ɶ�������C�в��ܷ�Ӧ��
�۽�����Ӧ��ȡC����Һ�����������ᾧ�����ˡ�ϴ�ӡ�����õ�Na2S2O3•5H2O��
��5����ɶ����ò�Ʒ�ļ���ʵ�飺
| �Ʋ� | ���������� | ���� |
| �����е����γɷֿ����У� Na2S Na2CO3 Na2SO3 Na2SO4 | ��ȡWg��Ʒ���ϡ��Һ�� ������Һ�еμӹ���BaCl2��Һ���а�ɫ�������ɣ����ˣ��ó�������Һ�� ��������м���������ᣬ������ȫ�ܽ⣬���д̼�����ζ����������� ������Һ�μ�2�ε�����Һ������μ�0.100 0mol•L-1��ı���Һ������Һ����ɫ������ȥ�����ĵ�ı���Һ���Ϊ18.10mL�� | ��Ʒ�����У� һ������Na2SO3�� ���ܺ���Na2S��Na2CO3�� һ��������Na2SO4�� �����ܷ����ķ�Ӧ�ǣ� 2S2O${\;}_{3}^{2-}$+I2�TS4O${\;}_{6}^{2-}$+2I- ��Ʒ���ȣ�$\frac{9}{w}$��100%�� |
��1�����ƴ�����Һ����4.21g���������������ᷴӦ�����ʵ�NaOH�������250.00mL����Һ�����õ����������ձ�������������ͷ�ιܡ�250mL ����ƿ��
��2����0.4mol•L-1�ı�������Һע��ྻ����ʽ�ζ����У�ʹҺ��̶��ڡ�0�����ϣ��̶���������ת����ʹ�ζ��ܼ��첿�ֳ�����Һ��Ȼ�����Һ�汣����0�̶Ȼ�0�̶����£����¶������ü�ʽ�ζ�������ƿ�м���20.00mL������Һ�����Ӽ��μ�����ָʾ����
��3��ʵ������
| ������ | ����Һ�������mL�� | ����0.4000mol•L-1����������mL�� | |
| �ζ�ǰ��mL�� | �ζ���mL�� | ||
| 1 | 20.00 | 2.10 | 22.00 |
| 2 | 20.00 | 0.90 | 21.00 |
��4�������������ζ�ǰ������ʽ�ζ��ܶ������յ������ȷ�����ý��ƫ�ͣ�ƫ�ߡ�ƫ�ͣ�
| A�� | ��ʽ�ζ���ֻ��ˮϴ | |
| B�� | ��ƿֻ��ˮϴ | |
| C�� | ʹ�ü�����ָʾ�� | |
| D�� | ��һ�ι۲���ʽ�ζ��ܵĿ̶�ʱ���� |
 ijУ��ѧ��ȤС��̽��SO2��FeCl3��Һ�ķ�Ӧ������װ������ͼ��ʾ���г���������ȥ����
ijУ��ѧ��ȤС��̽��SO2��FeCl3��Һ�ķ�Ӧ������װ������ͼ��ʾ���г���������ȥ������1��ʵ��ǰ��Ӧ�ȼ��װ�õ������ԣ�ʵ���в�����β��Ӧͨ��NaOH��Һ��
��2��ʵ���������Ҫ����100mL1mol/L FeCl3��Һ��δ��Ũ�����ữ��������Ҫ�IJ��������У��ձ�����Ͳ������������ͷ�ιܡ�100mL������ƿ��
��3����С��ͬѧ��5mL lmol/L FeCl3��Һ��ͨ��������SO2����Һ���ճ�dz��ɫ���ٴ�Һ©����������μ���NaOHϡ��Һ�����Թ�B�в�����ʵ�������ǿ�ʼ������Ȼ�������ɫ������
��4����С��ͬѧ�ڣ�3����ʵ��ʱ��������Һ��Ϊdz��ɫ��Ҫ�ϳ�ʱ�䣬�ڴ��ڼ�ͬѧ�ǹ۲쵽����������Һ���ػ�ɫ��ɺ���ɫ��û�й۲쵽���������������Һ��dz��ɫ��
���������ϡ�Fe�� HSO3��2+��������Һ�гʺ���ɫ�Ҿ��н�ǿ�Ļ�ԭ�ԣ��ܱ�Fe3+����ΪSO${\;}_{4}^{2-}$��Fe��HSO3��2+��Fe3+����Һ�з�Ӧ�����ӷ���ʽ��Fe3++H2O+Fe��HSO3��2+�T2Fe2++SO42-+3H+��
��5��Ϊ��̽��������̺���ɫ��Ϊdz��ɫ��ʱ�䣬��С��ͬѧ����������ʵ�飺
| ����� | ��5mL��1mol•LFeCl3��Һ��ͨ��SO2���壬��Һ������Ϊ����ɫ�����ڽ϶�ʱ������Һ��ɫ��Ϊdz��ɫ�� |
| ����� | ��5mL�������Ƶ�1mol•L-1��FeCl3��Һ����Ũ�����ữ����ͨ��SO2���壬��Һ������Ϊ����ɫ����ʱ���ڷ�����Һ��ɫ���dz��ɫ�� |
��6���ۺ�����ʵ��̽�����̣����Ի�õ�ʵ����ۣ�
I��SO2��FeCl3��Һ��Ӧ���ɺ���ɫ�м����Fe��HSO3��2+���ӣ�
����ɫ�м����ת���dz��ɫ��Һ��һ�������Ĺ��̣�
���ȡ����FeCl3��Һ�����Ի�����dz��ɫ���ֵ�ʱ�䣮
| A�� | һCH3�������ĵ���ʽΪ 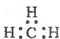 | B�� | �������Ľṹ��ʽ��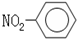 | ||
| C�� | ��ȩ�Ľṹʽ��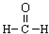 | D�� | �Ҵ��ķ���ʽ��CH3CH2OH |
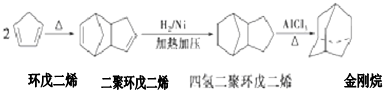
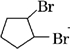 +2NaOH$��_{��}^{��}$
+2NaOH$��_{��}^{��}$ +2NaBr��
+2NaBr��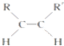 $\stackrel{O_{2}}{��}$ $\stackrel{H_{2}O/Zn}{��}$RCHO+R��CHO����д�����з�Ӧ����Ľṹ��ʽ��
$\stackrel{O_{2}}{��}$ $\stackrel{H_{2}O/Zn}{��}$RCHO+R��CHO����д�����з�Ӧ����Ľṹ��ʽ�� $\stackrel{O_{2}}{��}$ $\stackrel{H_{2}O/Zn}{��}$
$\stackrel{O_{2}}{��}$ $\stackrel{H_{2}O/Zn}{��}$ ��
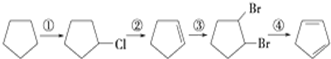
�� ���������DZ����ϵ�ϩ�����������Ը��������Һ�������Ȼ���д��A���п��ܵĽṹ��ʽ�������������칹����
���������DZ����ϵ�ϩ�����������Ը��������Һ�������Ȼ���д��A���п��ܵĽṹ��ʽ�������������칹����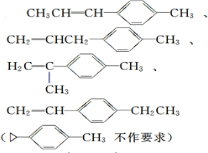 ��
��